Beyond Hartree-Fock: A Practical Guide to Post-Hartree-Fock Methods for Accurate Molecular Calculations in Drug Discovery
This article provides a comprehensive overview of post-Hartree-Fock (post-HF) methods, essential for achieving high-accuracy quantum chemical predictions in molecular calculations.
Beyond Hartree-Fock: A Practical Guide to Post-Hartree-Fock Methods for Accurate Molecular Calculations in Drug Discovery
Abstract
This article provides a comprehensive overview of post-Hartree-Fock (post-HF) methods, essential for achieving high-accuracy quantum chemical predictions in molecular calculations. Tailored for researchers and drug development professionals, it covers the foundational theory behind electron correlation, details key methodological approaches from MP2 to CCSD(T), and addresses practical challenges and optimization strategies for their application. The scope extends to troubleshooting computational bottlenecks, validating results against benchmarks, and exploring emerging trends, including the integration of machine learning and the prospective role of quantum computing, offering a vital resource for leveraging these powerful tools in biomedical research.
The Quest for Accuracy: Understanding Electron Correlation and the Limits of Hartree-Fock
The Hartree-Fock (HF) method serves as the foundational approximation in quantum chemistry for solving the electronic structure of molecules. However, its mean-field approach, where each electron experiences only the average electrostatic field of all other electrons, inherently neglects the instantaneous repulsive interactions between electrons [1] [2]. This neglected component of the electron-electron interaction is what defines the electron correlation problem. The correlation energy is formally defined as the difference between the exact, non-relativistic energy of a system and its Hartree-Fock energy: ( E{\text{corr}} = E{\text{exact}} - E_{\text{HF}} ) [3] [2] [4]. Although this energy difference typically constitutes a small fraction (around 1%) of the total electronic energy, its contribution is crucial for achieving chemical accuracy in computational predictions, as it directly influences molecular properties, reaction energetics, and the description of chemical bonding [3] [2].
The limitations of the HF method become particularly evident in specific chemical scenarios. For instance, the dissociation of the H₂ molecule is poorly described by restricted Hartree-Fock (RHF), which fails to correctly separate the molecule into two hydrogen atoms [5]. Similarly, the HF approximation often fails to predict the stability of anions where the binding mechanism relies on electron correlation effects, such as in the case of the C₂⁻ anion [5]. These qualitative and quantitative failures underscore the necessity for post-Hartree-Fock methods, which are designed to recover a significant portion of the missing correlation energy [1] [6].
Defining Electron Correlation
Physical Interpretation and the Coulomb Hole
Electron correlation describes the interaction between electrons in a quantum system, specifically how the motion of one electron is influenced by the instantaneous positions of all others [4]. In the HF approximation, the probability of finding two electrons at a given separation is effectively overestimated at small distances and underestimated at large distances because the model does not account for their mutual Coulombic repulsion [4]. The concept of the Coulomb hole visually represents this deficiency. It is defined as the difference in the intracule density distribution (the probability distribution of interelectronic distances) between a correlated wavefunction and the Hartree-Fock wavefunction [3]. This hole illustrates how correlated electrons "avoid" each other more effectively than the HF model predicts.
Dynamic and Static Correlation
Electron correlation is broadly categorized into two types, each with distinct physical origins and requiring different theoretical treatments [2] [4].
- Dynamic Correlation: This arises from the short-range, instantaneous repulsive interactions between electrons as they move. It is a ubiquitous effect present in all electronic systems and is characterized by rapid fluctuations in electron positions. Methods like Møller-Plesset Perturbation Theory and Coupled-Cluster theory are particularly effective at capturing dynamic correlation [6] [2].
- Static (Non-Dynamical) Correlation: This occurs in systems where the ground electronic state cannot be accurately described by a single Slater determinant. This is common in situations with (near-)degenerate electronic configurations, such as in molecules with stretched or broken bonds (e.g., during dissociation), diradicals, and many transition metal complexes [2] [5] [4]. Static correlation is a qualitative failure of the single-determinant picture and requires a multi-reference description from the outset, as provided by Multi-Configurational Self-Consistent Field (MCSCF) methods [6] [4].
Table 1: Key Characteristics of Electron Correlation Types
| Feature | Dynamic Correlation | Static Correlation |
|---|---|---|
| Physical Origin | Instantaneous electron-electron repulsion [2] | Near-degeneracy of electronic configurations [2] [4] |
| Primary Methods | MP2, CCSD(T), CISD [6] [2] | CASSCF, MCSCF [6] [4] |
| Typical Systems | Closed-shell molecules near equilibrium geometry [2] | Dissociating bonds, diradicals, transition metal complexes [2] [5] |
Post-Hartree-Fock methods comprise a suite of computational approaches developed to address the electron correlation problem. They can be broadly classified into several families based on their underlying theoretical principles.
Wavefunction-Based Correlation Methods
These methods build upon the HF wavefunction by introducing excitations into virtual orbitals.
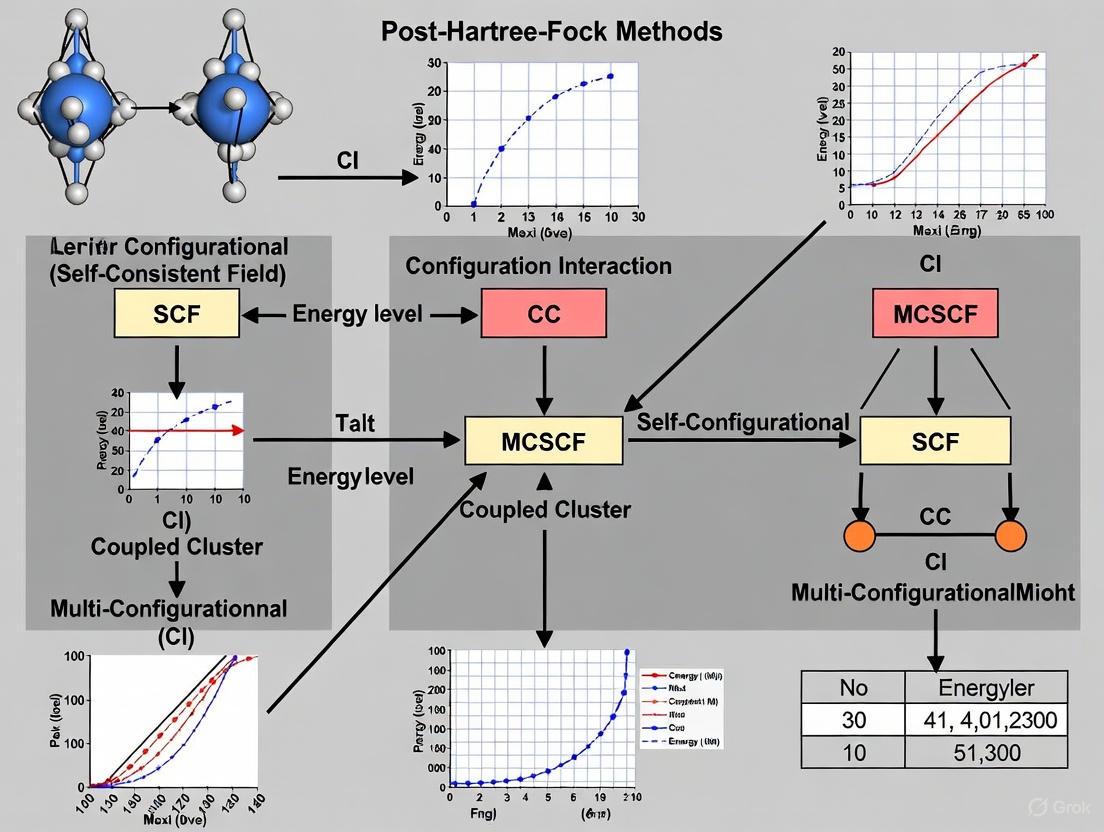
- Configuration Interaction (CI): The CI method expands the total wavefunction as a linear combination of the HF reference determinant and excited-state determinants (e.g., single (S), double (D), etc., excitations) [6] [7]. The coefficients of this expansion are determined variationally. Full CI, which includes all possible excitations, provides the exact solution for a given basis set but is computationally intractable for all but the smallest systems [3] [2]. Truncated methods like CISD are more practical but lack size-consistency, meaning their accuracy deteriorates with increasing system size [6] [7].
- Coupled-Cluster (CC) Theory: This method uses an exponential ansatz for the wavefunction (e.g., ( \Psi{CC} = e^{T} \Phi0 )) to ensure size-consistency [7]. The cluster operator ( T ) generates single, double, triple, etc., excitations. The CCSD method includes singles and doubles, while the CCSD(T) method, often called the "gold standard" of quantum chemistry, adds a perturbative treatment of triples. This method offers excellent accuracy for dynamic correlation but at a high computational cost [6] [7].
- Møller-Plesset Perturbation Theory (MPn): This is a class of non-variational methods that treat electron correlation as a perturbation to the HF Hamiltonian [6]. The second-order correction, MP2, is one of the most widely used post-HF methods due to its favorable balance of cost and accuracy, capturing a significant portion of dynamic correlation [6] [2].
- Multi-Configurational Methods: For systems with strong static correlation, methods like the Multi-Configurational Self-Consistent Field (MCSCF) and its variant Complete Active Space SCF (CASSCF) are employed [2] [4]. These methods optimize both the orbital coefficients and the configuration expansion coefficients within a carefully selected active space of orbitals, providing a qualitatively correct zero-order wavefunction. Dynamical correlation can then be added on top via methods like CASPT2 (CAS with second-order perturbation theory) [6].
Table 2: Comparison of Key Post-Hartree-Fock Methods
| Method | Theoretical Principle | Handles Correlation | Key Advantage | Key Limitation |
|---|---|---|---|---|
| MP2 [6] | Perturbation Theory (2nd order) | Dynamic | Low cost, good scaling [6] | Fails for static correlation, not variational [6] |
| CISD [6] [7] | Variational (Single + Double excitations) | Primarily Dynamic | Simple concept, variational [7] | Not size-consistent [7] |
| CCSD(T) [6] [7] | Exponential cluster operator | Dynamic | High accuracy, size-consistent [7] | Very high computational cost [7] |
| CASSCF [2] [4] | Variational (Multi-reference) | Static | Corrects for near-degeneracy [4] | Choice of active space is non-trivial [2] |
Practical Protocols for Post-HF Calculations
Protocol 1: Assessing Dynamic Correlation with MP2
The MP2 method provides a cost-effective first assessment of electron correlation effects.
- Aim: To obtain an initial estimate of the dynamical correlation energy for a molecular system near its equilibrium geometry.
- Prerequisites: A converged Hartree-Fock calculation.
- Workflow:
- Geometry Optimization: Perform a geometry optimization at the HF level to find a stable molecular structure.
- HF Energy Calculation: Execute a single-point energy calculation at the HF level to obtain the reference energy, ( E{\text{HF}} ).
- MP2 Correlation Energy Calculation: Using the HF orbitals as a basis, compute the second-order Møller-Plesset energy correction, ( E{\text{MP2}} ).
- Total Energy Evaluation: The total MP2 energy is given by ( E{\text{total}} = E{\text{HF}} + E_{\text{MP2}} ).
- Interpretation: The MP2 energy is typically lower than the HF energy. The correlation energy is ( E{\text{corr}}(\text{MP2}) = E{\text{MP2}} ). This protocol is suitable for systems where static correlation is not significant.
Protocol 2: Handling Strong Correlation with CASSCF
For molecules with known multi-reference character (e.g., diradicals, dissociating bonds), a CASSCF calculation is the appropriate starting point.
- Aim: To generate a qualitatively correct reference wavefunction that accounts for static correlation.
- Prerequisites: A foundational understanding of the system's electronic structure to define the active space.
- Workflow:
- Active Space Selection: This is the most critical step. Select a set of active electrons and active orbitals (e.g., 2 electrons in 2 orbitals for a stretched H₂ molecule, or the π-system in butadiene). This is denoted as (ne, no), e.g., (2,2).
- State Specification: Define the number of states and spin symmetry (e.g., singlet, triplet) to be averaged.
- Orbital Optimization: Perform the CASSCF calculation to self-consistently optimize both the molecular orbitals and the configuration coefficients within the active space.
- Analysis: Inspect the resulting natural orbitals and their occupation numbers. Non-integer occupation numbers (e.g., not close to 2 or 0) indicate strong static correlation.
- Interpretation: The CASSCF energy includes static but not dynamic correlation. For quantitative results, a subsequent dynamic correlation method like CASPT2 or MRCI must be applied.
The Scientist's Toolkit: Essential Computational Reagents
Table 3: Key Reagents and Parameters for Post-HF Calculations
| Item | Function/Description | Considerations for Selection |
|---|---|---|
| Basis Set | A set of mathematical functions (e.g., Gaussian-type orbitals) used to represent molecular orbitals [7]. | Larger basis sets (e.g., triple-ζ, quadruple-ζ) improve accuracy but drastically increase cost. Correlation-consistent basis sets (e.g., cc-pVXZ) are designed for post-HF methods [7]. |
| Active Space (for CASSCF) | The set of chemically relevant molecular orbitals and electrons included in the full CI expansion [2]. | Selection requires chemical insight. It should include orbitals involved in bond breaking/forming, frontier orbitals, and unpaired electrons. |
| Reference Wavefunction | The initial guess wavefunction, typically from a converged HF calculation. | For open-shell systems, an Unrestricted HF (UHF) reference can be used, but may suffer from spin contamination. |
| Integral Grid | Numerical grid used for evaluating integrals in post-HF algorithms, particularly in Density Functional Theory (DFT). | A finer grid is needed for higher accuracy, at the cost of increased computation time. |
The Hartree-Fock method, while foundational, is fundamentally insufficient for quantitative quantum chemistry due to its neglect of electron correlation. This limitation manifests in erroneous predictions for bond dissociation energies, properties of anions, and systems with degenerate or near-degenerate electronic states. The development of post-Hartree-Fock methods—including Configuration Interaction, Coupled-Cluster theory, Møller-Plesset Perturbation Theory, and Multi-Configurational approaches—provides a systematic pathway for recovering the missing correlation energy. The choice of an appropriate post-HF method depends critically on the nature of the system and the type of correlation (dynamic or static) that dominates. While these advanced methods come with increased computational cost, they are indispensable tools for achieving the accuracy required for modern molecular design, including applications in drug development and materials science.
In quantum chemistry, the Hartree-Fock (HF) method provides a foundational wave function-based approach for computing molecular electronic structures. It approximates the many-electron wave function as a single Slater determinant, where each electron moves in the average field created by all other electrons [8]. However, this mean-field approximation neglects electron correlation, which refers to the instantaneous, repulsive interactions between electrons. The energy discrepancy arising from this simplification is termed the correlation energy, defined as the difference between the exact non-relativistic energy of a system and its Hartree-Fock energy: ( E{\text{corr}} = E{\text{exact}} - E_{\text{HF}} ) [2]. While this correlation energy is typically a small fraction of the total electronic energy, recovering it is crucial for achieving chemical accuracy in predicting molecular properties, reaction energies, and binding affinities [2].
Electron correlation is conventionally divided into two primary types: dynamic correlation and static correlation. Accurately describing both forms is essential for modeling complex chemical systems, particularly in drug discovery where precise energy calculations underpin the design of novel therapeutics [8]. Post-Hartree-Fock methods comprise a suite of computational strategies developed specifically to address these electron correlation effects, going beyond the limitations of the basic HF approximation [1].
Defining Dynamic and Static Correlation
Dynamic Correlation
Dynamic correlation arises from the instantaneous Coulombic repulsion between electrons, which causes them to avoid each other as they move through space [2]. It reflects the rapid fluctuations in electron positions and represents the cumulative effect of numerous, small electron-electron repulsions that are not captured by the HF mean-field potential. This type of correlation is pervasive in all molecular systems and is particularly significant in systems with weakly interacting electrons [2].
From a computational perspective, dynamic correlation is often described as "short-range" and can be efficiently treated using perturbation theory or coupled cluster methods [2]. For example, in the reaction coordinate of a simple bond formation or cleavage, dynamic correlation accounts for the energy correction needed to properly describe the electron repulsion in the vicinity of the equilibrium bond length.
Static Correlation
Static correlation, also known as non-dynamic or near-degeneracy correlation, occurs when multiple electronic configurations possess similar energies [2]. This phenomenon is prominent in systems with near-degenerate orbitals, such as molecules with stretched or breaking bonds, diradicals, and many transition metal complexes [2]. In these cases, a single Slater determinant (as used in HF theory) provides a qualitatively incorrect description of the electronic structure.
Static correlation is considered a "long-range" effect and requires multi-reference methods for its accurate treatment [2]. For instance, in the dissociation of a H₂ molecule, the HF method fails dramatically as the bond is stretched, whereas a multi-configurational approach that includes both covalent and ionic configurations can correctly describe the dissociation limit.
Table 1: Comparative Analysis of Dynamic and Static Correlation
| Feature | Dynamic Correlation | Static Correlation |
|---|---|---|
| Physical Origin | Instantaneous Coulomb repulsion between electrons [2] | Near-degeneracy of multiple electronic configurations [2] |
| Character | Short-range, local electron avoidance [2] | Long-range, qualitative electronic structure effect [2] |
| Dominant In | Systems with weakly interacting electrons near equilibrium geometry [2] | Stretched bonds, diradicals, transition metal complexes [2] |
| Primary Treatment Methods | Møller-Plesset Perturbation Theory (MP2, MP4), Coupled Cluster (CCSD, CCSD(T)) [9] [1] | Multi-Configurational SCF (MCSCF), Complete Active Space SCF (CASSCF) [2] |
| Impact on Wavefunction | Small corrections to a single-reference wavefunction | Requires wavefunction with multiple dominant configurations |
Methodological Approaches and Protocols
The development of post-Hartree-Fock methods is largely driven by the need to treat dynamic and static correlation with varying degrees of accuracy and computational efficiency. The relationship between these methods can be visualized as a strategic decision tree.
Diagram Title: Method Selection Workflow for Electron Correlation
Protocol 1: Single-Reference Methods for Dynamic Correlation
This protocol details the application of single-reference methods, which are appropriate when dynamic correlation dominates and a single HF configuration provides a qualitatively correct description of the system [2].
A. Møller-Plesset Perturbation Theory (MP2)
Principle: MP2 is a second-order perturbation theory that treats the electron correlation as a small perturbation to the HF Hamiltonian. It is one of the most computationally efficient post-HF methods for capturing dynamic correlation [10] [1].
Procedure:
- Converge a Restricted HF (RHF) calculation: Perform a standard HF calculation to obtain a set of canonical molecular orbitals and orbital energies. Ensure the wavefunction is stable.
- Transform two-electron integrals: Transform the atomic orbital basis two-electron integrals (( g_{pqrs} )) into the molecular orbital basis. This step is often the computational bottleneck.
- Compute the MP2 correlation energy: Calculate the second-order energy correction using the following standard formula: [ E^{(2)} = \frac{1}{4} \sum{ijab} \frac{ | \langle ij || ab \rangle |^2 }{ \epsiloni + \epsilonj - \epsilona - \epsilon_b } ] where ( i,j ) denote occupied orbitals, ( a,b ) denote virtual orbitals, ( \langle ij || ab \rangle ) are antisymmetrized two-electron integrals in the molecular orbital basis, and ( \epsilon ) are the HF orbital energies [1].
- Calculate total energy: The total MP2 energy is given by ( E{\text{MP2}} = E{\text{HF}} + E^{(2)} ).
B. Coupled Cluster Singles and Doubles (CCSD)
Principle: The Coupled Cluster method uses an exponential wavefunction ansatz ( e^{\hat{T}} | \Psi{\text{HF}} \rangle ) to model electron correlation. The cluster operator ( \hat{T} = \hat{T}1 + \hat{T}2 + \ldots ) generates all possible excitations from the reference HF determinant. CCSD includes all single (( \hat{T}1 )) and double (( \hat{T}_2 )) excitations [9] [1].
Procedure:
- Converge a Restricted HF calculation: As with MP2, start with a well-converged RHF wavefunction.
- Set up the coupled cluster equations: The equations are derived by projecting the Schrödinger equation with the exponential ansatz onto the reference and all singly and doubly excited determinants.
- Solve amplitude equations iteratively: Solve the following non-linear equations for the ( t ) amplitudes (excitation strengths) iteratively until self-consistency is achieved: [ \langle \Phi{i}^{a} | \bar{H} | \Phi{\text{HF}} \rangle = 0 \quad \text{and} \quad \langle \Phi{ij}^{ab} | \bar{H} | \Phi{\text{HF}} \rangle = 0 ] where ( \bar{H} = e^{-\hat{T}} H e^{\hat{T}} ) is the similarity-transformed Hamiltonian.
- Compute the CCSD energy: The correlation energy is calculated as ( E{\text{corr}} = \langle \Phi{\text{HF}} | \bar{H} | \Phi{\text{HF}} \rangle - E{\text{HF}} ).
Protocol 2: Multi-Reference Methods for Static Correlation
This protocol is selected when significant static correlation is present, such as in bond-breaking reactions or systems with open-shell transition metals [2].
A. Complete Active Space Self-Consistent Field (CASSCF)
Principle: CASSCF is a specific type of Multi-Configurational SCF (MCSCF) method. It divides molecular orbitals into an inactive space (doubly occupied), an active space (with variable occupancy), and a virtual space (unoccupied). A Full Configuration Interaction (FCI) calculation is performed within the active space, while the orbitals are optimized simultaneously [2].
Procedure:
- Define the Active Space: This is the most critical step. Select ( N ) electrons in ( M ) orbitals to form the active space, denoted as CAS(( N, M )). This space must include all orbitals essential for the chemical process (e.g., bonding/antibonding orbital pairs for bond breaking, d-orbitals in transition metals).
- Perform an initial guess: Obtain initial orbitals, often from an HF calculation.
- Solve the CI problem: Within the active space, construct and diagonalize the CI matrix to obtain the configuration interaction coefficients and the energy for the current set of orbitals.
- Optimize the orbitals: Update the molecular orbitals to minimize the total energy. This step combines aspects of HF orbital optimization with the multi-configurational wavefunction.
- Iterate to convergence: Repeat steps 3 and 4 until both the CI coefficients and the orbitals are self-consistent, yielding the final CASSCF energy and wavefunction.
B. Multi-Reference Perturbation Theory (e.g., CASPT2)
Principle: A CASSCF calculation correctly treats static correlation but often lacks dynamic correlation. CASPT2 adds a second-order perturbation theory correction on top of the CASSCF reference wavefunction to account for dynamic correlation [2].
Procedure:
- Perform a CASSCF calculation: Follow Protocol 2A to obtain a converged multi-reference wavefunction.
- Use the CASSCF wavefunction as the reference: The zeroth-order Hamiltonian is built using the CASSCF orbitals and energies.
- Compute the second-order energy correction: The first-order wavefunction and second-order energy are calculated by considering excitations out of the entire CASSCF reference space. This step is computationally demanding but captures the bulk of the dynamic correlation missing in CASSCF.
- Calculate the total energy: The final total energy is ( E{\text{CASPT2}} = E{\text{CASSCF}} + E^{(2)} ).
Table 2: Key Reagent Solutions for Post-Hartree-Fock Calculations
| Research Reagent / Resource | Function and Description |
|---|---|
| Gaussian Basis Sets (e.g., cc-pVDZ, cc-pVTZ) | Pre-optimized sets of atom-centered Gaussian functions used to expand molecular orbitals. Larger "triple-zeta" basis sets offer better resolution but increase cost [10]. |
| Active Space (for CASSCF) | A carefully selected set of molecular orbitals and electrons in which a full configuration interaction is performed. It is the central "reagent" for treating static correlation [2]. |
| Pseudopotentials / Effective Core Potentials (ECPs) | Replace the core electrons of heavy atoms with an effective potential, reducing computational cost while maintaining accuracy for valence electron effects. |
| Two-Electron Integrals (( g_{pqrs} )) | The computational representation of electron-electron repulsion, calculated over quartets of basis functions. They are fundamental inputs for all correlated methods [9] [10]. |
| Quantum Chemistry Software (e.g., Gaussian, Q-Chem, ORCA, Molpro) | Software packages that implement the complex algorithms for SCF, integral transformation, and post-HF solvers, providing user-friendly interfaces [8]. |
Application in Drug Discovery
The accurate treatment of electron correlation is not merely an academic exercise but has profound implications in drug discovery, where predicting molecular properties and interactions with high fidelity is essential [8].
QM/MM Simulations: In enzyme catalysis, the reactive site (e.g., a covalent inhibitor forming a bond with a catalytic residue) often exhibits strong static correlation, necessitating a multi-reference QM method (like CASSCF) for the active site. This QM region is embedded within a larger protein environment treated with molecular mechanics (MM), balancing accuracy and computational feasibility [8].
Binding Affinity Prediction: The strength of non-covalent interactions between a drug candidate and its protein target—such as hydrogen bonding, π-π stacking, and dispersion forces—is heavily influenced by dynamic correlation. Methods like MP2 or CCSD(T) within a QM/MM framework can provide superior accuracy compared to HF or pure MM force fields, especially for "undruggable" targets with metalloenzymes or unusual bonding [8].
Reaction Mechanism Elucidation: For covalent inhibitors, the process of bond formation and breaking along the reaction pathway involves a transition from static to dynamic correlation dominance. Multi-reference methods are critical for correctly modeling the transition state and reaction energy barrier, guiding the rational design of more effective and selective inhibitors [8].
Post-Hartree-Fock (post-HF) methods encompass a suite of computational quantum chemistry approaches designed to overcome the central limitation of the Hartree-Fock (HF) approximation: the neglect of electron correlation. The HF method, while providing a qualitative description of electronic structure, treats electron-electron interactions only in an average sense and fails to capture the correlated motion of electrons. This missing electron correlation energy is crucial for quantitative predictions of molecular properties, reaction energies, and spectroscopic phenomena [11] [6]. In practical terms, HF accounts for the majority of the exact total energy, but the missing correlation component, though small, is chemically significant [7]. Post-HF methods systematically recover this correlation energy, bridging the gap between mean-field approximations and the exact solution of the Schrödinger equation within a given basis set.
The importance of these methods extends across computational chemistry, physics, and materials science, with growing applications in drug development for accurately modeling molecular interactions, excitation energies, and properties of excited states. This overview details the theoretical foundations, methodological categories, practical protocols, and performance characteristics of mainstream post-HF approaches, providing researchers with a framework for selecting and implementing these methods.
Theoretical Foundations of Electron Correlation
Electron correlation is conventionally separated into two distinct types: static (non-dynamical) and dynamic correlation. Static correlation arises in systems with (near-)degenerate electronic configurations, such as those encountered during bond dissociation or in transition metal complexes. A single Slater determinant provides a qualitatively inadequate description in these cases. Dynamic correlation, conversely, accounts for the instantaneous, short-range repulsion between electrons that is averaged in the HF picture. This separation, while somewhat artificial, informs the development and application of different post-HF strategies [6].
The electron correlation energy is formally defined as the difference between the exact, non-relativistic energy of a system and its HF energy calculated with a complete basis set [11]. Accurately capturing this energy presents a significant challenge because its magnitude is small relative to the total energy, yet its contribution to chemically relevant energy differences is substantial.
Categories of Post-Hartree-Fock Methods
Post-HF methods can be broadly classified into several categories based on their theoretical approach, each with distinct strengths and limitations suited to particular chemical problems. Table 1 provides a comparative summary of these methods.
Table 1: Overview of Major Post-Hartree-Fock Methods
| Method | Theoretical Approach | Handled Correlation Type | Key Strength | Key Limitation | Computational Scaling |
|---|---|---|---|---|---|
| MPn | Many-Body Perturbation Theory | Primarily Dynamic | Size-consistent; systematic improvement | Divergence in strongly correlated systems; not variational | MP2: O(N⁵), MP3: O(N⁶), MP4: O(N⁷) |
| CI | Configuration Interaction | Static & Dynamic (depending on truncation) | Variational; conceptually simple | Not size-extensive if truncated; exponential cost | CISD: O(N⁶), FCI: Factorial |
| CC | Coupled-Cluster | Static & Dynamic | Size-extensive; gold standard for small molecules | Non-variational; high computational cost | CCSD: O(N⁶), CCSD(T): O(N⁷) |
| CASSCF | Multi-Reference Wavefunction | Primarily Static | Handles strong correlation; chemically intuitive active space | Depends on active space choice; misses dynamic correlation | Factorial with active space size |
| CASPT2/ NEVPT2 | Multi-Reference Perturbation Theory | Static & Dynamic | Adds dynamic correlation to CASSCF | Costly; depends on CASSCF reference | High (typically O(N⁵) or worse) |
Perturbation Theory: Møller-Plesset (MP) Methods
Møller-Plesset perturbation theory is a cornerstone of post-HF methods, treating electron correlation as a perturbation to the HF Hamiltonian. The second-order correction, MP2, is the most widely used due to its favorable balance of cost and accuracy. MP2 captures a substantial portion of the dynamical correlation energy and is size-consistent. However, MP methods can exhibit divergent behavior for systems with significant static correlation, such as open-shell transition metal complexes, and are not variational [6]. The MP2 method is often employed as a proof-of-concept for more efficient protocols due to its relatively low computational cost [11].
Wavefunction Expansion: Configuration Interaction (CI)
The Configuration Interaction (CI) method constructs the many-electron wavefunction as a linear combination of Slater determinants, generated by exciting electrons from occupied HF orbitals to virtual orbitals. Full CI (FCI), which includes all possible excitations, provides the exact solution within the chosen basis set but is computationally feasible only for the smallest systems [6] [7]. Truncated CI methods, such as CISD (including single and double excitations), offer a practical compromise. A major drawback of truncated CI is its lack of size-extensivity, meaning the energy does not scale correctly with system size, leading to non-cancellation of errors in energy difference calculations [7].
Exponential Ansatz: Coupled-Cluster (CC) Methods
Coupled-Cluster theory employs an exponential wavefunction ansatz (e.g., ( \Psi{CC} = e^{T} \Phi{0} )) and is widely regarded as the most accurate general-purpose method for single-reference systems. The CCSD method (including single and double excitations) is size-extensive. The inclusion of a perturbative treatment of triple excitations, CCSD(T), often referred to as the "gold standard," delivers exceptional accuracy for thermochemical properties [11] [7]. The primary limitation of CC methods is their high computational cost, which restricts their application to systems of modest size.
Multi-Reference Methods: CASSCF and Beyond
For systems with strong static correlation, multi-reference methods are essential. The Complete Active Space Self-Consistent Field (CASSCF) method performs a full CI within a carefully selected set of active orbitals, which typically include the orbitals directly involved in the chemical process of interest. CASSCF provides a qualitatively correct wavefunction but recovers only a small fraction of the dynamic correlation energy [12] [6]. To address this, multi-reference perturbation theories like CASPT2 and NEVPT2 are used to add dynamic correlation on top of the CASSCF reference. These methods are powerful but require significant expertise in selecting an appropriate active space [12] [6].
Quantitative Performance of Post-HF Methods
The performance of post-HF methods can be quantified by their accuracy in predicting electron correlation energies. Recent research has demonstrated that information-theoretic approach (ITA) quantities, derived from the electron density, can predict post-HF correlation energies with linear regression [LR(ITA)], achieving chemical accuracy at the cost of a HF calculation. Table 2 summarizes the performance of LR(ITA) for various system types.
Table 2: Accuracy of LR(ITA) in Predicting MP2 Electron Correlation Energies for Various Systems [11]
| System Type | Example Systems | Best-Performing ITA Quantities | Linear Correlation (R²) | Root Mean Square Deviation (RMSD) |
|---|---|---|---|---|
| Isomers | 24 Octane Isomers | Fisher Information (I_F) |
> 0.990 | < 2.0 mH |
| Linear Polymers | Polyyne, Polyene | Shannon Entropy (S_S), Fisher Information (I_F) |
~1.000 | ~1.5 - 4.0 mH |
| Molecular Clusters | (H₂O)ₙ, (CO₂)ₙ | Onicescu information energy (E_2, E_3) |
1.000 | 2.1 - 9.3 mH |
| Metallic/Covalent Clusters | Beₙ, Mgₙ, Sₙ | Multiple ITA quantities | > 0.990 | ~17 - 42 mH |
Experimental Protocols
Protocol 1: Accurate Modeling of a Solid-State Color Center (NV⁻ in Diamond)
This protocol outlines a WFT-based approach for studying point defects with strong multi-determinant character, as demonstrated for the NV⁻ center in diamond [12].
Research Reagent Solutions
- Software: Quantum chemistry package capable of CASSCF/NEVPT2 (e.g., MOLPRO, ORCA, MOLFDIR).
- Cluster Model: A finite diamond lattice cluster, passivated by hydrogen atoms. The size must be determined via convergence tests.
- Active Space: For NV⁻, a CASSCF(6e,4o) is used, comprising four defect orbitals (a₁, a₁*, eₓ, e_y) originating from the dangling bonds of the three carbon atoms and the nitrogen atom adjacent to the vacancy.
- Basis Set: A standard Gaussian-type orbital basis set (e.g., cc-pVDZ, 6-311++G(d,p)).
Step-by-Step Workflow
Cluster Construction and Partial Optimization:
- Generate a hydrogen-terminated nanodiamond cluster of increasing size.
- Optimize atomic positions only in the immediate vicinity of the vacancy while keeping the outer shells fixed at the perfect diamond geometry to reflect the stiffness of the crystal. This is critical for convergence.
State-Specific CASSCF Geometry Optimization:
- For each electronic state of interest (e.g., the ground triplet state ( ^3A_2 )), perform a state-specific (SS) CASSCF geometry optimization. This optimizes the molecular orbitals and CI coefficients for a single state, yielding an accurate equilibrium geometry for that specific state.
State-Averaged CASSCF Single-Point Calculation:
- At the optimized geometry, perform a state-averaged (SA) CASSCF calculation. This involves requesting multiple roots (e.g., five triplet and eight singlet roots for NV⁻) with equal weights to obtain a consistent set of orbitals for describing multiple electronic states.
Dynamic Correlation Correction with NEVPT2:
- Using the SA-CASSCF wavefunction as a reference, perform a NEVPT2 calculation. This step incorporates the dynamic correlation effects from the electrons not included in the active space, which is crucial for quantitative accuracy.
Property Calculation:
- Calculate the properties of interest, such as excitation energies, zero-phonon lines (ZPLs), and fine structure, from the final CASSCF-NEVPT2 energies and wavefunctions.
The following workflow diagram illustrates this multi-step protocol:
Protocol 2: Predicting Correlation Energy via Information-Theoretic Approach (ITA)
This protocol describes the use of density-derived ITA quantities to predict post-HF correlation energies, avoiding expensive post-HF computations [11].
Research Reagent Solutions
- Software: A quantum chemistry program to perform HF calculations (e.g., NWChem, Gaussian).
- ITA Scripts: Code to compute ITA quantities (Shannon entropy, Fisher information, etc.) from the HF electron density.
- Reference Data: A small set of high-level (e.g., MP2, CCSD(T)) calculations on a training set of molecules to establish the linear regression model.
Step-by-Step Workflow
Training Set Calculation:
- For a set of representative molecules (e.g., octane isomers, water clusters), perform a standard HF calculation.
- For the same set, perform a high-level post-HF calculation (e.g., MP2) to obtain the reference electron correlation energy.
ITA Quantity Computation:
- From the HF electron density of each molecule, compute a set of 11 ITA quantities, such as Shannon entropy ((SS)), Fisher information ((IF)), and Onicescu information energy ((E2), (E3)).
Linear Regression Model Fitting (LR(ITA)):
- For each ITA quantity, perform a linear regression against the reference post-HF correlation energies to obtain a scaling equation (slope and intercept) for that quantity.
Prediction for New Systems:
- For a new, unknown system, perform only a HF calculation.
- Compute the ITA quantities from the HF density.
- Use the pre-established linear regression equations to predict the post-HF correlation energy.
Current Research and Future Outlook
The field of post-HF methods is actively evolving to address the dual challenges of computational cost and application scope. One promising direction is the information-theoretic approach (ITA), which uses physically inspired density-based descriptors to predict correlation energies. The LR(ITA) protocol has shown success for diverse systems, including polymers and molecular clusters, achieving chemical accuracy at the cost of a HF calculation [11]. This represents a potential paradigm shift towards machine learning-inspired, descriptor-based prediction of quantum chemical properties.
For large-scale systems, fragmentation methods like the generalized energy-based fragmentation (GEBF) method are crucial. These methods decompose a large system into smaller, tractable fragments, and the total energy is assembled from fragment calculations. The accuracy of LR(ITA) has been shown to be comparable to GEBF for large benzene clusters, highlighting its potential for massive systems [11].
Another critical frontier is the development of more efficient computational kernels. Research into approximate Fock exchange operators using low-rank decomposition and two-level nested self-consistent field iterations aims to drastically reduce the memory and computational bottlenecks associated with the nonlocal exchange operator, which is fundamental to both HF and hybrid DFT [13]. These advances are essential for extending the reach of high-accuracy electronic structure theory to biologically relevant systems and functional materials.
Post-Hartree-Fock methods form an essential hierarchy in the computational chemist's toolkit, enabling the systematic and controlled recovery of electron correlation energy. From the cost-effective MP2 to the highly accurate CCSD(T) and the robust multi-reference CASSCF/NEVPT2, each method offers a unique balance of accuracy, computational cost, and applicability. The choice of method depends critically on the chemical problem at hand: single-reference closed-shell molecules versus multi-reference systems like reaction transition states or open-shell transition metal complexes.
Emerging trends, including the information-theoretic approach and advanced computational approximations, are pushing the boundaries of system size and complexity that can be treated with high accuracy. As these methods continue to mature and integrate with high-performance computing and machine learning, their role in drug discovery and materials design is poised to expand significantly, providing researchers with ever more powerful tools to probe and predict molecular behavior.
In the field of computational chemistry, the pursuit of accurate predictions of molecular structure, energetics, and properties is fundamentally governed by the trade-off between computational cost and accuracy. This balance is particularly pronounced in post-Hartree-Fock methods, which were developed specifically to improve upon the limitations of the Hartree-Fock (HF) approximation by adding electron correlation effects [1]. The Hartree-Fock method itself provides a mean-field approximation that neglects the instantaneous repulsions between electrons, modeling them as interacting only with an average field [14] [15]. While HF establishes the theoretical foundation for modern electronic structure theory, its neglect of electron correlation limits its accuracy for many chemical problems, including molecular dissociation, excited states, and non-covalent interactions [16] [1].
Post-Hartree-Fock methods address this limitation by systematically accounting for electron correlation, but at a significantly increased computational cost. As these methods form the core of a broader thesis on advanced molecular calculations, understanding their specific cost-accuracy profiles is essential for selecting appropriate methodologies for research applications in areas such as drug discovery and materials design [16] [17]. This article provides a structured analysis of this fundamental trade-off, presenting quantitative data, detailed protocols, and practical guidance to inform methodological choices in scientific research.
Quantitative Analysis of Methodological Trade-Offs
The computational cost of quantum chemistry methods typically scales with system size, often expressed as a power of the number of basis functions (M) used to represent electron orbitals. The following table summarizes the characteristic scaling and accuracy of prominent methods.
Table 1: Characteristic Scaling and Accuracy of Quantum Chemistry Methods
| Method | Computational Scaling | Key Description | Typical Applications |
|---|---|---|---|
| Hartree-Fock (HF) | O(M⁴) [14] | Mean-field approximation; neglects electron correlation [1]. | Suitable for initial geometry optimizations; provides reference orbitals for post-HF methods [15]. |
| Density Functional Theory (DFT) | O(M³) to O(M⁴) [16] | Incorporates electron correlation via exchange-correlation functionals; favorable cost-accuracy balance [16]. | Workhorse for ground-state properties of medium/large systems (e.g., reaction mechanisms, material properties) [16] [18]. |
| Møller-Plesset Perturbation Theory (MP2) | O(M⁵) [16] | Adds electron correlation via 2nd-order perturbation theory [1]. | Accurate for non-covalent interactions and thermochemistry; often used for system pre-screening [16] [11]. |
| Coupled-Cluster Singles, Doubles & Perturbative Triples (CCSD(T)) | O(M⁷) [16] | "Gold standard" for single-reference systems; high accuracy but very high cost [16]. | Benchmark calculations for smaller systems (<50 atoms) [16] [11]. |
The practical implications of this scaling are profound. For instance, a recent benchmark study on predicting molecular hyperpolarizability compared Hartree-Fock and various Density Functional Theory (DFT) functionals [19]. The results demonstrated that for simple push-pull chromophores, the HF method with a modest 3-21G basis set achieved a 45.5% Mean Absolute Percentage Error (MAPE) but required only 7.4 minutes per molecule, and, crucially, provided a perfect pairwise ranking of molecules [19]. This highlights its potential as an efficient fitness function in evolutionary design algorithms where relative ordering is more critical than absolute accuracy. In contrast, more sophisticated functionals like CAM-B3LYP and M06-2X with the same basis set offered no significant improvement in accuracy for this specific property but doubled or tripled the computational cost [19].
Table 2: Illustrative Benchmarking Data for Hyperpolarizability Calculations [19]
| Method | Basis Set | Mean Absolute Percentage Error (MAPE) | Computational Time per Molecule (minutes) | Pairwise Rank Agreement |
|---|---|---|---|---|
| HF | STO-3G | 60.5% | 2.7 | Perfect (10/10 pairs) |
| HF | 3-21G | 45.5% | 7.4 | Perfect (10/10 pairs) |
| B3LYP | 3-21G | 50.1% | 14.9 | Perfect (10/10 pairs) |
| CAM-B3LYP | 3-21G | 47.8% | 28.1 | Perfect (10/10 pairs) |
| M06-2X | 3-21G | 48.4% | 35.0 | Perfect (10/10 pairs) |
Beyond the choice of electronic structure method, the selection of a basis set is a critical factor in defining the cost-accuracy balance. The basis set size directly controls the number of basis functions (M), which in turn dictates the cost of the calculation. Moving from a minimal basis set (e.g., STO-3G) to a split-valence basis set (e.g., 3-21G) provides the most significant gain in accuracy per unit of computation [19]. Further expansions, such as adding polarization and diffuse functions (e.g., 6-311++G(d,p)), yield diminishing returns and can dramatically increase the number of two-electron integrals that need to be computed, a process that scales with O(M⁴) [11] [14].
Protocol for Selecting a Computational Methodology
Workflow for Method Selection
The following diagram outlines a systematic workflow for selecting an appropriate computational method based on system size, property of interest, and available resources.
Detailed Protocol 1: High-Throughput Screening for Drug Discovery
Objective: To rapidly and accurately screen a large library of drug candidates for binding affinity.
System Preparation:
- Obtain 3D molecular structures of ligands from databases or generate them using molecular building tools.
- Perform initial geometry optimization using molecular mechanics (MM) or semiempirical quantum methods (e.g., GFN2-xTB) to eliminate steric clashes [16].
Initial Quantum Chemical Pre-optimization:
- Employ a cost-effective quantum method for further refinement. HF or a hybrid DFT functional (e.g., B3LYP) with a moderate basis set (e.g., 6-31G is often sufficient [19] [18].
- Conduct geometry optimization and frequency calculations to confirm the presence of a true minimum on the potential energy surface.
Electronic Property Calculation:
- Calculate electronic properties (e.g., electrostatic potential, HOMO-LUMO energies) from the pre-optimized structures using the same method as in step 2. These properties can serve as descriptors for machine learning models [17].
Binding Affinity Prediction:
- Option A (ML-Augmented): Input the quantum-derived descriptors into a pre-trained machine learning model (e.g., a graph neural network or random forest) to predict binding affinities [17]. This approach bypasses expensive explicit binding site calculations.
- Option B (QMMM): For a more detailed but costly analysis, embed the ligand in the protein active site using a hybrid Quantum Mechanics/Molecular Mechanics (QM/MM) scheme, where the ligand is treated with DFT and the protein with a molecular mechanics force field [16].
Validation:
- Validate the screening protocol by comparing predictions against experimental binding data or high-level CCSD(T) calculations for a small subset of representative molecules [16].
Detailed Protocol 2: Achieving Benchmark Accuracy with LR(ITA)
Objective: To accurately predict post-Hartree-Fock correlation energies for molecular clusters or polymers at a fraction of the computational cost.
Reference Data Generation (Training Set):
- Select a representative set of molecular structures (e.g., 24 octane isomers, protonated water clusters H⁺(H₂O)ₙ, or acenes of varying length) [11].
- For these structures, perform a single-point energy calculation at the Hartree-Fock (HF) level with a standard basis set (e.g., 6-311++G(d,p)) [11].
- For the same structures, perform a more expensive post-HF calculation (e.g., MP2, CCSD, or CCSD(T)) with the same basis set to obtain the reference electron correlation energy [11].
Information-Theoretic Descriptor Calculation:
- From the HF electron density obtained in the previous step, compute a set of information-theoretic approach (ITA) quantities. These include:
- Shannon Entropy (Sₛ): Characterizes the global delocalization of electron density.
- Fisher Information (I_F): Quantifies the local sharpness and inhomogeneity of the density.
- Onicescu Information Energy (E₂, E₃): Measures the concentration of the density distribution.
- Relative Rényi Entropy (R₂ᵣ, R₃ᵣ): Quantifies the difference between two density distributions [11].
- From the HF electron density obtained in the previous step, compute a set of information-theoretic approach (ITA) quantities. These include:
Model Training:
- Construct a linear regression model (LR(ITA)) for the training set, where the post-HF correlation energy is the dependent variable and one or more of the ITA quantities are the independent variables [11].
- Validate the model's accuracy by examining the coefficient of determination (R²) and the root-mean-squared deviation (RMSD) between predicted and calculated correlation energies.
Prediction for New Systems:
- For a new system of interest, perform only an HF calculation.
- Compute the same ITA quantities from the HF density.
- Use the pre-trained linear regression model to predict the post-HF correlation energy, effectively obtaining CCSD(T)-level accuracy at near-HF cost [11].
The Scientist's Toolkit: Essential Research Reagents
Table 3: Key Software and Computational "Reagents" for Post-Hartree-Fock Research
| Tool / Resource | Category | Primary Function | Relevance to Cost-Accuracy Trade-off |
|---|---|---|---|
| GPU-Accelerated Fock Matrix Builders [14] | HPC Software | Dramatically speeds up the integral evaluation and Fock matrix construction in HF/DFT calculations. | Reduces the cost of the baseline SCF calculation, making subsequent post-HF steps more accessible. |
| Linear Regression (LR) & ITA Quantities [11] | Algorithmic Approach | Predicts high-level correlation energies using low-level density descriptors. | Directly addresses the cost-accuracy problem by providing benchmark-quality energies at low cost. |
| Hybrid DFT Functionals (e.g., B3LYP, CAM-B3LYP, PBE0) [16] [19] | Theoretical Method | Provides a balanced description of electron correlation for diverse molecular properties. | Offers a practical compromise, being more accurate than HF and less costly than MP2 or CCSD(T). |
| Polarized Basis Sets (e.g., 6-31G(d), 6-311++G(d,p)) [11] [19] | Basis Set | Improves the description of electron distribution by adding angular flexibility and diffuse character. | Allows for systematic improvement of accuracy at a known computational cost increase, enabling controlled trade-offs. |
| Fragment-Based Methods (e.g., GEBF) [11] | Scalability Method | Enables quantum chemical calculations on large systems by decomposing them into smaller fragments. | Extends the application of accurate post-HF methods to large molecular clusters that would otherwise be intractable. |
The fundamental trade-off between computational cost and accuracy is an inescapable and defining aspect of computational chemistry, particularly in the realm of post-Hartree-Fock methods. Navigating this trade-off effectively is not about finding a single "best" method, but rather about making strategic choices informed by the specific research question, system size, and available computational resources. As demonstrated, strategies range from the pragmatic selection of method and basis set combinations to the adoption of innovative approaches like machine learning augmentation and information-theoretic descriptors. The ongoing integration of high-performance computing and artificial intelligence with foundational quantum chemical principles promises to further push the boundaries of this trade-off, enabling researchers to tackle increasingly complex problems in molecular science with greater confidence and efficiency.
A Deep Dive into Key Post-HF Methods and Their Applications in Drug Discovery
Non-covalent interactions (NCIs) are fundamental weak forces that govern molecular recognition, protein folding, supramolecular assembly, and drug-receptor binding. Accurate quantum mechanical treatment of these interactions requires sophisticated electron correlation methods beyond the mean-field approximation. Møller-Plesset perturbation theory, particularly at second order (MP2) and beyond, provides a balanced approach for modeling NCIs with reasonable computational cost [20] [21]. This application note examines the performance, limitations, and practical implementation of MP2 and higher-order methods for NCI prediction in chemical and pharmaceutical research contexts.
The critical importance of electron correlation for NCIs stems from the dominant role of dispersion forces in many molecular complexes. While Hartree-Fock (HF) theory completely misses dispersion, and density functional theory (DFT) requires empirical corrections, MP2 naturally incorporates these effects through its perturbative treatment of electron-electron correlations [21]. However, standard MP2 exhibits systematic overestimation of certain NCIs, necessitating methodological refinements including regularization, orbital optimization, and higher-order corrections [20] [22] [21].
Theoretical Foundation
MP2 Theory and Formulation
The MP2 correlation energy expression derives from Rayleigh-Schrödinger perturbation theory using the HF Hamiltonian as the zeroth-order operator:
Figure 1: Computational workflow for conventional MP2 theory. The method builds upon Hartree-Fock solutions through a well-defined perturbative procedure.
The MP2 correlation energy is expressed as:
$$ E{\text{MP2}} = -\frac{1}{4} \sum{ijab} \frac{|\langle ij || ab \rangle|^2}{\Delta_{ij}^{ab}} $$
where i,j denote occupied orbitals, a,b virtual orbitals, $\langle ij || ab \rangle$ represents antisymmetrized two-electron integrals, and $\Delta{ij}^{ab} = \epsilona + \epsilonb - \epsiloni - \epsilon_j$ is the orbital energy gap [20]. This formulation treats electron correlation as pairwise additive contributions from double excitations, providing a natural description of dispersion interactions missing in HF theory.
Limitations of Conventional MP2
Despite its utility, MP2 exhibits systematic deficiencies for certain NCI types:
- π-stacked complexes: MP2 notoriously overestimates interaction energies in conjugated systems like slipped benzene dimers and DNA base pairs, with errors exceeding 100% for large aromatics like the coronene dimer [20].
- Transition metal complexes: Dative bonds in organometallic compounds show overestimated binding energies due to non-additive correlation effects [20].
- Large-gap dependence: The method becomes unreliable as frontier orbital energy gaps decrease, manifesting divergent behavior for small-gap systems [20] [22].
These limitations stem from MP2's treatment of correlation as purely pairwise additive, neglecting higher-order collective effects that become important in delocalized systems with small energy denominators [20].
Methodological Advances
Regularized MP2 Methods
κ-regularization addresses MP2's divergence issues by damping contributions from small energy denominators:
$$ E{\kappa-\text{MP2}}(\kappa) = -\frac{1}{4} \sum{ijab} \frac{|\langle ij || ab \rangle|^2}{\Delta{ij}^{ab}} \left(1 - e^{-\kappa(\Delta{ij}^{ab})}\right)^2 $$
The regularization parameter $\kappa$ (typically 1.1-1.45 Eh⁻¹) attenuates terms with small denominators while preserving the standard MP2 expression for large gaps [20] [21]. This approach significantly improves performance for NCIs, reducing errors by approximately 50% across benchmark sets [21] [23].
Orbital-Optimized Variants
Orbital-optimized MP2 (OOMP2) determines orbitals self-consistently in the presence of the MP2 correlation potential, reducing spin contamination and improving descriptions of symmetry-breaking problems [21]. Combining OOMP2 with κ-regularization (κ-OOMP2) further enhances performance, particularly for systems where HF references exhibit artifactual symmetry-breaking [21].
Higher-Order Perturbation Theory
Third-order MP (MP3) and scaled variants like MP2.5 (which averages MP2 and MP3 energies) provide improved treatment of non-additive correlations:
$$ E{\text{MP2.5}} = \frac{1}{2}(E{\text{MP2}} + E_{\text{MP3}}) $$
These methods demonstrate enhanced accuracy for NCIs, particularly when combined with improved reference orbitals from κ-OOMP2 or density functional theory [21] [23].
Performance Benchmarking
Quantitative Assessment Across NCI Types
Table 1: Performance comparison of MP-based methods for non-covalent interactions (mean absolute errors in kcal/mol) [21] [23]
| Method | Hydrogen Bonding | Dispersion | Halogen Bonding | Mixed | Overall |
|---|---|---|---|---|---|
| MP2 | 0.24 | 0.89 | 0.31 | 0.52 | 0.67 |
| κ-MP2 | 0.18 | 0.41 | 0.25 | 0.31 | 0.32 |
| OOMP2 | 0.26 | 0.78 | 0.28 | 0.48 | 0.59 |
| κ-OOMP2 | 0.19 | 0.35 | 0.22 | 0.26 | 0.29 |
| MP2.5 | 0.12 | 0.28 | 0.15 | 0.19 | 0.21 |
| MP2.5:κ-OOMP2 | 0.07 | 0.12 | 0.09 | 0.08 | 0.10 |
Data compiled from testing across 19 benchmark sets (A24, S22, S66, X40, etc.) covering diverse NCI types [21] [23]. Results demonstrate the significant improvement achieved through regularization and reference orbital optimization.
Comparison with Higher-Level Methods
Table 2: Method scalability and accuracy trade-offs for NCI prediction [20] [22] [21]
| Method | Computational Scaling | Typical System Size | NCI Accuracy | Key Limitations |
|---|---|---|---|---|
| HF | O(N⁴) | ~100 atoms | Poor (no dispersion) | Neglects electron correlation |
| MP2 | O(N⁵) | ~50-100 atoms | Moderate | Overestimates dispersion |
| κ-MP2 | O(N⁵) | ~50-100 atoms | Good | Parameter dependence |
| MP2.5:κ-OOMP2 | O(N⁶) | ~30-80 atoms | Excellent | Increased computational cost |
| CCSD(T) | O(N⁷) | ~20-30 atoms | Excellent (gold standard) | Prohibitive for large systems |
| CCSD(cT) | O(N⁷) | ~20-30 atoms | Superior for large systems | Addresses CCSD(T) overcorrelation |
Recent developments like CCSD(cT) address the overcorrelation issues in CCSD(T) for large, polarizable systems by including additional diagrammatic terms that screen the bare Coulomb interaction [22]. For the coronene dimer, CCSD(cT) reduces binding energy errors by nearly 2 kcal/mol compared to CCSD(T), achieving chemical accuracy against diffusion Monte Carlo benchmarks [22].
Experimental Protocols
Standard Protocol for κ-Regularized MP2 Calculations
Objective: Compute accurate NCI energies for molecular complexes using κ-MP2.
Required Resources:
- Quantum chemistry software (e.g., Psi4, Q-Chem, ORCA)
- Aug-cc-pVTZ basis set (recommended)
- Molecular geometries (optimized at appropriate level)
Procedure:
- Geometry Preparation: Obtain optimized structures of monomers and complex using DFT with dispersion correction (e.g. ωB97M-V) or HF/MP2 with appropriate basis.
- Single-Point Energy Calculation:
- Perform HF calculation to obtain reference orbitals and orbital energies
- Compute MP2 correlation energy using κ-regularization (κ = 1.1-1.45 Eh⁻¹)
- For optimal results, use κ-OOMP2 with κ = 1.45 Eh⁻¹ [21]
- Binding Energy Calculation:
- Compute total energies for complex (Ecomplex) and monomers (EA, EB)
- Calculate interaction energy: ΔE = Ecomplex - (EA + EB)
- Apply basis set superposition error (BSSE) correction via counterpoise method
- Validation: Compare against benchmark values for representative systems (e.g., S66 subset)
Expected Results: κ-MP2 typically reduces MP2 overbinding by 30-60% for dispersion-dominated complexes while maintaining accuracy for hydrogen-bonded systems [20] [21].
Advanced Protocol: MP2.5 with Optimized Reference Orbitals
Objective: Achieve CCSD(T)-level accuracy for NCIs at reduced computational cost.
Procedure:
- Reference Orbital Generation:
- MP3 Energy Calculation:
- Compute MP3 correlation energy using optimized reference orbitals
- Scale third-order contribution by 0.5 (MP2.5 = MP2 + 0.5*(MP3-MP2))
- Binding Energy Analysis:
- Calculate interaction energies as in Protocol 5.1
- For large systems, consider local correlation approximations (DLPNO-CCSD(T)) for validation [22]
Performance: MP2.5:κ-OOMP2 achieves RMSD of 0.10 kcal/mol for S66 dataset, rivaling CCSD(T) accuracy at O(N⁶) cost [21] [23].
The Scientist's Toolkit
Table 3: Essential computational resources for MP-based NCI studies
| Resource Type | Specific Tools | Application Purpose | Key Considerations |
|---|---|---|---|
| Software Packages | Psi4, Q-Chem, ORCA, Gaussian | Quantum chemistry calculations | Psi4 offers excellent MP2.5 implementation; Q-Chem supports κ-OOMP2 |
| Basis Sets | aug-cc-pVTZ, aug-cc-pVDZ | Electron wavefunction expansion | Augmented correlation-consistent basis sets essential for NCIs |
| Benchmark Sets | S22, S66, A24, HSG | Method validation and parameterization | Provide diverse NCI types for balanced assessment |
| Analysis Tools | NCIplot, QTAIM, SAPT | Interaction decomposition and visualization | Reveal nature and strength of specific non-covalent contacts |
| Reference Data | CCSD(T)/CBS, DMC | High-accuracy benchmarks | Essential for method validation where experimental data is scarce |
Application in Drug Discovery
MP2-based methods provide critical insights for structure-based drug design, particularly for targeting challenging binding sites:
- Fragment-based screening: MP2.5 methods accurately rank fragment binding affinities by capturing dispersion contributions to binding [8] [24]
- Halogen bonding: κ-MP2 correctly models σ-hole interactions important in drug-receptor recognition [21] [25]
- Dispersion-dominated binding: For targets with hydrophobic pockets, MP2.5 provides superior affinity predictions compared to standard DFT [8] [24]
Case studies demonstrate successful application in optimizing kinase inhibitors and targeting metalloenzymes where accurate NCI treatment is essential for binding affinity predictions [8].
MP2 and its advanced variants represent a sweet spot in the accuracy-cost trade-off for NCI prediction in drug discovery applications. The methodological developments in regularization, orbital optimization, and higher-order corrections have addressed many of conventional MP2's limitations while maintaining computational feasibility for pharmaceutical-sized systems.
Future directions include:
- Integration with machine learning for rapid screening of NCI energies across chemical space [24]
- Quantum computing implementations of post-Hartree-Fock methods for potentially exponential speedup [26]
- Advanced local correlation methods enabling MP2.5/CCSD(T) accuracy for entire drug-receptor complexes [22]
As these technologies mature, MP-based methods will continue bridging the gap between computational efficiency and chemical accuracy for non-covalent interactions in pharmaceutical research.
Coupled-Cluster with Singles, Doubles, and perturbative Triples (CCSD(T)) is widely regarded as the "gold standard" in quantum chemistry for computing molecular energies and properties. This status is attributed to its remarkable ability to deliver high accuracy—often within 1 kJ/mol or 1 kcal/mol of experimental values—for molecules with predominantly single-reference electronic character. The method achieves this by systematically treating electron correlation effects through a wavefunction ansatz that includes all single and double excitations from a reference determinant (usually Hartree-Fock) and incorporates a non-iterative perturbative correction for connected triple excitations.
Despite its superior accuracy, the application of CCSD(T) has been historically limited by its steep computational scaling, which formally reaches for the (T) correction, where N represents the system size. This has traditionally restricted conventional implementations to systems of approximately 20-25 atoms. However, recent methodological and computational advances have significantly extended the reach of CCSD(T), enabling applications to molecules with 50-75 atoms and beyond, while maintaining its gold-standard accuracy. These developments are making CCSD(T) an increasingly powerful tool for researchers and drug development professionals tackling complex chemical problems.
Recent Methodological Advances Extending the Reach of CCSD(T)
The prohibitive computational cost of conventional CCSD(T) implementations has motivated the development of several cost-reduction strategies that dramatically extend its applicability while preserving high accuracy.
Key Approximation Techniques
Frozen Natural Orbitals (FNOs) compress the virtual molecular orbital space by discarding orbitals with low occupation numbers, as determined from a lower-level wavefunction (typically MP2). This leads to a significant reduction in computational cost, as the number of virtual orbitals is the primary determinant of the steep scaling. The error introduced by this truncation can be systematically controlled and corrected. [27] [28]
Density Fitting (DF) or Resolution-of-the-Identity (RI) approximates the four-center two-electron repulsion integrals using expansions in an auxiliary basis set. This reduces the memory, storage, and computational burdens associated with handling these integrals. [27]
Natural Auxiliary Functions (NAFs) further compress the auxiliary basis set used in DF, providing additional speedups without sacrificing accuracy. This is particularly effective when combined with FNOs, as the reduced orbital space requires less auxiliary description. [27] [28]
Explicitly Correlated (F12) Methods improve the slow convergence of correlation energy with basis set size by introducing terms in the wavefunction that depend explicitly on the interelectronic distance (r~12~). This allows for the use of smaller basis sets to achieve near-complete-basis-set-limit accuracy, offering substantial computational savings. [28]
Table 1: Summary of Key Cost-Reduction Techniques for CCSD(T)
| Technique | Underlying Principle | Primary Benefit | Reported Speedup |
|---|---|---|---|
| Frozen Natural Orbitals (FNO) | Truncation of the virtual orbital space based on natural occupation numbers. | Reduces scaling with the number of virtual orbitals. | Up to an order of magnitude [27] |
| Density Fitting (DF) | Approximation of two-electron integrals using an auxiliary basis. | Reduces storage, I/O, and pre-factor costs. | ~5-10x for integral processing |
| Natural Auxiliary Functions (NAF) | Compression of the DF auxiliary basis set. | Further reduces cost of DF integral assembly. | 1.5-3x on top of DF [28] |
| Explicitly Correlated F12 | Explicit inclusion of the interelectronic distance in the wavefunction. | Drastically improves basis set convergence. | Enables use of smaller basis sets for CBS-quality results [28] |
The combination of FNO and NAF approximations with efficient, parallelized algorithms has been shown to deliver overall speedups of 5 to 10 times for triple-ζ basis sets, making CCSD(T) calculations on systems with 50-75 atoms feasible on affordable computing resources within a few days. [27] [28] A very recent preprint (2025) confirms that these hybrid parallel approaches enable calculations on systems of up to 60 atoms and 2500 orbitals, which were previously beyond reach without local approximations. [29]
Application Protocols for Specific Chemical Systems
The accurate application of CCSD(T) requires carefully designed protocols tailored to different types of chemical systems and properties of interest.
Protocol for Thermochemistry and Reaction Energies
This protocol is designed for computing accurate reaction energies, barrier heights, and atomization energies.
- Geometry Optimization: Optimize the molecular geometry of all reactants and products using a robust, lower-cost method. Density Functional Theory (DFT) with a functional like ωB97M-V and a triple-ζ basis set (e.g., def2-TZVP) is a suitable choice. [30]
- Reference Energy Calculation: Perform a single-point Hartree-Fock calculation with a large correlation-consistent basis set (e.g., aug-cc-pVTZ) to obtain the reference energy and orbital set.
- FNO-CCSD(T) Energy Calculation:
- Perform a MP2 calculation to generate the virtual orbital density matrix.
- Diagonalize the density matrix to obtain Natural Orbitals (NOs) and their occupation numbers.
- Retain the subset of NOs with occupation numbers above a conservative threshold (e.g., corresponding to an energy error of <1 µE~h~). This defines the FNO space. [27]
- Execute the CCSD(T) calculation within the truncated FNO space.
- (Optional but Recommended) Compute a MP2-level correction, ΔMP2, which is the difference between the MP2 energy in the full space and the FNO-truncated space. Add this correction to the final FNO-CCSD(T) energy to improve accuracy. [28]
- Basis Set Extrapolation (For Highest Accuracy): Repeat Step 3 with a series of basis sets (e.g., aug-cc-pVTZ and aug-cc-pVQZ) and extrapolate the correlation energy to the complete basis set (CBS) limit using established formulas. The F12 approach can serve as an alternative to this extrapolation. [28]
Protocol for Non-Covalent Interactions (NCIs)
NCIs, such as π-stacking and hydrogen bonding, are crucial in drug binding and materials science. Their accurate description is notoriously challenging.
- Geometry and Counterpoise Correction: Use CCSD(T)-quality geometries if available, often from high-level DFT. To account for Basis Set Superposition Error (BSSE), the counterpoise correction is essential. [30]
- High-Level Single-Point Calculations:
- For the dimer and each monomer, perform a FNO-CCSD(T) calculation using a large, diffuse-function-augmented basis set (e.g., aug-cc-pVTZ). The monomer calculations must be performed in the full dimer basis (counterpoise correction).
- The interaction energy is calculated as: E~int~ = E~dimer~ - E~monomer A (in dimer basis)~ - E~monomer B (in dimer basis)~.
- Accuracy Assessment for Large Systems: For very large π-stacked systems (e.g., acene dimers), be aware that post-CCSD(T) contributions (e.g., full triples, quadruple excitations) might become non-negligible. The linear evolution of the correlation energy with monomer size can be used as a probe to assess the behavior of CCSD(T) for large systems. [30]
Table 2: Recommended Computational Protocols for Different Chemical Problems
| Application | Recommended Method | Recommended Basis Set | Key Considerations | Target Accuracy |
|---|---|---|---|---|
| General Thermochemistry | FNO-CCSD(T) + ΔMP2 correction | aug-cc-pVTZ / aug-cc-pVQZ | Use tight FNO truncation thresholds; CBS extrapolation is beneficial. | ~1 kJ/mol [27] |
| Non-Covalent Interactions | FNO-CCSD(T) with counterpoise correction | aug-cc-pVTZ or cc-pVTZ-F12 | Essential to use basis sets with diffuse functions; monitor performance via interaction energy slopes. | ~0.1-0.5 kcal/mol |
| Transition Metal Reactions | FNO-CCSD(T) / PNO-LCCSD(T) | cc-pVTZ / cc-pVQZ for non-metals; aug-cc-pwCVTZ for metals | Open-shell systems require specific implementations; static correlation may be a concern. | ~2-4 kJ/mol |
| Extended Polymers & Clusters | FNO-CCSD(T) or Linear Regression (ITA) | 6-311++G(d,p) or larger | For very large systems, information-theoretic approach (ITA) can predict correlation from HF. [11] | Chemical accuracy possible [11] |
Workflow Visualization
Successful application of advanced coupled-cluster methods requires a suite of well-defined "research reagents" in the form of software, basis sets, and computational protocols.
Table 3: Essential Software and Basis Sets for CCSD(T) Calculations
| Tool / Reagent | Type | Primary Function | Key Features / Use Case |
|---|---|---|---|
| MRCC | Software Suite | Performs canonical and local correlation CC calculations. | Capable of high-level methods like CCSDT(Q); used for benchmarking. [30] |
| CFOUR | Software Suite | High-level quantum chemical calculations. | Used for demanding canonical post-CCSD(T) calculations. [30] |
| ORCA | Software Suite | Versatile quantum chemistry package. | Performs DLPNO-CCSD(T) and DFT geometry optimizations. [30] |
| cc-pVXZ Family | Basis Set | Systematic basis sets for correlation-consistent calculations. | The cornerstone for CCSD(T) studies (X = D, T, Q, 5...). [31] [30] |
| aug-cc-pVXZ | Basis Set | Correlation-consistent sets with diffuse functions. | Essential for anions, excited states, and non-covalent interactions. [30] |
| def2 Series | Basis Set | Efficient, generally contracted basis sets. | Good balance of cost and accuracy; popular for molecular systems. [31] |
| FNO-CCSD(T) | Method/Protocol | Reduced-cost CCSD(T) via virtual space truncation. | Default choice for systems of ~30-75 atoms. [27] [28] |
| CCSD(F12*) | Method/Protocol | Explicitly correlated CCSD with efficient triples. | Preferred for achieving CBS limits with smaller basis sets. [28] |
| DLPNO-CCSD(T) | Method/Protocol | Local correlation approximation for large systems. | Enables calculations on systems with hundreds of atoms. [30] |
The accurate prediction of drug-receptor binding affinities and reaction mechanisms is a central challenge in computational drug discovery. While classical molecular mechanics (MM) methods offer speed, they lack the quantum mechanical (QM) precision required to model electronic phenomena such as charge transfer, bond breaking/formation, and polarization [8] [10]. Ab initio quantum chemistry methods, particularly post-Hartree-Fock (post-HF) methods, address this need by providing a more physically realistic description of electron correlation, which is neglected in the foundational Hartree-Fock (HF) approximation [6] [1]. The HF method's mean-field approach, where each electron interacts with the average field of the others, leads to significant errors in the calculation of interaction energies, a critical shortcoming for predicting ligand binding [8] [7]. Post-HF methods systematically improve upon HF by accounting for the instantaneous, correlated motion of electrons, thereby offering a pathway to high-accuracy predictions of binding affinities and reaction pathways that are crucial for rational drug design [6] [32].
The applicability of conventional post-HF methods to large biological systems has historically been limited by their formidable computational cost and poor scaling with system size [6] [7]. However, recent methodological advances are bridging this gap. The development of fragmentation approaches, such as the Molecules-in-Molecules (MIM) method, combined with efficient approximations like Domain-based Local Pair Natural Orbital (DLPNO) techniques, now enables post-HF quality calculations on systems as large as protein-ligand complexes [32] [33]. This article details the application of these advanced post-HF protocols, providing researchers with a framework for achieving benchmark accuracy in modeling drug-receptor interactions.
Quantitative Performance of Post-HF Methods
The selection of an appropriate electronic structure method requires a careful balance between accuracy and computational feasibility. The performance of various methods for key properties relevant to drug discovery is summarized in Table 1.
Table 1: Performance Benchmarking of Quantum Chemical Methods for Drug Discovery Applications
| Method | Electron Correlation Treatment | Typical Binding Energy Error | Computational Scaling | Best for Applications Involving |
|---|---|---|---|---|
| Hartree-Fock (HF) | None (Mean-Field) | High (Systematic Underestimation) [8] | O(N⁴) [8] | Initial geometries, charge distributions [8] |
| Møller-Plesset 2nd Order (MP2) | Perturbation Theory [6] | Moderate (~a few kcal/mol) [6] | O(N⁵) | Dynamical correlation; relatively small systems [6] |
| Coupled-Cluster Singles, Doubles & Perturbative Triples (CCSD(T)) | Exponential Cluster Operator [7] | Very Low (< 1 kcal/mol) - "Gold Standard" [33] | O(N⁷) | High-fidelity benchmark calculations for small models [7] |
| DLPNO-CCSD(T) | Local Approximation to CCSD(T) [33] | Low (~1 kcal/mol) [33] | ~O(N⁴) to O(N⁵) [33] | Accurate single-point energies for large systems [32] [33] |
| Configuration Interaction Singles & Doubles (CISD) | Variational (Limited Excitations) [6] | Moderate (Lacks Size-Extensivity) [7] | O(N⁶) | Small system wavefunctions; historical use [6] |
| Complete Active Space SCF (CASSCF) | Variational (Active Space) [6] | High for Dynamical Correlation | Exponential with active space | Static (Non-Dynamical) Correlation; bond breaking, excited states [6] |
| Density Functional Theory (DFT) | Approximate Functional [8] | Functional-Dependent (Low with Hybrids) [8] | O(N³) [8] | General-purpose ground-state properties, reaction mechanisms [8] |
Recent innovations have successfully brought post-HF accuracy to protein-ligand systems. A 2024 study achieved errors of less than 1 kcal mol⁻¹ for protein-ligand binding affinities by combining the three-layer MIM fragmentation scheme (MIM3) with DLPNO-based post-HF methods [33]. This approach demonstrated remarkable correlation with experimental data, with R² values of approximately 0.90 and 0.78 for the CDK2 and BZT-ITK datasets, respectively [33]. The MIM-DLPNO framework effectively overcomes the traditional scalability barrier, making CCSD(T)-level accuracy feasible for drug-sized molecules.
Experimental Protocols
Protocol 1: Calculating Protein-Ligand Binding Affinity Using a MIM-DLPNO-CCSD(T)/MM Approach
This protocol describes a hybrid quantum mechanics/molecular mechanics (QM/MM) approach utilizing the Molecules-in-Molecules (MIM) fragmentation method to compute highly accurate protein-ligand binding affinities at the post-HF level [32] [33].
1. System Preparation
- Initial Coordinates: Obtain the atomic coordinates of the protein-ligand complex from a high-resolution X-ray crystal structure or a refined molecular dynamics (MD) snapshot.
- MM Setup: Prepare the entire system using a classical molecular mechanics force field (e.g., AMBER, CHARMM). Add hydrogen atoms, solvate the complex in a water box, and add counterions to neutralize the system.
- QM Region Definition: Identify the ligand and key protein residues within ~3-5 Å of the ligand that are involved in specific interactions (e.g., hydrogen bonds, salt bridges, π-stacking). This defines the "core" region for the high-level QM treatment.
2. MIM Fragmentation Scheme Setup
- Layer 1 (High-Level, Core): This layer contains the ligand and the immediate binding pocket residues critical for interaction. Apply the DLPNO-CCSD(T) method with a medium-sized basis set (e.g., cc-pVTZ) for the highest accuracy on the central interaction.
- Layer 2 (Medium-Level, Intermediate): This layer includes protein residues and solvent molecules that are farther from the ligand but still part of the local environment. Use a faster but still correlated method like DLPNO-MP2 with a smaller basis set (e.g., cc-pVDZ or 6-31G) [32] [33].
- Layer 3 (Low-Level, Environment): The remainder of the protein and solvent is treated at a low level of theory, such as HF or a semi-empirical method, or even with the MM force field in a QM/MM scheme.
3. Binding Energy Calculation via the Supermolecular Approach
- Perform three separate MIM energy calculations:
- Ecomplex: For the entire protein-ligand complex.
- Eprotein: For the protein alone.
- E_ligand: For the isolated ligand.
- To enhance computational efficiency, strategically cancel out common subsystem energy terms between the complex and the protein calculations, as these largely remain unchanged upon ligand binding [32].
- Calculate the binding affinity (ΔE_bind) using the supermolecular equation:
- ΔEbind = Ecomplex - (Eprotein + Eligand)
4. Analysis and Validation
- Compare the computed ΔE_bind against experimentally determined binding free energies (e.g., from Isothermal Titration Calorimetry - ITC).
- Decompose the interaction energy to identify residues contributing most significantly to binding, providing insights for lead optimization.
The workflow for this protocol is illustrated in Figure 1 below.
Figure 1: Workflow for MIM-DLPNO-CCSD(T)/MM Binding Affinity Calculation.
Protocol 2: Investigating a Reaction Mechanism in an Enzyme Active Site using CASSCF/CASPT2
This protocol is designed for studying reaction mechanisms, such as covalent inhibition or enzymatic catalysis, where electronic rearrangements and bond breaking/forming are critical. It is particularly important when dealing with transition metal centers or systems with significant multi-configurational character (static correlation) [6].
1. Active Site Model Construction
- Cluster Approach: Extract a finite cluster model from the protein structure that includes the ligand (or substrate), the catalytic residues, key cofactors (e.g., a heme group, Zn²⁺ ion), and a portion of the surrounding protein backbone. Saturated dangling bonds with hydrogen atoms.
- QM/MM Approach (Alternative): For a more realistic environment, embed a high-level QM region (the active site) within a MM-treated protein and solvent environment.
2. Geometry Exploration
- Use a more efficient method (e.g., DFT) to optimize the geometries of the reactants, products, intermediates, and transition states along the proposed reaction coordinate.
- Perform frequency calculations to confirm that intermediates have no imaginary frequencies and transition states have a single imaginary frequency.
3. Active Space Selection (Crucial Step)
- For the CASSCF calculation, judiciously select the active space, denoted as (ne, mo), where ne is the number of active electrons and mo is the number of active molecular orbitals.
- This requires chemical intuition. For example, for a reaction involving the breaking of a sigma bond and formation of a pi bond, the active space should include the relevant bonding and antibonding orbitals and their electrons.
- Example: For the study of a cytochrome P450 reaction, the active space might include the Fe 3d orbitals, the porphyrin π orbitals, and the frontier orbitals of the substrate.
4. Multi-Reference Wavefunction Calculation
- CASSCF Calculation: Perform a CASSCF calculation on the optimized structures to obtain a wavefunction that correctly describes static correlation and near-degeneracy effects within the active space. This yields the correct qualitative description of the reaction pathway [6].
- Dynamic Correlation Correction (CASPT2): Because CASSCF recovers mainly static correlation, follow with a CASPT2 (Complete Active Space Perturbation Theory 2nd Order) calculation. This adds dynamical correlation energy as a perturbation, which is essential for quantitative accuracy of reaction barriers and energies [6].
5. Reaction Profile and Analysis
- Construct the potential energy surface (PES) by plotting the CASPT2//CASSCF energies for all stationary points (reactants, TS, intermediates, products).
- Analyze the CASSCF wavefunctions and natural orbitals for each stationary point to verify the electronic state and understand the bond formation/cleavage process.
The logical relationship between the methodological choices in this protocol is shown in Figure 2.
Figure 2: Methodology for Investigating Reaction Mechanisms with CASSCF/CASPT2.
The Scientist's Toolkit: Essential Research Reagents & Software
Successful implementation of the protocols above requires a suite of specialized software tools and computational resources. The key components are listed in Table 2.
Table 2: Essential Tools for Post-HF Research in Drug Discovery
| Tool Category | Example Software | Specific Function / Note |
|---|---|---|
| Quantum Chemistry Packages | ORCA, Gaussian, GAMESS(US), MOLPRO, MOLFDIR [6], COLUMBUS [6] | Perform core electronic structure calculations (HF, MP2, CC, CI, CASSCF). ORCA is noted for its efficient DLPNO implementations. |
| Fragmentation Methods | Molecules-in-Molecules (MIM) [32] [33] | Enables post-HF calculations on large systems by dividing them into smaller, tractable fragments. |
| QM/MM Software | AMBER, CHARMM, GROMACS (with QM/MM plugins), CP2K | Integrates QM treatment of the active site with MM treatment of the biological environment. |
| Molecular Visualization & Modeling | Chimera, ChimeraX, PyMOL, VMD, GaussView | For system preparation, visualization of structures/orbitals, and analysis of results. |
| High-Performance Computing (HPC) | Local Clusters, National Supercomputing Centers, Cloud Computing (AWS, Azure, GCP) | Essential for all but the smallest post-HF calculations due to high computational cost. |
| Basis Set Libraries | Basis Set Exchange (BSE) website | A repository for standard basis sets (e.g., cc-pVXZ, 6-31G). |
Fragment-Based and Linear-Scaling Approaches for Large Biomolecular Systems
The investigation of large biomolecular systems represents a significant challenge in computational chemistry and drug discovery. The accurate application of post-Hartree-Fock methods to these systems has traditionally been constrained by their steep computational scaling, which often renders explicit calculations prohibitively expensive [6]. In response, two complementary strategies have emerged as transformative approaches: fragment-based methodologies and linear-scaling computational techniques.
Fragment-based drug discovery (FBDD) provides a conceptual framework that aligns with the practical constraints of computational efficiency. By decomposing large molecular systems into smaller, tractable chemical units, FBDD enables the systematic exploration of chemical space and interaction motifs that would be computationally intractable at full scale [34] [35]. This approach synergizes powerfully with advances in linear-scaling algorithms and machine learning potentials, which collectively overcome traditional limitations in system size and complexity [36] [16].
These integrated methodologies are reshaping the landscape of biomolecular simulation and rational drug design, particularly for challenging target classes such as protein-protein interactions and membrane receptors [37] [34]. This document provides detailed application notes and experimental protocols for implementing these approaches within the rigorous theoretical context of post-Hartree-Fock research.
Quantitative Benchmarking of Computational Approaches
The selection of appropriate computational methods requires careful consideration of their respective accuracy, scalability, and specific applicability to fragment-based workflows. The following table summarizes key methodological characteristics for large biomolecular systems.
Table 1: Computational Method Benchmarks for Biomolecular Applications
| Method Class | Representative Methods | Theoretical Scaling | Key Applications in FBDD | Accuracy Limitations |
|---|---|---|---|---|
| Post-Hartree-Fock | MP2, CCSD(T), CASSCF [6] | O(N⁵) to O(N⁷) [6] | Benchmarking fragment interactions, reaction energies | Limited to small fragments or model systems |
| Density Functional Theory | DFT with dispersion corrections (DFT-D3, DFT-D4) [16] | O(N³) | Fragment geometry optimization, binding energy estimation [16] | Functional-dependent errors for dispersion, charge transfer |
| Linear-Scaling QM | FMO, ONIOM, F-SAPT [37] [16] | O(N) to O(N²) | Protein-ligand interaction decomposition, fragment linking optimization [37] | Boundary effects in fragmentation, approximation errors |
| Neural Network Potentials | Egret-1, AIMNet2 [38] | O(N) | High-throughput fragment screening, molecular dynamics [38] | Training data dependency, transferability limitations |
| Molecular Mechanics | MD simulations, FEP [35] | O(N) to O(N²) | Fragment binding pose sampling, relative binding affinity [35] | Force field parameterization limitations |
The practical application of these methods generates substantial quantitative data requiring systematic organization. The following table presents characteristic performance metrics for key methodologies discussed in this protocol.
Table 2: Characteristic Performance Metrics for Fragment-Based Workflows
| Methodology | System Size Range | Typical Time Scale | Accuracy Metrics | Key Applications |
|---|---|---|---|---|
| Ultra-Large Library Docking [36] | Billions of compounds | Hours to days [39] | ~30% hit rate improvement over HTS [36] | Initial fragment hit identification |
| FEP Calculations [35] | 50-500 atoms | Hours to days per transformation | ~1 kcal/mol accuracy in ΔΔG [39] | Fragment optimization, affinity prediction |
| F-SAPT Analysis [37] | 50-200 atoms | Minutes to hours per complex | Quantitative interaction decomposition | Rational fragment growth and linking |
| Neural Network Potentials [38] | 100-100,000 atoms | Seconds to minutes per evaluation | Near-DFT accuracy at MM speed [38] | Conformational sampling, property prediction |
| t-SMILES Generation [40] | Variable fragment libraries | Rapid sequence generation | 100% theoretical validity [40] | De novo fragment-based design |
Experimental Protocols
Protocol 1: Fragment-Based Hit Identification Using Ultra-Large Virtual Screening
Principle: This protocol leverages ultra-large virtual screening to identify fragment hits from gigascale chemical libraries, combining docking and machine learning for efficient exploration of chemical space [36].
Materials:
- Target protein structure (X-ray, cryo-EM, or homology model)
- Pre-processed fragment library adhering to "Rule of 3" criteria (MW <300 Da, cLogP <3, HBD <3, HBA <3, rotatable bonds <3) [35]
- High-performance computing resources with GPU acceleration
- Virtual screening platform (e.g., OpenEye, Schrödinger) [39]
Procedure:
- Target Preparation:
- Prepare protein structure using standard protein preparation workflows (hydrogen addition, bond order assignment, optimization of hydrogen bonding networks)
- Define binding site using experimental data or pocket detection algorithms
- Generate grid maps for docking calculations
Library Curation:
- Apply "Rule of 3" filters to ensure fragment-like properties [35]
- Enumerate reasonable tautomers and protonation states at physiological pH
- Generate multi-conformer representations for each fragment
Iterative Screening:
Hit Validation:
- Select 100-500 top-ranking fragments for experimental validation
- Validate hits using biophysical methods (SPR, MST, ITC) [35]
- Determine binding affinity (KD) and ligand efficiency (LE >0.3 kcal/mol per heavy atom)
Structural Characterization:
- Pursue co-crystallization of fragment-target complexes for hit validation
- Use structural data to inform fragment optimization campaigns
Figure 1: Fragment-Based Hit Identification Workflow
Protocol 2: Fragment Optimization Using Free Energy Perturbation and F-SAPT Analysis
Principle: This protocol employs free energy perturbation (FEP) and functional-group symmetry-adapted perturbation theory (F-SAPT) to guide rational fragment optimization with quantitative accuracy [37] [35].
Materials:
- Experimentally validated fragment hits with confirmed binding mode
- Protein-fragment complex structure (from X-ray crystallography or homology modeling)
- FEP-enabled molecular dynamics software (e.g., Schrodinger, OpenMM)
- Quantum chemistry software with SAPT capabilities (e.g., Promethium) [37]
Procedure:
- Initial Complex Preparation:
- Prepare protein-fragment complex using structural data
- Solvate system in explicit water model with appropriate ion concentration
- Equilibrate system using molecular dynamics (100 ns production run)
FEP Map Construction:
- Design congeneric series around fragment core structure
- Define perturbation paths between related fragments
- Calculate relative binding affinities (ΔΔG) using FEP/REST protocols
F-SAPT Analysis:
- Extract protein-fragment interaction regions from MD trajectories
- Perform F-SAPT calculations on representative snapshots
- Decompose interaction energies into physically meaningful components:
- Electrostatic contributions
- Exchange-repulsion
- Induction/polarization
- Dispersion interactions
Structure-Based Design:
- Identify key interaction hotspots using F-SAPT results
- Design fragment extensions targeting favorable interactions
- Prioritize synthetic targets based on FEP predictions and synthetic accessibility
Iterative Optimization Cycle:
- Synthesize top-priority analogues (typically 10-50 compounds per cycle)
- Experimental validation of binding affinities (SPR, ITC)
- Structural characterization of key complexes
- Refine computational models based on new data
Figure 2: Fragment Optimization Using FEP and F-SAPT
Protocol 3: Machine Learning-Enhanced Fragment-Based de novo Design
Principle: This protocol integrates fragment-based molecular representation with deep learning generation models for de novo ligand design, using the t-SMILES framework to ensure high validity and novelty [40].
Materials:
- Curated fragment libraries (e.g., BRICS, MMPA, or custom fragments) [40]
- t-SMILES encoding software
- Transformer-based language model architecture
- High-throughput molecular simulation platform (e.g., Rowan) [38]
Procedure:
- Fragment Library Preparation:
- Fragment known bioactive molecules using algorithm (JTVAE, BRICS, MMPA, Scaffold) [40]
- Apply drug-likeness filters and remove problematic functional groups
- Characterize fragments by physicochemical properties and structural features
t-SMILES Encoding:
- Convert fragments to t-SMILES representation using TSSA, TSDY, or TSID algorithms [40]
- Generate multiple descriptions per molecule to create augmented training set
- Tokenize sequences for transformer model input
Model Training:
- Pre-train transformer model on large chemical database (ChEMBL, ZINC)
- Fine-tune on target-specific bioactive molecules if available
- Implement attention mechanisms to capture fragment importance
Goal-Directed Generation:
- Condition generation on desired properties (potency, selectivity, ADMET)
- Use reinforcement learning or Bayesian optimization for multi-objective optimization
- Generate 10,000-100,000 candidate molecules
Validation and Selection:
- Filter generated molecules for synthetic accessibility and drug-likeness
- Predict binding poses using molecular docking
- Select top 100-500 candidates for further evaluation
- Synthesize and test highest-priority candidates (10-50 compounds)
Successful implementation of fragment-based and linear-scaling approaches requires access to specialized computational tools and experimental resources. The following table details key components of the integrated workflow.
Table 3: Essential Research Reagents and Computational Resources
| Category | Specific Tools/Resources | Key Functionality | Application Context |
|---|---|---|---|
| Virtual Screening Platforms | OpenEye Toolkits [39], Schrödinger Platform [36] | Ultra-large library docking, high-throughput screening | Initial fragment hit identification |
| Quantum Chemistry Software | Promethium (F-SAPT) [37], ORCA, Gaussian | Post-HF calculations, interaction energy decomposition | Fragment binding mode analysis, QM/MM simulations |
| Machine Learning Potentials | Rowan Platform (Egret-1, AIMNet2) [38], Neural Network Potentials | Near-quantum accuracy at molecular mechanics speed | Conformational sampling, property prediction |
| Molecular Representation | t-SMILES frameworks [40] | Fragment-based molecular representation for deep learning | De novo molecular generation, chemical space exploration |
| Biophysical Screening | SPR (Biacore), MST, ITC, NMR [35] | Label-free detection of weak fragment binding | Experimental validation of computational predictions |
| Structural Biology | X-ray Crystallography, Cryo-EM [35] | Atomic-resolution structure determination | Fragment binding mode elucidation |
| Free Energy Calculations | FEP-enabled MD software [35] | Relative binding affinity prediction | Fragment optimization and lead compound selection |
The integration of fragment-based approaches with linear-scaling computational methods represents a paradigm shift in the study of large biomolecular systems. By leveraging the principles of decomposition and efficient algorithmic scaling, these protocols enable researchers to overcome traditional limitations in system size and complexity while maintaining the theoretical rigor of post-Hartree-Fock quantum chemistry.
The workflows presented herein—from ultra-large virtual screening to machine learning-enhanced de novo design—provide a comprehensive framework for advancing drug discovery against challenging biological targets. As computational resources continue to grow and algorithms become increasingly sophisticated, these approaches will undoubtedly expand their impact, potentially democratizing the drug discovery process and enabling the cost-effective development of safer, more effective therapeutics [36].
Overcoming Computational Hurdles: Strategies for Efficient Post-HF Calculations
In the realm of post-Hartree-Fock methods for molecular calculations, the selection of an appropriate basis set is a critical step that directly influences the accuracy and computational cost of a study. A basis set is a set of functions, typically centered on atomic nuclei, used to represent molecular orbitals by turning partial differential equations into algebraic equations suitable for computational implementation [41] [42]. The fundamental challenge for researchers lies in navigating the trade-off between completeness and feasibility; larger basis sets provide a more complete description of the electron distribution and correlation effects but lead to a rapid, often prohibitive, increase in computational demand [42]. This application note provides a structured framework for selecting basis sets that are both scientifically rigorous and computationally practical, with a specific focus on applications in advanced wavefunction-based methods.
Basis Set Fundamentals and Types
Core Concepts and Terminology
Basis sets are composed of basis functions that approximate atomic orbitals. The key to their performance lies in their composition and the types of functions they include:
- Gaussian-Type Orbitals (GTOs): The most common type of function in quantum chemistry, favored because the product of two GTOs can be expressed as a linear combination of other GTOs, greatly simplifying the computation of multi-center integrals [41] [42]. They are less accurate than Slater-Type Orbitals (STOs) near the atomic nucleus and in the "tail" region, but this is mitigated by using multiple GTOs to represent a single atomic orbital [41].
- Contraction: A contracted Gaussian function is a fixed linear combination of primitive Gaussian functions. This strategy allows for a more accurate representation of atomic orbitals while keeping the number of variational parameters manageable [43] [44].
- Polarization Functions: These are functions with angular momentum one unit higher than the highest occupied atomic orbital (e.g., d-functions on carbon, or p-functions on hydrogen). They allow the electron density to shift away from the nucleus, providing the flexibility needed to model the distortion of atomic electron clouds during chemical bonding [41].
- Diffuse Functions: These are Gaussian functions with small exponents, giving them a more extended spatial distribution. They are crucial for accurately modeling anions, excited states, van der Waals interactions, and other phenomena where electrons are far from the nucleus [41] [44].
Hierarchy of Common Basis Set Families
Basis sets are organized into families of increasing size and accuracy. The most relevant for post-Hartree-Fock calculations are summarized in Table 1.
Table 1: Common Basis Set Families for Molecular Calculations
| Basis Set Family | Key Characteristics | Common Notation Examples | Typical Use Cases |
|---|---|---|---|
| Pople-style [41] | Split-valence; computationally efficient for Hartree-Fock and DFT. | 6-31G, 6-31G, 6-31+G | Geometry optimizations, frequency calculations on medium-to-large systems. |
| Dunning's cc-pVXZ [43] [41] | Correlation-consistent polarized Valence X-Zeta; systematically approaches the complete basis set (CBS) limit. | cc-pVDZ (DZ), cc-pVTZ (TZ), cc-pVQZ (QZ) | High-accuracy post-HF calculations (e.g., CCSD(T)), property convergence studies. |
| Karlsruhe (def2) [43] | Well-balanced and optimized for general-purpose use, including with DFT. | def2-SVP, def2-TZVP, def2-QZVP | DFT and post-HF calculations on a wide range of systems, including organometallics. |
| Atomic Natural Orbital (ANO) [44] | Uses ANOs from correlated calculations; very compact and accurate. | ANO-RCC | Spectroscopy, heavy elements, and systems requiring high accuracy with a relatively small set. |
| Minimal Basis Sets [41] | One basis function per atomic orbital; fast but inaccurate for most applications. | STO-3G | Very large systems, preliminary conformational searches. |
The "X" in Dunning's cc-pVXZ denotes the zeta-level, corresponding to the number of basis functions for each valence atomic orbital. The sequence DZ (double-zeta), TZ (triple-zeta), QZ (quadruple-zeta), 5Z, etc., represents a systematic increase in the number of basis functions, allowing calculations to converge toward the CBS limit [41]. Augmented versions (e.g., aug-cc-pVXZ) include an additional shell of diffuse functions on all atoms [43].
Quantitative Comparison of Basis Set Performance
The choice of basis set significantly impacts computed molecular properties. Table 2 illustrates the convergence of the C-O bond length and vibrational frequencies in CO₂ at the Hartree-Fock level with Dunning basis sets [45]. This demonstrates that while properties generally improve with larger basis sets, the gains must be weighed against the increased computational cost.
Table 2: Basis Set Convergence for CO₂ Properties (Hartree-Fock Level) [45]
| Basis Set | C-O Bond Length (Å) | Vibrational Frequency #1 (cm⁻¹) | Vibrational Frequency #2 (cm⁻¹) | Vibrational Frequency #3 (cm⁻¹) |
|---|---|---|---|---|
| cc-pVDZ (DZ) | 1.1406 | 761.3 | 1513.3 | 2580.3 |
| cc-pVTZ (TZ) | 1.1319 | 782.4 | 1573.4 | 2695.4 |
| cc-pVQZ (QZ) | 1.1283 | 789.8 | 1593.6 | 2734.7 |
For post-Hartree-Fock methods, the recovery of correlation energy is paramount. As illustrated in one study, smaller basis sets like 6-31G can show severe deviations of up to 33 kcal/mol from experimental reaction energies compared to larger, polarized sets [46]. Furthermore, the Basis Set Superposition Error (BSSE), an artificial lowering of energy in molecular systems due to the use of incomplete basis sets, can severely affect dissociation energy calculations, sometimes contributing up to 60% of the computed binding energy when large diffuse functions are used [46]. The Counterpoise (CP) correction is a common, though not always perfect, method to correct for BSSE [44].
A Protocol for Systematic Basis Set Selection
Selecting a basis set is not a one-size-fits-all process. The following workflow provides a logical, step-by-step protocol for making an informed choice, balancing accuracy and computational feasibility.
Diagram 1: A logical workflow for selecting a basis set, showing the iterative process of benchmarking and refinement.
Step-by-Step Protocol
Step 1: Assess System Characteristics
- Size: The number of atoms is the primary driver of computational cost. For large systems (e.g., drug-sized molecules), a double-zeta polarized basis may be the practical limit for post-HF calculations [47].
- Electronic Nature: Systems with lone pairs, anions, or low-energy excited states require diffuse functions (e.g., aug-cc-pVDZ). Weakly interacting complexes and van der Waals systems are particularly sensitive to this [44].
- Element Composition: For atoms beyond the second period, effective core potentials (ECPs) and matching basis sets (e.g., LANL2DZ, def2-ECPs) are often used to reduce computational cost while maintaining accuracy for valence electrons [43].
Step 2: Define Accuracy Requirements
- Target Properties: The required basis set depends heavily on the molecular property of interest [31].
- Geometry: Often converged at the triple-zeta level with polarization [47] [45].
- Vibrational Frequencies: Typically require at least a triple-zeta basis set for quantitative accuracy [45].
- Reaction Energies/Bond Dissociation: Highly sensitive to correlation energy recovery; require high-level methods and large basis sets (TZ or larger) with careful BSSE correction [46] [31].
- Hyperfine Coupling Constants: Specialized basis sets like EPR-II and EPR-III are optimized for these properties [43].
Step 3: Establish Computational Constraints
- CPU Time and Memory: Computational cost for post-HF methods scales as O(N⁵) to O(N⁷) with system size (N), and basis set size is a key factor in N [47]. A 30% reduction in basis set size can lead to a ~50% decrease in calculation time [46].
- Software and Algorithms: Exploit efficiency gains from resolution-of-the-identity (RI) or density fitting approximations, and chain-of-sphere (COSX) integrations, for which specific auxiliary basis sets are available [47].
Step 4: Select Initial Basis Set Family and Zeta-Level
- For Post-Hartree-Fock Methods: Dunning's cc-pVXZ family is the gold standard due to its systematic construction for correlation energy recovery and smooth extrapolation to the CBS limit [41] [31] [47].
- Start with cc-pVDZ for preliminary scans or very large systems.
- Use cc-pVTZ for most research-grade calculations of energies and properties.
- Use cc-pVQZ and/or CBS extrapolation for high-accuracy benchmarking.
- For DFT Calculations: Pople-style (e.g., 6-311+G*) or Karlsruhe (def2-TZVP) basis sets are highly efficient and often recommended [47] [48]. The pcseg- family is also specifically optimized for use with DFT [47].
Step 5: Conduct a Benchmarking Study
- On a Model System: Select a smaller, chemically relevant model system (e.g., a dipeptide for a protein study) [48].
- Systematic Variation: Perform single-point energy or geometry optimization calculations on the model system using a range of basis sets (e.g., cc-pVDZ → cc-pVTZ → cc-pVQZ) at your target level of theory (e.g., CCSD(T)) [45].
- Check for Convergence: Plot the target property (e.g., energy, bond length, frequency) against basis set size. The property is considered converged when the change from one zeta-level to the next falls below a predefined threshold (e.g., 1 kJ/mol for energy) [31] [45].
Step 6: Final Selection and Production Run
- Select the smallest basis set that provides converged results for your target property within the computational constraints.
- Justify your choice in any reporting by referencing the benchmarking process or literature evidence for similar systems [48].
The Scientist's Toolkit: Essential Basis Set Reagents
Table 3: Key "Research Reagent" Basis Sets for Post-Hartree-Fock Calculations
| Basis Set | Function | Typical Application in Protocol |
|---|---|---|
| cc-pVDZ | Double-zeta polarized basis. | Initial benchmark point; calculations on very large systems where accuracy can be sacrificed for speed. |
| cc-pVTZ | Triple-zeta polarized basis. | The recommended starting point for most production-level post-HF energy and property calculations. |
| aug-cc-pVDZ | Augmented double-zeta basis. | Studying anions, excited states, or weak interactions where a TZ calculation is too expensive. |
| aug-cc-pVTZ | Augmented triple-zeta basis. | High-accuracy studies of properties requiring diffuse functions (e.g., electron affinities, Rydberg states). |
| cc-pCVDZ | Core-polarized double-zeta basis. | Calculations where core-valence correlation is important (e.g., spin-orbit coupling, hyperfine structure). |
| def2-TZVP | General-purpose triple-zeta basis. | A robust alternative to cc-pVTZ, especially when using RI approximations or for transition metals. |
| EPR-II / EPR-III | Specialized for hyperfine coupling. | DFT calculations of hyperfine coupling constants and other magnetic properties [43]. |
Basis set selection is a cornerstone of reliable computational chemistry. There is no single "best" basis set; the optimal choice is a strategic decision based on the specific chemical system, the target properties, and the available computational resources. By adhering to a systematic protocol—involving system assessment, definition of requirements, and rigorous benchmarking—researchers can make defensible and efficient choices. This disciplined approach ensures that computational efforts in post-Hartree-Fock research are both feasible and capable of delivering chemically meaningful insights, particularly in demanding fields like drug development where predictive accuracy is paramount.
The accurate calculation of electron correlation energy is a central challenge in quantum chemistry, critical for predicting molecular properties, reaction mechanisms, and binding affinities in drug discovery. Traditional post-Hartree-Fock (post-HF) methods, while accurate, suffer from computational costs that skyrocket with system size, rendering them prohibitive for large molecular systems. This application note details two innovative approximation strategies—Information-Theoretic Approach (ITA) and Machine Learning (ML) surrogates—that are revolutionizing molecular calculations by achieving high accuracy at substantially reduced computational cost. These approaches represent a paradigm shift, enabling researchers to bypass traditional bottlenecks while maintaining the precision required for pharmaceutical development and materials design.
Information-Theoretic Approach (ITA) for Electron Correlation Energy Prediction
Theoretical Foundation
The Information-Theoretic Approach (ITA) reframes the electron correlation problem through the lens of information theory by treating the electron density as a continuous probability distribution. This framework utilizes quantitative descriptors that encode both global and local features of the electron density distribution, providing physically interpretable, basis-set agnostic measures for correlation energy prediction [11] [49].
The core ITA quantities include:
- Shannon Entropy ((S_S)): Characterizes the global delocalization of electron density.
- Fisher Information ((I_F)): Quantifies local inhomogeneity and the sharpness of density features.
- Ghosh-Berkowitz-Parr Entropy ((S_{GBP})): Provides an alternative entropy measure.
- Onicescu Information Energy ((E2), (E3)): Represents information energy content.
- Relative Rényi Entropy ((R{2r}), (R{3r})): Measures distinguishability between densities.
- Relative Shannon Entropy ((I_G)): Kullback-Leibler divergence for density differences.
- Relative Fisher Information ((G1), (G2), (G_3)): Additional discrimination capabilities [11] [49].
LR(ITA) Protocol and Performance
The Linear Regression ITA (LR(ITA)) protocol establishes strong linear relationships between Hartree-Fock-level ITA quantities and post-HF correlation energies (MP2, CCSD, CCSD(T)), enabling high-accuracy prediction at HF computational cost [11].
Table 1: LR(ITA) Performance for Octane Isomers (6-311++G(d,p) Basis Set)
| ITA Quantity | Method | R² | RMSD (mH) |
|---|---|---|---|
| (S_S) | MP2 | 0.878 | 1.9 |
| (S_S) | CCSD | 0.897 | 1.3 |
| (S_S) | CCSD(T) | 0.893 | 1.5 |
| (I_F) | MP2 | 0.987 | 0.6 |
| (I_F) | CCSD | 0.989 | 0.4 |
| (I_F) | CCSD(T) | 0.988 | 0.5 |
| (S_{GBP}) | MP2 | 0.964 | 1.0 |
| (S_{GBP}) | CCSD | 0.974 | 0.6 |
| (S_{GBP}) | CCSD(T) | 0.972 | 0.8 |
For polymeric systems, LR(ITA) demonstrates exceptional accuracy with R² values approaching 1.000 and RMSDs of ~1.5 mH for polyyne, ~3.0 mH for polyene, and <4.0 mH for all-trans-polymethineimine [11]. For more challenging systems like acenes and 3D molecular clusters (Beₙ, Mgₙ, Sₙ), while strong linear correlations persist (R² > 0.990), single ITA quantities capture insufficient information for quantitative accuracy, with RMSDs increasing to ~10-42 mH [11].
Experimental Protocol: LR(ITA) Implementation
Objective: Predict post-HF electron correlation energies using ITA quantities derived from Hartree-Fock calculations.
Software Requirements: Quantum chemistry package with Hartree-Fock and post-HF capability (e.g., Gaussian, GAMESS, PySCF); Custom Python scripts for ITA quantity calculation and linear regression.
Step-by-Step Workflow:
Molecular Geometry Optimization
- Perform geometry optimization at HF/6-311++G(d,p) level
- Verify convergence criteria (energy gradient < 10⁻⁴ a.u.)
Reference Electron Correlation Energy Calculation
- Compute reference correlation energies using post-HF methods (MP2, CCSD, or CCSD(T))
- Use consistent 6-311++G(d,p) basis set for direct comparison
- For large systems, employ Generalized Energy-Based Fragmentation (GEBF) for linear-scaling reference values [11]
ITA Quantity Calculation from HF Density
- Calculate electron density ( \rho(r) ) from converged HF wavefunction
- Compute all 11 ITA quantities using numerical integration:
- Shannon entropy: ( SS = -\int \rho(r) \ln \rho(r) dr )
- Fisher information: ( IF = \int \frac{|\nabla \rho(r)|^2}{\rho(r)} dr )
- Additional quantities as defined in reference [11]
Linear Regression Model Development
- Construct dataset of ITA quantities (X) vs. reference correlation energies (Y)
- Perform linear regression: ( E_{corr} = m \times ITA + b )
- Validate model with k-fold cross-validation
- Assess quality via R² coefficient and RMSD
Prediction for New Systems
- Compute HF-level ITA quantities for target system
- Apply LR(ITA) model to predict correlation energy
- For molecular clusters, verify transferability across sizes
LR(ITA) Workflow: Information-theoretic quantities from HF calculations predict correlation energies.
Machine Learning Surrogates for Electronic Structure Methods
One-Electron Reduced Density Matrix Learning
Machine learning approaches are creating surrogate electronic structure methods that bypass traditional self-consistent field procedures. By leveraging the bijective maps established by density functional theory and reduced density matrix functional theory, ML models can learn the fundamental relationship between external potential and the one-electron reduced density matrix (1-rdm) [50].
The γ-learning approach directly learns the map: ( \hat{\gamma}[\hat{v}] = \sum{i}^{N{sample}} \hat{\beta}i K(\hat{v}i, \hat{v}) ) where ( \hat{v} ) is the external potential, ( K ) is the kernel function, and ( \hat{\beta}_i ) are regression coefficients [50].
This method enables accurate prediction of 1-rdms for surrogate electronic structure methods ranging from local DFT to full configuration interaction. From predicted density matrices, molecular observables, energies, and atomic forces can be computed using either standard quantum chemistry or secondary ML models [50].
Unrestricted Coupled Cluster for Reactive Chemistry
Recent advances enable high-throughput unrestricted coupled cluster calculations for reactive systems, providing gold-standard data for machine learning interatomic potentials (MLIPs). Automated workflows address key challenges including:
- Hartree-Fock reference construction using stability analysis
- Basis set corrections for energies and forces
- Automatic filtering of unreliable calculations near symmetry breaking points [51]
Table 2: UCCSD(T) Dataset for Organic Molecules
| Parameter | Specification |
|---|---|
| Theory Level | Unrestricted CCSD(T) |
| Configurations | 3,119 |
| Content | Energies and Forces |
| Molecules | Organic (C, H, N, O) |
| Maximum Atoms | 16 |
| Application | MLIP Training |
MLIPs trained on UCCSD(T) data show significant improvement over DFT-based models, with >0.1 eV/Å better force accuracy and >0.1 eV improvement in activation energy reproduction—critical for modeling chemical reactions [51].
Experimental Protocol: ML Surrogate Development
Objective: Develop machine learning surrogate models for electronic structure methods using 1-rdm learning.
Software Requirements: QMLearn or equivalent ML framework; Quantum chemistry software (PySCF, Q-Chem); Data preprocessing tools.
Step-by-Step Workflow:
Training Set Generation
- Select diverse molecular set (small to medium-sized)
- Perform electronic structure calculations for target method (DFT, HF, CI)
- Extract 1-rdm matrices and external potential representations
- Include configurational diversity (equilibrium and non-equilibrium geometries)
Descriptor Representation
- Represent external potentials and 1-rdms using Gaussian-type orbitals
- Employ internal reference frame for rotational/translational invariance
- Matrix element-based representation for efficient learning [50]
Model Training
- Implement kernel ridge regression or neural networks
- γ-learning: Direct 1-rdm prediction from external potential
- γ+δ-learning: Learn energy/force functionals from predicted 1-rdm
- Optimize hyperparameters via cross-validation
Model Validation
- Compare predicted vs. calculated energies and forces
- Validate on unseen molecular systems
- Assess conservation laws in molecular dynamics
- Calculate spectroscopic properties from predicted densities
Application Phase
- Input molecular structure and external potential
- Predict 1-rdm using trained model
- Compute properties directly or via secondary functionals
- Perform energy-conserving molecular dynamics
ML Surrogate Development: Training machine learning models to predict electronic structure.
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Computational Tools for Advanced Molecular Calculations
| Tool/Resource | Type | Function | Application Context |
|---|---|---|---|
| 6-311++G(d,p) | Basis Set | Balanced accuracy/cost for electron correlation | LR(ITA) protocol development [11] |
| Generalized Energy-Based Fragmentation (GEBF) | Method | Linear-scaling energy computation for large systems | Benchmarking molecular clusters [11] |
| QMLearn | Software Package | ML surrogate model development | 1-rdm learning and prediction [50] |
| SparQ Tool | Quantum Info Tool | Quantum information analysis of wavefunctions | Electron correlation analysis [52] |
| Unrestricted CCSD(T) Dataset | Data Resource | Gold-standard energies/forces for organic molecules | Training reactive ML potentials [51] |
| Kernel Ridge Regression | Algorithm | Non-linear learning for 1-rdm maps | γ-learning implementation [50] |
Information-theoretic approaches and machine learning surrogates represent transformative methodologies for electron correlation energy prediction in molecular systems. The LR(ITA) protocol demonstrates that simple density-based descriptors can predict post-HF correlation energies with chemical accuracy at Hartree-Fock cost, validated across diverse systems from isomers to molecular clusters. Concurrently, ML surrogate methods using 1-rdm learning effectively bypass traditional SCF procedures, enabling rapid computation of electronic properties with high fidelity. For drug discovery professionals, these approaches offer practical pathways to incorporate high-level electron correlation effects into molecular design workflows, potentially accelerating the development of novel therapeutics through more accurate binding affinity prediction and reaction modeling. As both fields continue to advance, their integration promises even greater efficiencies in computational molecular design.
High-Performance Computing (HPC) and Algorithmic Optimizations for Scalability
The accurate calculation of electronic structure properties through post-Hartree-Fock methods is fundamental to advancements in quantum chemistry, materials science, and drug discovery. These methods address the critical limitation of the Hartree-Fock approach by accounting for electron correlation effects [6]. However, post-Hartree-Fock calculations are computationally demanding, with traditional implementations often exhibiting unfavorable scaling with system size, restricting their application to small molecules [6] [53]. The integration of High-Performance Computing (HPC) and the development of novel, low-scaling algorithms are therefore essential for enabling high-accuracy simulations of large, biologically and industrially relevant systems. This Application Note details contemporary strategies—spanning algorithmic innovations, specialized software, and advanced hardware utilization—that are pushing the boundaries of scalable electronic structure calculations, thereby providing researchers with practical methodologies for tackling increasingly complex molecular systems.
Algorithmic Foundations and Performance Analysis
Low-Scaling Post-Hartree-Fock Algorithms
Traditional post-Hartree-Fock methods, such as MP2 and coupled-cluster, are plagued by high computational costs, often scaling as O(N⁵) or worse, where N represents the system size [6]. To overcome this bottleneck, low-scaling algorithms that leverage mathematical approximations without significantly compromising accuracy have been developed.
Tensor Numerical Methods: These methods represent multidimensional functions and operators using low-rank tensor formats, drastically reducing computational complexity. For instance, the tensor-structured numerical Hartree-Fock solver can compute the two-electron integrals (TEI) tensor with O(n log n) complexity for a grid of size n×n×n, a significant improvement over conventional methods [54]. This approach is not restricted to Gaussian-type orbitals and allows for high-precision calculations on large 3D grids.
Resolution-of-the-Identity (RI) with Numerical Laplace Transform: Implemented in software like CP2K, this combination is key to low-scaling SOS-MP2 (Spin-opposite-scaled Møller–Plesset second-order perturbation theory) and RPA (Random Phase Approximation) methods [55]. The RI technique, employing a short-range metric, approximates four-center electron repulsion integrals using three-center integrals, enhancing sparsity and computational efficiency. A numerical Laplace transform further reduces scaling by replacing an integral with a weighted sum over a few grid points (typically 6-8), making methods like SOS-MP2 sub-cubic scaling [55].
Linear-Scaling Fragmentation Methods: For massive molecular clusters, the Generalized Energy-Based Fragmentation (GEBF) method enables the calculation of post-Hartree-Fock electron correlation energies by decomposing a large system into smaller, tractable fragments. The total property is then obtained by combining the results of these subsystem calculations, achieving linear scaling [49].
Quantitative Performance of Correlation Energy Predictions
The LR(ITA) protocol offers an efficient pathway to predict post-Hartree-Fock correlation energies using information-theoretic approach (ITA) quantities computed at the Hartree-Fock level. The following table summarizes the accuracy of this method for different molecular systems, demonstrating its potential as a low-cost predictive tool.
Table 1: Accuracy of LR(ITA) in Predicting MP2 Electron Correlation Energies for Various Systems [49]
| System Type | Example Systems | R² Value | Root Mean Squared Deviation (RMSD) |
|---|---|---|---|
| Isomers | 24 Octane Isomers | ~0.987 (using IF) | 0.6 mH |
| Linear Polymers | Polyyne, Polyene | ~1.000 | 1.5 - 3.0 mH |
| Molecular Clusters | (C₆H₆)ₙ, (CO₂)ₙ, H⁺(H₂O)ₙ | Similar accuracy to GEBF | N/A |
HPC-Enabled Scalability
Leveraging modern HPC architectures is crucial for achieving practical scalability. Recent demonstrations show that quantum chemistry software can be optimized to run on state-of-the-art supercomputers.
Table 2: HPC Performance and Scaling of Quantum Chemistry Codes
| Software/Code | HPC Architecture | Application Scope | Achieved Scalability |
|---|---|---|---|
| VeloxChem | LUMI Supercomputer (AMD Instinct MI250X GPUs) | Full protein-ligand QM interactions, Spectroscopy [56] | Full-machine scaling for systems of hundreds to thousands of atoms |
| FSIM | MPI, OpenMP | Pedagogical Hartree-Fock with McMurchie-Davidson integrals [15] [57] | Modular framework for parallel execution studies |
| CP2K Low-Scaling | CPU-based HPC clusters | SOS-MP2, RPA for periodic systems (solids, liquids) [55] | Sub-cubic scaling; applicable to systems with hundreds of atoms |
Detailed Experimental Protocols
Protocol 1: Low-Scaling SOS-MP2 Molecular Dynamics of Liquid Water with CP2K
This protocol outlines the steps for performing a short molecular dynamics simulation of liquid water using the low-scaling SOS-MP2 method, a correlated post-Hartree-Fock approach [55].
1. System Preparation and Input Configuration
- Coordinate File: Prepare an XYZ file containing the atomic coordinates of the water system (e.g., 32 molecules in a cubic box of side 9.85 Å).
- Input File Structure: The CP2K input file should be structured with specific sections:
&GLOBAL: Define the project name (PROJECT water32) and run type (RUN_TYPE MD).&MOTION/&MD: Specify the number of MD steps (STEPS 3).&FORCE_EVAL/&DFT: This is the core section for the electronic structure calculation.
2. Basis Set and Potential Setup
- Basis Sets: Use pre-optimized, correlation-consistent basis sets (e.g.,
cc-TZfor oxygen and hydrogen) and their matching RI auxiliary basis sets (e.g.,RI_TZ) for efficiency. These are included via theBASIS_SET_FILE_NAMEkeyword. - Pseudopotentials: Specify the appropriate pseudopotential file (
POTENTIAL_FILE_NAME).
3. Self-Consistent Field (SCF) Calculation
- Method: Perform a pure Hartree-Fock calculation by setting
&XC_FUNCTIONALtoNONEand including&HFwithFRACTION 1.0. - Convergence: Use tight convergence criteria (e.g.,
EPS_SCF 1.0E-6) and a sufficient number of iterations (MAX_SCF 40). - Periodicity: For periodic HF, use a truncated Coulomb operator as the HFX potential with a cutoff radius smaller than half the box length (
POTENTIAL_TYPE TRUNCATEDandCUTOFF_RADIUS 4.5).
4. SOS-MP2 Energy and Force Calculation
- Activation: Within the
&XCsection, add a&WF_CORRELATIONsubsection. - Laplace Quadrature: Use the
&RI_SOS_MP2subsection to specify the number of grid points for the numerical Laplace transform (QUADRATURE_POINTS 6). - Opposite-Spin Scaling: Set the scaling parameter for the opposite-spin component (e.g.,
SCALE_S 1.3). - Low-Scaling Algorithm: Enable the efficient algorithm with
&LOW_SCALINGand set a memory cut parameter (MEMORY_CUT 3). - RI Metric: In the
&RI/&RI_METRICsubsection, define a short-range metric. For sparse systems like water, a truncated Coulomb metric with a 1.5-2.0 Å cutoff is recommended over the overlap metric for better accuracy.
5. Execution and Analysis
- Run: Execute the calculation using
mpirun -np [number_of_cores] cp2k.popt [input_file].inp. - Output: Monitor the output for SOS-MP2 energy contributions and forces, which are used to propagate the MD simulation.
Protocol 2: HPC-Driven Quantum Chemistry with VeloxChem on GPU Clusters
This protocol describes the setup for running large-scale quantum chemical calculations, such as those for protein-ligand systems, on GPU-accelerated HPC infrastructure using VeloxChem [56].
1. Software and Hardware Environment
- Software: Install the VeloxChem quantum chemistry package, compiled with GPU support (e.g., for AMD Instinct MI250X GPUs).
- HPC Access: Secure access to a GPU-based supercomputing cluster, such as LUMI.
2. System Modeling and Basis Set Selection
- System Size: The code is optimized for massive systems (hundreds to thousands of atoms). Prepare the geometry of the large molecular system (e.g., a protein-ligand complex).
- Basis Set: Choose a suitable Gaussian-type orbital basis set (e.g.,
cc-pVTZ) that balances accuracy and computational cost for the target system.
3. Job Configuration for HPC
- Input Script: Prepare an input file specifying the desired calculation (e.g., ground-state energy, molecular gradients, or spectroscopic properties).
- Resource Request: In the job submission script (e.g., Slurm or PBS), request multiple GPU nodes. For example, to utilize the entire LUMI supercomputer, a full-node job submission is required.
- Example Slurm directives:
4. Execution and Result Collection
- Launch: Submit the job to the queue using the scheduler command (e.g.,
sbatch job_script.sh). - Performance Monitoring: Utilize the HPC's profiling tools to monitor GPU utilization, memory usage, and inter-node communication efficiency.
- Output Analysis: Upon completion, analyze the output files for energies, properties, and performance metrics. The key advantage is the ability to obtain correlated-level (e.g., MP2, CCSD(T)) accuracy for systems previously considered intractable.
Workflow and Relationship Diagrams
HPC Workflow for Low-Scaling Post-HF Calculations
The following diagram illustrates the logical workflow for setting up and running a low-scaling post-Hartree-Fock calculation on a high-performance computing system, integrating the key components from the protocols above.
Diagram Title: HPC Workflow for Low-Scaling Post-HF Calculations
This diagram outlines the modular software architecture of the FSIM framework, which is designed for clarity and educational exploration of HPC concepts in quantum chemistry [15].
Diagram Title: FSIM HPC Framework Modular Architecture
Table 3: Key Software, Hardware, and Basis Set "Reagents" for HPC Quantum Chemistry
| Category | Item | Function and Application Note |
|---|---|---|
| Software & Libraries | CP2K | A molecular dynamics and electronic structure package featuring highly efficient low-scaling SOS-MP2 and RPA methods for periodic and molecular systems. [55] |
| VeloxChem | A quantum chemistry software designed from the ground up for HPC environments, with optimized GPU support for large-scale systems like protein-ligand complexes. [56] | |
| FSIM | A pedagogical, object-oriented C++ framework for the Hartree-Fock method and McMurchie-Davidson integrals. Serves as an ideal platform for learning and prototyping HPC strategies (MPI, OpenMP). [15] | |
| HPC Hardware | AMD Instinct MI250X | GPU accelerator used in the LUMI supercomputer. Delivers the high computational throughput and memory bandwidth required for scaling to thousands of atoms. [56] |
| Basis Sets | cc-pVXZ, cc-TZ | Correlation-consistent basis set families. Essential for achieving high accuracy in post-Hartree-Fock correlation energy calculations. [55] |
| RI Auxiliary Basis (e.g., RI_TZ) | Specially optimized fitting basis sets used in the Resolution-of-the-Identity (RI) approximation to reduce the computational cost of two-electron integrals. [55] |
The pursuit of accurate and computationally feasible methods for modeling complex molecular systems is a central challenge in computational chemistry and drug discovery. While post-Hartree-Fock methods provide a gold standard for quantum mechanical accuracy, their prohibitive computational cost often restricts their application to small systems [16]. To overcome this limitation, the field has increasingly turned to hybrid and multi-level strategies that synergistically combine the accuracy of high-level quantum mechanics (QM) with the efficiency of lower-level methods such as molecular mechanics (MM) and machine learning (ML) [58] [16]. These integrated approaches enable researchers to study chemically reactive events in large, complex environments like proteins and solvated systems, which are critical for biomedical applications. This article details specific application notes and protocols for implementing these strategies, framed within the context of advanced electronic structure theory, to achieve optimal performance in predictive modeling.
Theoretical Foundation and Key Concepts
The Accuracy-Efficiency Trade-Off
The core challenge in molecular simulation is balancing the trade-off between computational accuracy and cost. Ab initio quantum chemical methods, including Hartree-Fock (HF), Density Functional Theory (DFT), and post-Hartree-Fock approaches like Møller-Plesset perturbation theory (MP2) and Coupled Cluster theory (CCSD(T)), offer high accuracy by solving the many-body Schrödinger equation but are computationally demanding [59] [16]. CCSD(T), often considered the benchmark for precision, has a computational cost that scales steeply with system size, limiting its routine use [16]. Conversely, Molecular Mechanics (MM) uses classical force fields to model molecular systems efficiently but fails to capture bond formation/breaking and electronic transitions [58].
The Hybrid QM/MM Paradigm
Hybrid Quantum Mechanical/Molecular Mechanical (QM/MM) methods bridge this divide by partitioning the system [58]. A small, chemically active region (e.g., a drug's functional group or an enzyme's active site) is treated with a high-level QM method, while the surrounding environment is modeled with a computationally efficient MM force field [58]. This allows for accurate modeling of reactive events within their native, complex environments. Advanced implementations often use electrostatic embedding, where the MM point charges polarize the QM electron density, providing a more realistic interaction model [58].
Table 1: Comparison of Electronic Structure Methods Used in Multi-Level Strategies
| Method | Theoretical Description | Accuracy | Computational Cost | Typical Use in Hybrid Schemes |
|---|---|---|---|---|
| Coupled Cluster (e.g., CCSD(T)) | Accounts for electron correlation via cluster operators; gold standard [16]. | Very High | Very High | Benchmarking; small core regions where maximum accuracy is critical. |
| Density Functional Theory (DFT) | Models electron density via exchange-correlation functionals [16]. | High (functional-dependent) | Medium-High | Primary QM engine for reactive regions in enzymes and solutions. |
| Hartree-Fock (HF) | Mean-field approximation neglecting electron correlation [16]. | Low-Medium | Medium | Base method for some QM/MM setups; often corrected. |
| Semiempirical Methods (e.g., GFN2-xTB) | Approximates QM integrals with empirical parameters [16]. | Medium | Low | Large QM regions; high-throughput screening; pre-optimization. |
Application Notes and Protocols
The following protocols outline specific workflows for applying hybrid strategies to problems in drug development and structural biology.
Protocol: Elucidating Covalent Drug Binding Mechanisms with QM/MM MD
Application Objective: To characterize the reaction mechanism and free energy profile of a covalent inhibitor binding to its biological target, such as a transition metal-based anticancer drug (e.g., RAPTA-C) interacting with a protein or DNA [58].
1. System Setup
- Initial Structure: Obtain a resolved 3D structure of the target biomacromolecule (e.g., from Protein Data Bank) and the drug molecule.
- Parameterization: Use force fields like AMBER ff14SB (for proteins) and ff99bsc0 (for DNA). Generate parameters for the drug molecule using compatible tools (e.g., GAUSSIAN for QM-derived charges) [58].
- Solvation: Solvate the entire system in a periodic box of explicit water molecules, using a model such as TIP3P [58].
- QM/MM Partitioning: Define the QM region to include the drug's reactive center and key amino acid side chains or nucleic acid residues involved in the binding reaction. The MM region comprises the rest of the protein, solvent, and ions.
2. Simulation Workflow
- Equilibration: Perform classical MD to equilibrate the solvent and MM environment.
- Enhanced Sampling QM/MM MD: Conduct hybrid QM/MM Molecular Dynamics using an enhanced sampling technique, such as umbrella sampling or metadynamics, to overcome the timescale limitation of rare reactive events [58]. The QM method is typically a DFT functional, while the MM region uses the classical force field.
- Free Energy Calculation: Compute the potential of mean force (PMF) along the reaction coordinate (e.g., forming bond distance) to obtain the reaction free energy profile (∆G) and activation barrier [58].
3. Analysis
- Analyze the PMF to identify transition states and intermediates.
- Inspect MD trajectories to characterize key atomic interactions stabilizing the reaction pathway.
Diagram 1: QM/MM MD protocol for covalent drug binding.
Protocol: Integrating Machine Learning with ab initio Calculations
Application Objective: To predict molecular properties (e.g., solubility, toxicity, binding affinity) with high accuracy and lower computational cost than pure ab initio methods [16] [60].
1. Data Generation and Curation
- Target Property: Define the property to be predicted (e.g., HOMO-LUMO gap, energy).
- Reference Data: Generate a dataset of molecular structures and their corresponding properties using a high-level ab initio method (e.g., CCSD(T) or DFT) for a representative training set.
2. Model Training and Validation
- Molecular Representation: Convert molecules into features. Use graphs (atoms as nodes, bonds as edges) or 3D geometry images for richer structural information [60].
- Learning Framework: Employ a Graph Neural Network (GNN) or a contrastive learning method like GraphGIM, which uses both 2D graphs and multi-view 3D geometry images to learn robust molecular representations [60].
- Training: Train the ML model to map the molecular representation to the target property.
- Validation: Rigorously test the model on a held-out test set not seen during training to assess predictive performance.
3. Prediction and Deployment
- Use the trained model to rapidly screen large virtual chemical libraries for molecules with desired properties.
Diagram 2: Machine learning with ab initio data workflow.
Protocol: Refining Predicted Protein Structures with MD
Application Objective: To refine and validate a computationally predicted protein structure (e.g., from AlphaFold2, Robetta, or I-TASSER) to obtain a reliable structural model for structure-based drug design [61].
1. Initial Structure Prediction
- Use one or more de novo (AF2, Robetta, trRosetta) or template-based (MOE, I-TASSER) tools to generate an initial 3D model of the target protein [61].
2. Molecular Dynamics Refinement
- System Preparation: Place the predicted structure in a solvated periodic box with ions.
- Equilibration: Perform energy minimization and gradual heating followed by equilibrium MD run under NPT conditions.
- Production MD: Run a multi-nanosecond MD simulation to allow the structure to relax and sample stable conformations.
3. Model Quality Assessment
- Convergence Metrics: Monitor the Root Mean Square Deviation (RMSD) of the protein backbone and the Radius of Gyration (Rg) to ensure structural stability [61].
- Quality Checks: Use validation tools like ERRAT and phi–psi Ramachandran plots to evaluate the stereochemical quality of the refined model before use in downstream applications [61].
The Scientist's Toolkit: Essential Research Reagents and Software
Table 2: Key Computational Tools for Hybrid and Multi-Level Strategies
| Tool Name | Type/Category | Primary Function in Research | Application Context |
|---|---|---|---|
| GROMACS [58] | Molecular Dynamics Engine | High-performance MD and QM/MM MD simulations for sampling and dynamics. | Simulating biomolecular dynamics; running QM/MM simulations with packages like MiMiC. |
| MiMiC [58] | QM/MM Framework | Flexible, multi-program framework for running advanced QM/MM simulations. | Coupling various QM and MM programs for studying catalysis and reactivity in biomolecules. |
| PyMOL [62] | Molecular Visualization | Rendering, analyzing, and presenting 3D molecular structures and trajectories. | Visualizing protein-ligand complexes, MD trajectories, and structural models. |
| Coupled Cluster Codes (e.g., CFOUR, MRCC) | Quantum Chemistry | Providing benchmark-level accuracy for energy and property calculations. | Generating reference data for ML training; calculating accurate energies for small systems. |
| GraphGIM [60] | Machine Learning Model | Molecular representation learning via contrastive learning on graphs and images. | Pre-training models for accurate prediction of molecular properties in drug discovery. |
| MOE [61] | Integrated Software Suite | Homology modeling, molecular modeling, and simulation for drug discovery. | Constructing protein structures via homology modeling; structure-based design. |
The integration of hybrid QM/MM simulations, machine learning, and classical molecular dynamics represents a powerful paradigm for tackling the complexity of molecular systems in drug development. By strategically combining the unparalleled accuracy of post-Hartree-Fock methods with the scale of molecular mechanics and the predictive speed of machine learning, researchers can achieve a balance that no single approach can offer. The protocols and tools detailed herein provide a roadmap for scientists to implement these multi-level strategies, driving forward the rational design of therapeutics and the understanding of biological processes at an atomistic level.
Benchmarking Performance and Validating Results for Reliable Predictions
Benchmarking Against Experimental Data and High-Level Theory
Benchmarking computational chemistry methods is a critical practice for validating their accuracy and establishing their domain of applicability. For post-Hartree-Fock methods, which aim to capture electron correlation effects neglected in mean-field approaches, benchmarking typically follows a dual-path strategy: comparison against high-level theoretical reference data and validation against experimental observations [16]. This protocol outlines detailed methodologies for conducting such benchmarks, enabling researchers to assess the performance of electronic structure methods for molecular calculations.
The fundamental challenge in quantum chemistry is the accurate and efficient computation of electron correlation energy [11] [63]. As the field progresses with new methodological developments, rigorous benchmarking becomes indispensable for guiding method selection, particularly for applications in drug development and materials design where predictive accuracy is paramount.
Benchmarking Methodologies
Theoretical Benchmarking Against High-Level Reference Data
Theoretical benchmarking involves comparing target methods against highly accurate wavefunction-based theories on carefully designed test sets. Coupled Cluster with Single, Double, and perturbative Triple excitations (CCSD(T)) is widely regarded as the "gold standard" for molecular energetics, providing reference data where experimental measurements are scarce or unreliable [16].
Table 1: High-Level Theoretical Methods for Benchmarking
| Method | Theoretical Description | Typical Applications | Scalability |
|---|---|---|---|
| CCSD(T) | Coupled Cluster with Single, Double, and perturbative Triple excitations | Energetics, spectroscopic constants | O(N⁷) |
| CASSCF/NEVPT2 | Complete Active Space Self-Consistent Field with N-electron Valence Perturbation Theory | Multiconfigurational systems, excited states | Depends on active space |
| MP2 | Møller-Plesset 2nd Order Perturbation Theory | Initial benchmarking, large systems | O(N⁵) |
For systems with strong multiconfigurational character, such as the NV⁻ center in diamond, the CASSCF/NEVPT2 protocol provides an alternative benchmarking approach [12]. This method combines active space selection with perturbation theory to capture both static and dynamic correlation effects in challenging electronic structures.
Experimental Benchmarking
Experimental benchmarking validates computational methods against empirically measured properties. Key benchmark properties include:
- Molecular hyperpolarizabilities for nonlinear optical materials [19]
- Zero-phonon lines and excitation energies for color centers [12]
- Molecular structures from techniques like Hirshfeld atom refinement [64]
- Reaction energies and barrier heights for kinetic studies
When designing experimental benchmarks, careful consideration must be given to the experimental conditions and uncertainties, as these directly impact the validation process [19].
Protocol 1: Information-Theoretic Approach for Correlation Energy Prediction
Background and Principle
The Information-Theoretic Approach (ITA) provides a cost-efficient method for predicting post-Hartree-Fock electron correlation energies using density-based descriptors derived from Hartree-Fock calculations [11] [49] [63]. This approach exploits linear relationships between information-theoretic quantities and correlation energies, enabling accurate predictions at a fraction of the computational cost of traditional post-Hartree-Fock methods.
Materials and Computational Setup
Table 2: Research Reagent Solutions for ITA Protocol
| Reagent/Resource | Function/Purpose | Implementation Notes |
|---|---|---|
| Quantum Chemistry Software (e.g., PySCF) | Performs electronic structure calculations | Requires Hartree-Fock and post-HF capability |
| ITA Quantity Calculator | Computes density-based descriptors | Custom code often required |
| Linear Regression Module | Establishes correlation between ITA quantities and target energies | Standard statistical packages |
| 6-311++G(d,p) Basis Set | Standardized basis for benchmarking | Balanced accuracy/cost trade-off |
Step-by-Step Procedure
System Selection and Preparation
- Select a representative set of molecular systems (e.g., 24 octane isomers, polymeric structures, molecular clusters)
- Generate optimized molecular geometries using Hartree-Fock or DFT methods
Reference Data Generation
- Calculate reference electron correlation energies using post-Hartree-Fock methods (MP2, CCSD, or CCSD(T)) with a consistent basis set (e.g., 6-311++G(d,p))
- For large molecular clusters, employ linear-scaling methods like Generalized Energy-Based Fragmentation (GEBF) to obtain accurate reference data [11]
ITA Quantity Computation
- Perform Hartree-Fock calculations for all systems
- Compute a set of 11 information-theoretic quantities from the electron density:
- Shannon entropy (Sₛ)
- Fisher information (IF)
- Ghosh-Berkowitz-Parr entropy (SGBP)
- Onicescu information energies (E₂, E₃)
- Relative Rényi entropies (R₂ʳ, R₃ʳ)
- Relative Shannon entropy (I_G)
- Relative Fisher information (G₁, G₂, G₃)
Linear Regression Analysis
- Establish linear relationships between ITA quantities and reference correlation energies
- Validate the predictive model using statistical measures (R² values, RMSD)
- Assess accuracy against chemical accuracy threshold (1 kcal/mol or 1.6 mH)
Diagram 1: ITA Correlation Energy Prediction Workflow
Applications and Validation
The LR(ITA) protocol has been successfully applied to diverse chemical systems:
- Octane isomers: Fisher information (I_F) achieves R² = 0.987 with RMSD of 0.6 mH for MP2 correlation energies [11]
- Polymeric systems: Linear relationships (R² ≈ 1.000) with RMSDs of 1.5-11 mH across polyyne, polyene, and acene structures [11]
- Molecular clusters: Accurate prediction for hydrogen-bonded, dispersion-bound, and metallic clusters with RMSDs ranging 2-42 mH depending on system type [11]
Protocol 2: Benchmarking Hyperpolarizability Calculations
Background and Principle
This protocol benchmarks computational methods for predicting molecular first hyperpolarizability (β), a key property in nonlinear optics and molecular electronics design [19]. The approach emphasizes pairwise ranking preservation, which is crucial for evolutionary design algorithms where relative ordering matters more than absolute accuracy.
Materials and Computational Setup
Table 3: Research Reagent Solutions for Hyperpolarizability Benchmarking
| Reagent/Resource | Function/Purpose | Implementation Notes |
|---|---|---|
| Push-Pull Chromophore Set | Representative test molecules | 5 compounds with experimental β values |
| Finite Field Method Module | Calculates hyperpolarizability | Numerical differentiation of dipole moments |
| Quantum Chemistry Code | Performs HF/DFT calculations | PySCF recommended |
| Experimental Reference Data | Validation benchmark | Kanis et al. dataset |
Step-by-Step Procedure
Test System Selection
- Curate a set of push-pull chromophores with reliable experimental hyperpolarizability values
- Ensure diversity in molecular size and β values (e.g., pNA to nitrostilbene derivatives)
Computational Method Evaluation
- Test multiple functionals (HF, PBE0, B3LYP, CAM-B3LYP, M06-2X) across various basis sets (STO-3G to 6-311G(d))
- Perform geometry optimizations at consistent theory levels
- Calculate hyperpolarizability using finite field method with field strength h = 0.001 au
Performance Metrics Calculation
- Compute Mean Absolute Percentage Error (MAPE) for absolute accuracy assessment
- Evaluate pairwise rank agreement using equation:
- Pairwise Agreement = (1/ⁿC₂) Σ 𝕀[sgn(βcalc,i - βcalc,j) = sgn(βexp,i - βexp,j)]
- Measure computational cost (wall-clock time)
Pareto Optimality Analysis
- Identify methods that provide optimal accuracy-speed trade-offs
- Select recommended methods for specific applications
Diagram 2: Hyperpolarizability Benchmarking Protocol
Results and Interpretation
For hyperpolarizability benchmarking of push-pull chromophores:
- HF/3-21G provides the best balance: 45.5% MAPE with perfect pairwise ranking in 7.4 minutes per molecule [19]
- All 30 tested method combinations maintained perfect pairwise agreement (10/10 pairs)
- Basis set selection has greater impact than functional choice for these systems
- Pareto analysis identifies HF/STO-3G and HF/3-21G as optimal trade-offs
Protocol 3: Multireference Systems Benchmarking for Color Centers
Background and Principle
Point defects in semiconductors, such as the NV⁻ center in diamond, present significant challenges for computational methods due to their multiconfigurational character [12]. This protocol describes a wavefunction theory-based benchmarking approach combining CASSCF with NEVPT2 to address both static and dynamic correlation effects.
Materials and Computational Setup
Table 4: Research Reagent Solutions for Color Center Benchmarking
| Reagent/Resource | Function/Purpose | Implementation Notes |
|---|---|---|
| Cluster Models | Finite representation of crystal environment | Hydrogen-terminated nanodiamonds |
| Active Space Orbitals | Treatment of static correlation | CASSCF(6e,4o) for NV⁻ center |
| NEVPT2 Module | Dynamic correlation correction | Second-order perturbation theory |
| Experimental Spectroscopy Data | Reference for excitation energies | Zero-phonon lines, fine structure |
Step-by-Step Procedure
Cluster Model Construction
- Generate hydrogen-terminated diamond clusters of increasing size
- Freeze atomic positions in outer shells to mimic crystalline environment
- Conduct convergence tests with respect to cluster size
Active Space Selection
- Identify defect-localized molecular orbitals (4 orbitals for NV⁻ center)
- Define active electrons (6 electrons for NV⁻ center)
- Establish CASSCF(6e,4o) active space
State-Specific Calculations
- Perform state-specific CASSCF for equilibrium geometries of target electronic states
- Calculate NEVPT2 dynamic correlation corrections
- Optimize geometries for individual electronic states
Property Calculation and Validation
- Compute energy levels of electronic states involved in polarization cycle
- Determine Jahn-Teller distortion effects on measurable properties
- Calculate fine structure of ground and excited states
- Compare with experimental zero-phonon lines and pressure dependence
Applications and Results
The CASSCF/NEVPT2 protocol successfully models challenging properties of the NV⁻ center:
- Accurate computation of electronic states involved in the polarization cycle
- Prediction of hitherto uncharacterized high-lying excited states
- Quantification of Jahn-Teller distortion effects on measurable properties
- Calculation of fine structure and pressure dependence of zero-phonon lines
Emerging Trends and Future Directions
The field of computational chemistry benchmarking is evolving with several promising developments:
- Machine Learning Integration: Hybrid models that combine physical principles with data-driven corrections are enhancing predictive accuracy while maintaining computational efficiency [16]
- Quantum Computing: Algorithms like Variational Quantum Eigensolver (VQE) and Quantum Phase Estimation (QPE) offer potential for strongly correlated systems, though current implementations face hardware limitations [16]
- Fragment-Based Methods: Approaches like the Fragment Molecular Orbital (FMO) method and Generalized Energy-Based Fragmentation (GEBF) enable accurate treatment of large systems [11] [16]
- Automated Method Selection: Benchmarking data is increasingly used to develop protocols for automatic method selection based on system characteristics [16] [19]
These advances are gradually narrowing the gap between computational predictions and experimental observations across diverse chemical applications.
The Hartree-Fock (HF) method serves as the foundational starting point in ab initio quantum chemistry, but it possesses a critical limitation: it does not account for electron correlation, the instantaneous repulsive interactions between electrons [1]. This missing correlation energy is essential for achieving quantitative accuracy in predicting molecular energies, structures, and properties. Post–Hartree–Fock methods comprise a set of computational approaches developed specifically to address this shortcoming by adding a more accurate description of electron repulsions [1] [7].
These methods are crucial in fields like drug development, where accurate predictions of molecular interaction energies, conformational preferences, and spectroscopic properties can significantly impact the design of new therapeutic molecules. The choice of an appropriate electronic structure method is a critical decision, balancing computational cost against the required accuracy. This application note provides a structured comparison of popular post-HF methods to guide researchers in making this choice effectively.
Key Post-HF Methods
- Møller-Plesset Perturbation Theory (MPn): This approach treats electron correlation as a small perturbation to the Hartree-Fock Hamiltonian. The second-order correction, MP2, is the most widely used due to its favorable balance of cost and accuracy. It is an economical method for partially correcting for the lack of dynamic electron correlation and can describe van der Waals forces [65]. Higher-order corrections (MP3, MP4) are available but less commonly used due to their increased computational cost without clear advantages for many applications [65].
- Coupled Cluster (CC) Theory: This method uses an exponential wavefunction ansatz to capture electron correlation in a size-extensive manner, meaning it performs equally well for systems of different sizes [7]. CCSD includes all single and double excitations. The CCSD(T) method, which adds a perturbative treatment of triple excitations, is often called the "gold standard" of quantum chemistry for its high accuracy in predicting energies and geometries [65].
- Configuration Interaction (CI): CI constructs the wavefunction as a linear combination of the HF determinant and excited-state determinants. While Full CI is exact within the chosen basis set, it is only feasible for very small systems [65] [7]. Truncated methods, like CISD, are limited by their lack of size-extensivity, which hampers their application to systems of varying size [7].
Critical Concepts for Applications
- Size-Consistency/Extensivity: A method is size-consistent if the energy of two infinitely separated molecules equals the sum of the energies of the individual molecules. This property is vital for studying reaction energies and interaction energies. CC methods are size-extensive, while truncated CI is not [7].
- Basis Set Dependence: The accuracy of all post-HF methods is intrinsically linked to the quality of the atomic basis set used [65] [7]. Larger, more flexible "correlation-consistent" basis sets (e.g., cc-pVXZ) are typically required to achieve meaningful results, which increases computational cost [65].
- Computational Scaling: The computational cost of these methods, often expressed as a formal scaling with the number of basis functions (N), rises dramatically, creating a practical limit on the size of systems that can be studied.
Comparative Analysis of Methods
The following tables provide a quantitative and qualitative comparison of key post-HF methods to guide method selection.
Table 1: Key Characteristics of Post-Hartree-Fock Methods
| Method | Key Description | Formal Computational Scaling | Size-Extensive? | Key Strengths | Key Limitations |
|---|---|---|---|---|---|
| MP2 | 2nd-order Møller-Plesset Perturbation Theory [65] | N⁵ [65] | Yes [65] | Good for dynamic correlation; good structures/IR spectra; describes van der Waals forces [65] | Can be unreliable for systems with significant non-dynamic correlation [65] |
| MP4 | 4th-order Møller-Plesset Perturbation Theory [65] | N⁷ | Yes | More complete correlation treatment than MP2 | Significantly more expensive than MP2; advantages not always clear [65] |
| CCSD | Coupled Cluster with Singles & Doubles [65] | N⁶ | Yes [7] | High accuracy for energies/geometries [65] | High cost; iterative calculations; scaling limits application [65] [7] |
| CCSD(T) | CCSD with perturbative Triples [65] | N⁷ | Yes [7] | "Gold standard" for accuracy; excellent for geometries/formation energies [65] | Very high computational cost; limited to small molecules [65] [7] |
| CISD | Configuration Interaction with Singles & Doubles [7] | N⁶ | No [7] | Conceptual simplicity; variational upper bound [7] | Lacks size-extensivity; performance degrades for larger systems [65] [7] |
Table 2: Typical Application Scope and Accuracy
| Method | Typical Max System Size (Heavy Atoms) | Recommended Basis Sets | Accuracy for Geometries (Bond Lengths) | Accuracy for Thermochemistry |
|---|---|---|---|---|
| MP2 | Tens of atoms (with local approx.) [65] | cc-pVTZ, cc-pVQZ [65] | Good (within ~0.02 Å for standard bonds) [65] | Moderate; can be unbalanced |
| CCSD | ~10s of atoms | cc-pVTZ, cc-pVQZ [65] | High (within ~0.01 Å) [65] | Good |
| CCSD(T) | ~10-20 atoms | cc-pVTZ, cc-pVQZ [65] | Very High (within ~0.005 Å) [65] | Very High (sub-kcal/mol) |
| CISD | ~10s of atoms | Depends on system | Moderate | Poor for large systems due to size-extensivity error [7] |
Decision Workflow for Method Selection
The following diagram provides a logical workflow for selecting an appropriate electronic structure method based on the research objective and system characteristics.
Detailed Protocols for Key Calculations
Protocol 1: MP2 Geometry Optimization and Frequency Calculation
This protocol is suitable for optimizing molecular structures and calculating vibrational frequencies for medium-sized molecules with a single-reference character.
- Objective: To determine a minimum-energy molecular structure and verify it is a true minimum by confirming the absence of imaginary frequencies in the vibrational spectrum.
- Software Requirements: Gaussian, ORCA, PSI4, Q-Chem, or other ab initio packages with MP2 and analytical gradient capabilities.
- Input Specifications:
- Method:
MP2 - Basis Set:
cc-pVTZfor a good balance of accuracy and cost. For higher accuracy, usecc-pVQZ[65]. - Job Type:
Optimization(Opt) followed byFrequency(Freq) calculation on the optimized geometry. - Charge and Multiplicity: Set appropriately for the system.
- Method:
- Step-by-Step Instructions:
- Build Initial Geometry: Construct a reasonable initial molecular structure using a graphical builder or from crystallographic data.
- Prepare Input File: The input file should specify the method, basis set, and job type. Example Gaussian Input:
- Run Calculation: Submit the job to the computational queue.
- Analyze Results:
- Geometry: Extract the optimized Cartesian coordinates from the output file.
- Energy: Note the final MP2 energy (in Hartrees).
- Frequencies: Confirm all vibrational frequencies are real (positive). A single imaginary frequency may indicate a transition state.
- Troubleshooting:
- Convergence Failure: Loosen the optimization convergence criteria (
Opt=Loose) or provide a better initial guess. - High Cost for Large Systems: Use the local MP2 approximation (LMP2) if available in the software (e.g., in Jaguar) [65].
- Convergence Failure: Loosen the optimization convergence criteria (
Protocol 2: High-Accuracy Single-Point Energy with CCSD(T)
This protocol is used to compute a highly accurate electronic energy for a structure (often pre-optimized with a cheaper method) to obtain benchmark-quality reaction energies, barrier heights, or interaction energies.
- Objective: To calculate a near-exact electronic energy for a molecular system at a given geometry.
- Software Requirements: CFOUR, MRCC, Gaussian, ORCA, or Dalton. Efficient CCSD(T) optimizations can be implemented in Dalton [65].
- Input Specifications:
- Method:
CCSD(T) - Basis Set:
cc-pVQZor larger. To approach the complete basis set (CBS) limit, use a composite scheme or extrapolation [65]. - Job Type:
Single-Point Energy(oftenSPorEnergy). - Frozen Core Approximation: Typically used to reduce cost by excluding core electrons from the correlation treatment (e.g.,
Frozen Core).
- Method:
- Step-by-Step Instructions:
- Obtain Input Geometry: Use a geometry optimized at a high level of theory (e.g., MP2/cc-pVTZ or CCSD/cc-pVTZ).
- Prepare Input File: The input is simpler than for an optimization. Example Dalton Input Structure (Geometry in Bohr) [65]: (Note: The molecule specification must precede this block.)
- Run Calculation: Submit the job. CCSD(T) calculations are computationally demanding and may require significant time and resources.
- Analyze Results: Extract the final CCSD(T) energy from the output. This energy can be used in thermodynamic cycles.
- Troubleshooting:
- Prohibitive Cost: If the system is too large for CCSD(T), consider using the cheaper DLPNO-CCSD(T) approximation available in packages like ORCA.
- Basis Set Limitations: If a large basis set is not feasible, apply a CBS extrapolation from results with cc-pVTZ and cc-pVQZ basis sets [65].
The Scientist's Toolkit: Essential Research Reagents
Table 3: Essential Software and Basis Sets for Post-HF Calculations
| Item Name | Type | Function/Description | Example Use Case |
|---|---|---|---|
| cc-pVXZ Family | Basis Set | Correlation-consistent polarized Valence X-tuple Zeta basis sets; systematically improve accuracy with increasing X (D,T,Q,5,...) [65]. | Achieving high accuracy in correlation energy calculations; CBS extrapolation [65]. |
| Frozen Core Approximation | Computational Technique | Reduces cost by excluding core electrons from post-HF correlation treatment. | Essential for applying CCSD(T) to molecules with atoms beyond the first row [65]. |
| Local Correlation (e.g., LMP2) | Algorithmic Improvement | Ignores correlation between distant electrons, reducing formal scaling [65]. | Enabling MP2 calculations on large molecules (e.g., drug-sized molecules like paclitaxel) [65]. |
| Gaussian, ORCA, PSI4 | Software Package | Comprehensive ab initio quantum chemistry programs. | Performing a wide range of post-HF calculations (MP2, CCSD(T), CI) in a user-friendly environment. |
| Dalton | Software Package | A program geared toward calculating molecular properties (NMR, UV/Vis, etc.) [65]. | Efficient CCSD(T) optimizations and advanced spectroscopic property calculations [65]. |
| Extrapolation to CBS | Computational Protocol | Estimates the complete basis set limit energy using calculations with two successive basis sets [65]. | Achieving near-exact energies without the prohibitive cost of an infinitely large basis set. |
Accurately predicting the properties of molecular clusters and polymers is a central challenge in computational chemistry, with significant implications for materials science and drug development. Post-Hartree-Fock (post-HF) methods, such as Møller-Plesset perturbation theory (MP2) and coupled cluster (CCSD and CCSD(T)), are considered the gold standard for computing electron correlation energy, which is crucial for predicting molecular behavior and interactions [11]. However, these methods are notoriously computationally expensive, with costs that skyrocket as system size increases, making their application to large, complex systems like polymers and molecular clusters prohibitively slow and resource-intensive [11]. This limitation creates a pressing need for alternative, cost-efficient methods that can deliver high accuracy across diverse chemical systems.
This case study explores the Information-Theoretic Approach (ITA) as a promising framework for predicting post-HF electron correlation energies at a fraction of the computational cost. We assess the accuracy of the Linear Regression ITA (LR(ITA)) protocol across a wide range of complex systems, including organic polymers and various types of molecular clusters, validating its performance against traditional post-HF calculations [11]. The findings are framed within a broader research thesis on advancing post-HF methodologies for efficient and accurate molecular calculations.
Computational Framework
The Information-Theoretic Approach (ITA) and LR(ITA) Protocol
The Information-Theoretic Approach (ITA) reframes the challenge of electron correlation by treating the electron density as a continuous probability distribution. It leverages a suite of physical descriptors to quantify information contained within the density, bypassing the explicit, costly computation of electron-electron interactions [11]. These descriptors are inherently basis-set agnostic and physically interpretable, providing deep insights into electronic structure.
Key ITA quantities used in this study include [11]:
- Shannon Entropy ((S_S)): Characterizes the global delocalization of the electron density.
- Fisher Information ((I_F)): Quantifies local inhomogeneity and the sharpness of density features.
- Ghosh, Berkowitz, and Parr Entropy ((S_{GBP})): Provides another measure of entropy based on electron density.
- Onicescu Information Energy ((E2) and (E3)): Reflects the concentration of the electron distribution.
- Relative Rényi Entropy ((R2^r) and (R3^r)) and Relative Shannon Entropy ((I_G)): Measure the distinguishability between different electron densities.
- Relative Fisher Information ((G1), (G2), and (G_3)): Gauges the local distinguishability between densities.
The LR(ITA) protocol establishes a strong linear relationship between these low-cost, Hartree-Fock-level ITA quantities and the high-level post-HF electron correlation energy (e.g., from MP2, CCSD, or CCSD(T) calculations). A linear regression model is trained on a set of molecules, creating a predictive tool that can estimate the correlation energy for new systems using only a Hartree-Fock calculation [11]. For very large systems, the protocol can be integrated with a linear-scaling method like the Generalized Energy-Based Fragmentation (GEBF) approach to further enhance computational efficiency without sacrificing accuracy [11].
Advanced Machine Learning in Molecular Property Prediction
Concurrently, the field of molecular property prediction has seen significant advances through deep learning. The Self-Conformation-Aware Graph Transformer (SCAGE) is one such architecture pretrained on approximately 5 million drug-like compounds [66]. Its multitask pretraining framework, M4, incorporates molecular fingerprint prediction, functional group prediction, 2D atomic distance prediction, and 3D bond angle prediction, enabling it to learn comprehensive molecular representations from structure to function [66]. While distinct from the LR(ITA) method, SCAGE represents the cutting edge in leveraging molecular information for accurate property prediction and provides a complementary perspective on the importance of sophisticated computational protocols.
Accuracy Assessment in Polymers
Systems and Computational Methodology
The accuracy of the LR(ITA) protocol was evaluated on a series of linear and quasi-linear organic polymers, which are characterized by delocalized electronic structures that pose a challenge for computational methods [11]. The systems studied were:
- Polyyne: Linear chains of alternating single and triple bonds.
- Polyene: Linear chains of alternating single and double bonds.
- All-trans-polymethineimine: A polymer with a conjugated backbone.
- Acene: Linear fused benzene rings.
The computational protocol was as follows:
- Geometry Optimization: Structures were optimized at the HF/6-311++G(d,p) level of theory.
- ITA Quantity Calculation: The 11 ITA quantities (e.g., (SS), (IF), (S_{GBP})) were computed from the HF electron density.
- Reference Correlation Energy: The target electron correlation energy was calculated using MP2/6-311++G(d,p).
- Model Building and Validation: Linear regression models were built for each ITA quantity, and their predictive accuracy was assessed by comparing the LR(ITA)-predicted correlation energies to the actual MP2 values.
Quantitative Results and Analysis
Table 1: Accuracy of LR(ITA) for Predicting MP2 Correlation Energies in Polymers
| Polymer System | Top ITA Quantities | Linear Correlation (R²) | Root Mean Squared Deviation (RMSD) |
|---|---|---|---|
| Polyyne | Multiple (excluding G2) | ≈1.000 | ~1.5 mH |
| Polyene | Multiple (excluding G2, I_G) | ≈1.000 | ~3.0 mH |
| All-trans-polymethineimine | Multiple (excluding G1, G2, I_G) | ≈1.000 | <4.0 mH |
| Acene | Multiple | ≈1.000 | ~10–11 mH |
The data demonstrates that the LR(ITA) protocol achieves remarkable predictive accuracy for the tested polymers, with near-perfect linear correlations (R² ≈ 1.000) for most ITA quantities [11]. The very low RMSDs for polyyne, polyene, and polymethineimine indicate that a single ITA quantity can capture sufficient information about the electron correlation energy in these systems with delocalized electronic structures [11]. The slightly higher, though still reasonable, RMSD for acenes suggests their more complex delocalized electronic structure requires more nuanced descriptors for quantitative prediction [11].
Figure 1: Experimental workflow for assessing LR(ITA) accuracy in polymers. The protocol uses HF calculations to derive ITA quantities, which are used in a linear regression model to predict MP2-level correlation energies.
Accuracy Assessment in Molecular Clusters
Systems and Computational Methodology
The study was extended to diverse molecular clusters to test the transferability and robustness of the LR(ITA) protocol. These clusters represent different types of dominant intermolecular interactions [11]:
- Metallic Clusters: Ben and Mgn clusters.
- Covalent Clusters: Sn clusters.
- Hydrogen-Bonded Clusters: Protonated water clusters, H+(H2O)n.
- Dispersion-Bound Clusters: Carbon dioxide (CO2)n and benzene (C6H6)n clusters.
The computational methodology remained consistent with the polymer study: ITA quantities from HF/6-311++G(d,p) calculations were used in a linear regression model to predict MP2/6-311++G(d,p) correlation energies.
Quantitative Results and Analysis
Table 2: Accuracy of LR(ITA) for Predicting MP2 Correlation Energies in Molecular Clusters
| Cluster Type | Example Systems | Linear Correlation (R²) | Root Mean Squared Deviation (RMSD) |
|---|---|---|---|
| Metallic | Ben, Mgn | >0.990 | ~28–37 mH |
| Covalent | Sn | >0.990 | ~26–42 mH |
| Hydrogen-Bonded | H+(H2O)n | ≈1.000 | 2.1–9.3 mH |
| Dispersion-Bound | (C6H6)n, (CO2)n | Similar accuracy to GEBF (Not specified) | Similar accuracy to GEBF (Not specified) |
The results show a key distinction. While strong linear correlations (R² > 0.990) exist for all cluster types, indicating the extensivity of the ITA quantities, the quantitative accuracy varies significantly [11]. For 3D metallic and covalent clusters, the RMSD errors are substantially larger, suggesting that a single ITA quantity cannot quantitatively capture enough information about the complex electron correlation energies in these systems [11].
In contrast, the LR(ITA) protocol performs exceptionally well for hydrogen-bonded systems like protonated water clusters, achieving near-perfect correlation and low RMSDs [11]. For large dispersion-bound benzene clusters, the LR(ITA) method demonstrated similar high accuracy to the linear-scaling Generalized Energy-Based Fragmentation (GEBF) method, underscoring its utility for large, weakly-bound systems [11].
Experimental Protocols
Protocol 1: LR(ITA) for Correlation Energy Prediction
This protocol details the steps for applying the LR(ITA) method to predict post-HF electron correlation energies for a set of molecules (e.g., a homologous series of polymers or clusters).
1. System Preparation and Geometry Optimization
- Input: Molecular structures (SMILES, Cartesian coordinates).
- Software: Quantum chemistry package (e.g., Gaussian, GAMESS, Psi4).
- Method: Perform geometry optimization and frequency calculation at the HF/6-311++G(d,p) level to ensure a stable minimum on the potential energy surface.
- Output: Optimized molecular geometry.
2. Reference Post-HF Single-Point Energy Calculation
- Method: Perform a single-point energy calculation on the optimized geometry using a post-HF method (e.g., MP2, CCSD, or CCSD(T)) with the 6-311++G(d,p) basis set.
- Output: Total energy from the post-HF method ((E{\text{post-HF}})) and the HF method ((E{\text{HF}})).
- Calculation: Compute the reference electron correlation energy as (E{\text{corr}} = E{\text{post-HF}} - E_{\text{HF}}).
3. Information-Theoretic Quantity Calculation
- Software: A code capable of computing ITA quantities from the electron density (e.g., a custom script interfacing with the quantum chemistry software's output).
- Input: The electron density from the HF/6-311++G(d,p) calculation.
- Action: Calculate the suite of 11 ITA quantities ((SS), (IF), (S{GBP}), (E2), (E3), (R2^r), (R3^r), (IG), (G1), (G2), (G_3)) for each molecule.
4. Linear Regression Model Building
- Software: Data analysis environment (e.g., Python with scikit-learn, R).
- Action: For each ITA quantity, perform a simple linear regression with the ITA quantity as the independent variable and the reference (E_{\text{corr}}) as the dependent variable.
- Output: A set of linear equations (one per ITA quantity) of the form (E_{\text{corr}}^{\text{predicted}} = m \times \text{ITA} + b).
5. Model Validation and Prediction
- Action: Use the regression models to predict the correlation energy for validation molecules or new systems of the same class.
- Validation Metrics: Calculate the coefficient of determination (R²) and Root Mean Squared Deviation (RMSD) between predicted and calculated correlation energies to assess accuracy.
Protocol 2: Accuracy Assessment with Generalized Energy-Based Fragmentation (GEBF)
For very large molecular clusters, this protocol integrates GEBF to make the reference MP2 calculation feasible.
1. System Fragmentation
- Software: A GEBF-enabled program.
- Action: Fragment the large target cluster into smaller, manageable subsystems according to the GEBF scheme.
2. Subsystem Calculations
- Action: Perform HF and MP2 calculations (using the 6-311++G(d,p) basis set) on each subsystem and relevant charge-embedded subsystems.
3. Energy Assembly
- Action: Re-assemble the total MP2 and HF energies of the target cluster from the subsystem energies using the GEBF formula.
- Output: The total MP2 correlation energy for the large cluster, serving as the reference value.
4. LR(ITA) Application and Comparison
- Action: Perform an HF calculation for the entire cluster, compute ITA quantities, and predict the correlation energy using the pre-trained LR(ITA) model.
- Action: Compare the LR(ITA)-predicted correlation energy to the GEBF-derived MP2 correlation energy to validate the accuracy of the LR(ITA) method for large systems.
Table 3: Key Reagents and Computational Resources for Molecular Accuracy Studies
| Item Name | Specifications / Function | Application Context |
|---|---|---|
| Basis Set | 6-311++G(d,p) - A triple-zeta basis set with diffuse and polarization functions. | Standard basis set for HF and post-HF calculations to ensure a balanced description of electron correlation [11]. |
| Reference Molecules | Octane Isomers (24 structures) - A set of molecules with the same formula but different connectivity. | Used as a benchmark set to validate the initial accuracy of the LR(ITA) protocol [11]. |
| Polymer Systems | Polyyne, Polyene, Polymethineimine, Acene - Linear or quasi-linear organic polymers. | Test systems for assessing accuracy in structures with delocalized electronic structures [11]. |
| Cluster Systems | (H₂O)ₙ, (C₆H₆)ₙ, (CO₂)ₙ, Beₙ, Mgₙ, Sₙ - Clusters with various bonding types. | Test systems for evaluating transferability across different intermolecular interactions [11]. |
| GEBF Software | Generalized Energy-Based Fragmentation method. | Linear-scaling method to obtain reference MP2 energies for large molecular clusters [11]. |
| SCAGE Model | Self-Conformation-Aware Graph Transformer - A deep learning model for molecular property prediction. | Provides an alternative, machine-learning-based approach for accurate property prediction with substructure interpretability [66]. |
| OMC25 Dataset | Open Molecular Crystals 2025 Dataset - Over 27 million DFT-relaxed molecular crystal structures. | A resource for training and benchmarking machine learning models on solid-state molecular systems [67]. |
Figure 2: Decision pathway for accuracy assessment of molecular clusters. The expected accuracy of the LR(ITA) protocol depends on the cluster's bonding type and size, guiding the choice of reference method.
This case study demonstrates that the Information-Theoretic Approach, particularly the LR(ITA) protocol, provides a robust and accurate framework for predicting post-Hartree-Fock electron correlation energies across a wide spectrum of complex systems. Its performance is exceptional for organic polymers and clusters dominated by hydrogen bonding or dispersion forces, achieving near-chemical accuracy at the computational cost of a Hartree-Fock calculation. The method shows slightly reduced but still informative quantitative accuracy for 3D metallic and covalent clusters. The integration of the LR(ITA) protocol with linear-scaling methods like GEBF extends its applicability to very large systems, which are otherwise intractable for conventional post-HF calculations. When combined with emerging machine learning tools like SCAGE and large-scale datasets such as OMC25, the ITA stands as a powerful component in the modern computational chemist's toolkit, paving the way for more efficient and insightful molecular calculations in drug development and materials science.
The accurate simulation of molecular electronic structure remains a fundamental challenge across computational chemistry, materials science, and drug discovery. Traditional ab initio methods, particularly those extending beyond the mean-field approximation of Hartree-Fock theory, face significant scalability limitations due to their steep computational cost with increasing system size and electron correlation effects. This document details emerging application notes and experimental protocols that leverage quantum computing and advanced machine learning (ML) potentials to overcome these barriers, providing a practical framework for researchers engaged in post-Hartree-Fock molecular calculations.
Application Notes: Current Landscape and Performance
Quantum Computing Applications
Recent advances demonstrate that quantum processors are transitioning from theoretical promise to verifiable utility in quantum chemistry, enabling explorations of complex electronic phenomena that challenge classical computational methods.
Table 1: Representative Quantum Computing Applications in Molecular Simulation
| Application Focus | Key System/Molecule | Methodology | Hardware/Qubit Count | Reported Performance/Accuracy |
|---|---|---|---|---|
| Spin Ladder Calculation [68] | Mn₄O₅Ca (Photosynthesis) | Simplified Hamiltonian + Multi-qubit Gates (Neutral Atoms) | N/A (Theoretical) | Reduced gate count & coherence time vs. 2-qubit gates |
| Molecular Geometry & NMR Simulation [69] | 15-Atom & 28-Atom Molecules | Quantum Echoes Algorithm (OTOC) | Google's Willow Chip | 13,000x faster vs. classical supercomputer; Matched traditional NMR |
| Biomolecular Conformer Energy Calculation [70] | Cyclohexane Conformers, H₁₈ Ring | DMET-SQD (Hybrid Quantum-Classical) | 27-32 Qubits (IBM) | Energy differences within 1 kcal/mol of CCSD(T)/HCI benchmarks |
Advanced Machine Learning Potentials
Machine learning models are creating new paradigms by acting as surrogates for costly quantum mechanical computations, with a recent shift towards predicting intermediate quantum properties.
Table 2: Emerging Machine Learning Potentials and Differentiable Frameworks
| ML Model/Framework | Target System | Learning Target | Key Innovation | Application/Performance |
|---|---|---|---|---|
| Differentiable Framework [71] | Molecular & Condensed-Phase | Effective Electronic Hamiltonian | Integration with PySCFAD for indirect training | Reproduces properties (energy levels, dipole moments) from ML Hamiltonian |
| PoLiGenX [72] | Protein-Ligand Complexes | 3D Ligand Pose & Structure | Diffusion Model conditioned on reference poses | Reduced steric clashes and strain energies in generated ligands |
| LAGNet [72] | Organic Molecules | Electron Density | Core suppression model & Lebedev-Angular Grid | Decreased storage and computation costs for electron density learning |
| AttenhERG & CardioGenAI [72] | Small Molecules | hERG Toxicity & Redesign | Attentive FP & Autoregressive Transformer | High-accuracy toxicity prediction & generative redesign of scaffolds |
Experimental Protocols
Protocol: Hybrid DMET-SQD for Molecular Energy Calculation
This protocol details the hybrid quantum-classical approach for calculating relative energies of molecular conformers, as validated on current quantum hardware [70].
1. System Fragmentation via Density Matrix Embedding Theory (DMET):
- Input Preparation: Generate an initial mean-field wavefunction (e.g., Hartree-Fock) for the entire molecular system.
- Fragment Selection: Chemically relevant fragment is selected from the full molecule.
- Embedding: The fragment is embedded into an approximate bath, representing the electronic environment of the rest of the molecule, creating a smaller embedded problem.
2. Quantum Subspace Diagonalization via SQD:
- State Preparation: On the quantum processor, prepare a set of quantum states (configurations) derived from the embedded problem.
- Quantum Circuit Execution: For each configuration, run a series of quantum circuits on the fragment's qubit mapping to measure the Hamiltonian matrix elements within the subspace.
- Error Mitigation: Apply techniques such as gate twirling and dynamical decoupling to mitigate coherent errors and decoherence.
3. Classical Post-Processing and Iteration:
- Subspace Diagonalization: Classically diagonalize the measured Hamiltonian subspace to obtain the ground-state energy of the embedded fragment.
- Self-Consistency Loop: The DMET procedure uses the SQD-computed fragment energy to update the chemical potential, iterating until energy convergence is achieved across the whole molecule.
Protocol: Differentiable Workflow for ML-Hamiltonian Learning
This protocol outlines the training of machine learning models to predict electronic Hamiltonians, enabling the computation of multiple molecular properties through a differentiable quantum chemistry workflow [71].
1. Model Training and Hamiltonian Prediction:
- Input: Atomic compositions and geometries.
- ML Model Execution: A neural network model (e.g., Graph Neural Network) processes the input to predict the matrix elements of an effective electronic Hamiltonian in a specified basis set.
- Output: The predicted Hamiltonian (H_ML).
2. Differentiable Quantum Chemistry Processing:
- PySCFAD Integration: The H_ML is passed to PySCFAD, a fully differentiable quantum chemistry package.
- Property Calculation: Standard quantum operations are performed on HML within PySCFAD to compute derived properties such as:
- Electronic energy levels (by solving HMLψ = Eψ)
- Dipole moments
- Polarizabilities
3. Loss Calculation and Backpropagation:
- Target Comparison: The computed properties are compared against reference data (e.g., from high-level CCSD(T) calculations or experiment).
- Differentiable Backpropagation: The loss function gradient is backpropagated through the PySCFAD operations and further into the ML model, allowing for end-to-end training against property data, even without direct Hamiltonian labels.
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Computational Tools and Resources
| Tool/Resource | Type | Primary Function | Relevance to Post-Hartree-Fock Research |
|---|---|---|---|
| IBM Quantum System One [70] | Quantum Hardware | Execution of hybrid quantum-classical algorithms | Access to ~27-32 qubit processors for running VQE, SQD, and other algorithms on real hardware. |
| Google Willow Chip [69] | Quantum Hardware | Running complex, verifiable quantum algorithms | Enables advanced algorithms like Quantum Echoes for molecular spectroscopy and dynamics. |
| PySCFAD [71] | Software Library | Fully differentiable quantum chemistry calculations | Core component for end-to-end training of ML models that predict quantum mechanical operators. |
| Tangelo [70] | Software Library | Quantum chemical workflows and DMET implementation | Facilitates the classical component and interface for hybrid DMET-SQD calculations. |
| Gnina (v1.3) [72] | Software Tool | Protein-ligand docking with CNN scoring | Provides ML-based scoring functions, including for covalent docking, in structure-based drug design. |
| PennyLane Datasets [73] | Data Resource | Curated quantum chemistry data and molecules | Offers standardized benchmark systems (e.g., H₂, Cr₂, Fe-S clusters) for algorithm development. |
| fastprop [72] | Software Tool | Molecular property prediction | Rapid descriptor-based ML for ADMET and physicochemical properties, useful for initial screening. |
The synergistic integration of quantum computing and machine learning potentials is forging a new path in computational molecular science. Quantum algorithms, whether run on today's noisy devices or future error-corrected processors, offer a fundamentally scalable approach for solving strongly correlated electron problems. In parallel, differentiable ML potentials are creating powerful, scalable surrogates that maintain quantum-mechanical rigor. For researchers, the practical protocols and tools outlined here provide a starting point for leveraging these emerging paradigms to tackle previously intractable problems in catalysis, drug discovery, and materials design, pushing the boundaries beyond conventional post-Hartree-Fock methods.
Conclusion
Post-Hartree-Fock methods are indispensable for achieving chemical accuracy in computational drug discovery, particularly for modeling intricate electronic interactions that underlie binding affinity and reactivity. While challenges of computational cost and system size persist, ongoing innovations in algorithmic efficiency, machine learning integration, and fragment-based approaches are steadily enhancing their accessibility. The future points toward a hybrid computational environment where classical post-HF methods, machine learning potentials, and nascent quantum computing synergistically push the boundaries of molecular simulation. For biomedical research, this progression promises more reliable in silico predictions of drug efficacy and safety, ultimately accelerating the development of novel therapeutics and deepening our understanding of complex biological systems at an atomic level.
References
- 1. Post–Hartree–Fock [https://en.wikipedia.org/wiki/Post%E2%80%93Hartree%E2%80%93Fock]
- 2. 6.2 Electron correlation and post-Hartree-Fock approaches [https://fiveable.me/computational-chemistry/unit-6/electron-correlation-post-hartree-fock-approaches/study-guide/uTuhO6yfiyCbUeYZ]
- 3. Electron Correlation - an overview [https://www.sciencedirect.com/topics/chemistry/electron-correlation]
- 4. Electronic correlation [https://en.wikipedia.org/wiki/Electronic_correlation]
- 5. When does Hartree-Fock fundamentally fail? [https://chemistry.stackexchange.com/questions/114303/when-does-hartree-fock-fundamentally-fail]
- 6. Post Hartree-Fock Methods - an overview [https://www.sciencedirect.com/topics/chemistry/post-hartree-fock-methods]
- 7. 2.4 Post Hartree-Fock techniques [https://web.ornl.gov/~pk7/thesis/pkthnode14.html]
- 8. Quantum Mechanics in Drug Discovery - PubMed Central - NIH [https://pmc.ncbi.nlm.nih.gov/articles/PMC12249871/]
- 9. Quantum Chemistry in a Nutshell II – Post-Hartree-Fock methods [https://www.quantum-computing-info.com/post-hartree-fock/]
- 10. Quantum Chemistry in Drug Discovery - Rowan Newsletter [https://rowansci.substack.com/p/quantum-chemistry-in-drug-discovery]
- 11. Predicting the Post-Hartree-Fock Electron Correlation ... [https://www.mdpi.com/1420-3049/30/17/3500]
- 12. Accurate and convergent energetics of color centers ... [https://www.nature.com/articles/s41524-025-01813-0]
- 13. Effective approximations for Hartree–Fock exchange ... [https://arxiv.org/html/2509.00733v1]
- 14. GPU-Accelerated Fock Matrix Computation with Efficient ... [https://www.mdpi.com/2076-3417/15/9/4779]
- 15. FSIM: A Pedagogical and Extensible HPC Framework for ... [https://arxiv.org/html/2510.24968v1]
- 16. Advancing predictive modeling in computational chemistry ... [https://link.springer.com/article/10.1007/s44371-025-00363-0]
- 17. Revolutionizing pharmacology: AI-powered approaches in ... [https://www.sciencedirect.com/science/article/pii/S259009862500020X]
- 18. A computational chemistry approach to modelling ... [https://www.sciencedirect.com/science/article/pii/S2352484720302560]
- 19. Benchmarking Hartree-Fock and DFT for Molecular ... [https://arxiv.org/html/2511.17767v1]
- 20. Regularized Second Order Møller-Plesset Theory [https://pmc.ncbi.nlm.nih.gov/articles/PMC10037552/]
- 21. and Third-Order Møller-Plesset Perturbation Theory for Non ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC9948597/]
- 22. Understanding discrepancies in noncovalent interaction ... [https://www.nature.com/articles/s41467-025-64104-8]
- 23. And Third-Order Møller-Plesset Perturbation Theories for ... [https://impact.ornl.gov/en/publications/exploring-the-limits-of-second-and-third-order-m%C3%B8ller-plesset-per/]
- 24. Quantum Chemistry Simulations: Accelerating Drug ... [https://quantumzeitgeist.com/quantum-chemistry-simulations-accelerating-drug-discovery/]
- 25. - Non Research Papers - Academia.edu covalent interactions [https://www.academia.edu/Documents/in/Non-covalent_interactions]
- 26. Post-Hartree-Fock method in Quantum Chemistry for ... [https://arxiv.org/abs/2011.01544]
- 27. Accurate Reduced-Cost CCSD(T) Energies [https://pmc.ncbi.nlm.nih.gov/articles/PMC7884001/]
- 28. Basis Set Limit CCSD(T) Energies for Extended Molecules ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC9835832/]
- 29. Enabling accurate and large-scale explicitly correlated ... [https://chemrxiv.org/engage/chemrxiv/article-details/67a7527d6dde43c9080ebe5d]
- 30. Another Angle on Benchmarking Noncovalent Interactions [https://pmc.ncbi.nlm.nih.gov/articles/PMC11912214/]
- 31. How should I compare different basis sets for post-Hartree- ... [https://mattermodeling.stackexchange.com/questions/4034/how-should-i-compare-different-basis-sets-for-post-hartree-fock-methods]
- 32. Towards Post-Hartree-Fock Accuracy for Protein-Ligand ... [https://chemrxiv.org/engage/chemrxiv/article-details/656133b329a13c4d47ec222d]
- 33. Toward Post-Hartree-Fock Accuracy for Protein-Ligand ... [https://pubmed.ncbi.nlm.nih.gov/38530869/]
- 34. Fragment-Based Drug Design: From Then until Now, and ... [https://pubmed.ncbi.nlm.nih.gov/39992814/]
- 35. Fragment-based drug discovery: A unified workflow for ... [https://www.ddw-online.com/fragment-based-drug-discovery-a-unified-workflow-for-efficient-lead-generation-38290-202510/]
- 36. Computational approaches streamlining drug discovery [https://www.nature.com/articles/s41586-023-05905-z]
- 37. Fragment-Based Drug Discovery | April 15-16, 2025 [https://www.drugdiscoverychemistry.com/25/fragment-based-drug-discovery]
- 38. Rowan | ML-Powered Molecular Design and Simulation [https://www.rowansci.com/]
- 39. OpenEye Scientific: Molecular Modeling Software [https://www.eyesopen.com/]
- 40. t-SMILES: a fragment-based molecular representation ... [https://www.nature.com/articles/s41467-024-49388-6]
- 41. Basis set (chemistry) - Wikipedia [https://en.wikipedia.org/wiki/Basis_set_(chemistry)]
- 42. 8.1 Introduction to Basis Sets [https://manual.q-chem.com/5.3/Ch8.S1.html]
- 43. Basis Sets | Gaussian.com [https://gaussian.com/basissets/]
- 44. Generation of Basis Sets for Accurate Molecular Calculations [https://www.mdpi.com/2079-3197/10/5/65]
- 45. Basis set convergence of molecular properties: Geometry ... [https://education.molssi.org/qm-tools/04-vib-freq/index.html]
- 46. (PDF) Basis set selection for molecular calculations [https://www.academia.edu/23638053/Basis_set_selection_for_molecular_calculations]
- 47. What considerations must be made when selecting a basis ... [https://mattermodeling.stackexchange.com/questions/13987/what-considerations-must-be-made-when-selecting-a-basis-set]
- 48. How to choose the best method and basis set for a calculation in computational chemistry? - Chemistry Stack Exchange [https://chemistry.stackexchange.com/questions/42900/how-to-choose-the-best-method-and-basis-set-for-a-calculation-in-computational-c]
- 49. Predicting the Post-Hartree-Fock Electron Correlation ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12430394/]
- 50. Machine learning electronic structure methods based on ... [https://www.nature.com/articles/s41467-023-41953-9]
- 51. Reactive Chemistry at Unrestricted Coupled Cluster Level [https://arxiv.org/html/2509.10872v1]
- 52. Quantum information theory on sparse wavefunctions and ... [https://arxiv.org/abs/2408.02631]
- 53. How to use quantum computers for biomolecular free ... [https://arxiv.org/html/2506.20587v1]
- 54. Tensor numerical methods in quantum : from chemistry – Hartree ... Fock [https://pubs.rsc.org/en/content/articlehtml/2015/cp/c5cp01215e]
- 55. Low-scaling post Hartree-Fock [https://manual.cp2k.org/trunk/methods/post_hartree_fock/low-scaling.html]
- 56. Advancing Quantum Chemistry through HPC and ... [https://www.amd.com/en/blogs/2025/advancing-quantum-chemistry-through-hpc-and-veloxchem-.html]
- 57. FSIM: A Pedagogical and Extensible HPC Framework for ... [https://ui.adsabs.harvard.edu/abs/2025arXiv251024968H/abstract]
- 58. Insights from hybrid QM/MM MD simulations [https://www.sciencedirect.com/science/article/abs/pii/S002195172500586X]
- 59. Approximation Strategies to the Many-Body Schrödinger ... [https://chemrxiv.org/engage/chemrxiv/article-details/6920a703ef936fb4a2e8d563]
- 60. GraphGIM: rethinking molecular graph contrastive learning via... [https://bmcbiol.biomedcentral.com/articles/10.1186/s12915-025-02249-0]
- 61. Comparison, Analysis, and Molecular Dynamics ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10525864/]
- 62. PyMOL | pymol.org [https://www.pymol.org/]
- 63. Predicting the Post-Hartree-Fock Electron Correlation ... [https://pubmed.ncbi.nlm.nih.gov/40942028/]
- 64. Benchmarking crystal structure refinement: A systematic ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12422757/]
- 65. Correlated Ab Initio Calculations: Tutorial and Assignments [https://people.chem.ucsb.edu/kahn/kalju/chem126/public/qm_el_corr.html]
- 66. A self-conformation-aware pre-training framework for ... [https://www.nature.com/articles/s41467-025-59634-0]
- 67. Open Molecular Crystals 2025 (OMC25) Dataset and Models [https://ai.meta.com/research/publications/open-molecular-crystals-2025-omc25-dataset-and-models/]
- 68. Researchers optimize simulations of molecules on quantum ... [https://arstechnica.com/science/2025/01/researchers-optimize-simulations-of-molecules-on-quantum-computers/]
- 69. Our Quantum Echoes algorithm is a big step toward real-world ... [https://chemistry.berkeley.edu/news/our-quantum-echoes-algorithm-big-step-toward-real-world-applications-quantum-computing]
- 70. Study Suggests Today’s Quantum Could Aid Computers ... Molecular [https://thequantuminsider.com/2025/07/15/study-suggests-todays-quantum-computers-could-aid-molecular-simulation/]
- 71. Exploring the design space of machine-learning models for ... [https://arxiv.org/abs/2504.01187]
- 72. Advanced machine learning for innovative drug discovery [https://jcheminf.biomedcentral.com/articles/10.1186/s13321-025-01061-w]
- 73. Top 20 molecules for quantum computing [https://pennylane.ai/blog/2024/01/top-20-molecules-for-quantum-computing]