From Heitler-London to Modern Computations: The Evolution and Renaissance of Valence Bond Theory
This article provides a comprehensive analysis of the Valence Bond (VB) theory, tracing its evolution from the foundational Heitler-London quantum mechanical model of the hydrogen molecule to its modern, computationally...
From Heitler-London to Modern Computations: The Evolution and Renaissance of Valence Bond Theory
Abstract
This article provides a comprehensive analysis of the Valence Bond (VB) theory, tracing its evolution from the foundational Heitler-London quantum mechanical model of the hydrogen molecule to its modern, computationally competitive form. Tailored for researchers, scientists, and drug development professionals, it explores the core principles, methodological advancements, and solutions to historical limitations that have spurred a VB renaissance. The scope includes a direct comparison with Molecular Orbital (MO) theory, clarifying their mathematical equivalence at high levels of theory and distinct conceptual advantages. The discussion extends to the theory's implications for understanding electronic structure, chemical reactivity, and potential applications in biomolecular modeling and drug design.
The Birth of a Quantum Theory: Tracing the Heitler-London Foundation and Historical Context
Long before the development of quantum mechanics provided a rigorous mathematical framework for chemical bonding, Gilbert N. Lewis proposed a revolutionary theory that would forever change how chemists visualize the formation of molecules. His 1916 seminal paper, "The Atom and the Molecule," introduced two fundamental concepts: the cubical atom model and the electron-pair bond [1] [2]. These pre-quantum ideas not only explained chemical valence with remarkable success but also laid the essential conceptual groundwork for what would later become modern valence bond theory [2] [3]. Within the context of Heitler-London approach modern valence bond comparison research, Lewis's work represents the critical chemical intuition that preceded and guided the subsequent quantum mechanical formulation [3]. This article explores Lewis's original models, their limitations, and their direct intellectual lineage to contemporary understanding of chemical bonding, providing a comparative analysis of how these early concepts measure against modern computational approaches.
Lewis's Cubical Atom Model: Architecture and Bonding Mechanisms
Core Principles of the Cubic Atom
Developed initially in 1902 but published in 1916, Lewis's cubical atom model proposed a novel architecture where electrons positioned at the eight corners of a cube constituted the valence shell of an atom [1] [4]. This arrangement naturally led to the octet rule, as eight electrons would complete the cube's vertices [2] [4]. The model was specifically designed to explain the phenomenon of valency, providing a tangible, visual representation of how atoms combine to form molecules [1]. In this conceptualization, the cube itself represented the atom's outer shell, with electrons at vertices participating in bonding interactions through sharing of edges or faces between adjacent atomic cubes [1]. This spatial configuration offered an immediate explanation for why elements tended to form certain numbers of bonds, as the geometry limited how cubes could connect.
Bonding Representations in the Cubic Framework
The cubical atom model provided distinct mechanisms for different bond types through specific spatial interactions between cubes:
- Ionic bonds formed through complete transfer of an electron from one cube to another without sharing an edge (Structure A) [1] [4]
- Single covalent bonds occurred when two atoms shared a cube edge, resulting in the sharing of two electrons (Structure C) [1] [4]
- Intermediate polar bonds were represented by sharing only one corner between cubes (Structure B) [1] [2]
- Double bonds formed when two cubic atoms shared a face, resulting in four electrons being shared [1] [4]
Lewis himself described the dynamic nature of bonding, viewing these representations as "two limiting types" with individual molecules potentially ranging "all the way from one limit to the other" [2]. This perspective remarkably foreshadowed the quantum mechanical concept of resonance that would later be developed by Pauling [2].
Table 1: Bonding Mechanisms in Lewis's Cubical Atom Model
| Bond Type | Cubic Representation | Electrons Shared | Molecular Example |
|---|---|---|---|
| Ionic | No shared edge | Complete transfer | NaCl |
| Single Covalent | Shared edge | 2 | H₂ |
| Polar | Shared corner | Unequal sharing | HCl |
| Double Bond | Shared face | 4 | O₂ |
Limitations and Transition to Electron-Dot Structures
The cubical model encountered significant limitations, most notably its inability to satisfactorily explain triple bonds [1] [2]. There was no geometrically consistent way for two cubes to share three parallel edges [1] [4]. This fundamental shortcoming prompted Lewis to abandon the rigid cubic representation in favor of a more flexible approach [2]. He subsequently introduced the electron-dot structures (also called Lewis structures), where bonds were represented by pairs of dots between atomic symbols [2]. This transition from a three-dimensional cubic model to two-dimensional diagrammatic representations marked a crucial simplification that retained the essential concept of electron-pair bonding while overcoming the geometric constraints of the cube [2]. In his later representations, Lewis arranged the four electron pairs in a tetrahedral fashion around atoms, making a direct connection to the three-dimensional molecular geometries observed by organic chemists [2].
The Electron-Pair Bond: Lewis's Enduring Legacy
Fundamental Concept and Chemical Implications
At the heart of Lewis's revolutionary theory was the proposition that a chemical bond forms through the interaction of two shared bonding electrons, creating an electron pair that glues atoms together in most molecular matter [2]. This concept of the electron pair as the "quantum unit of chemical bonding" represented a dramatic shift from earlier mechanical models of bonding, such as the "hook and eye" model that Linus Pauling had learned as a student [5]. Lewis's theory successfully explained why the most abundant compounds possess an even number of electrons and provided a logical foundation for understanding molecular stability [2]. The electron-pair concept elegantly rationalized the observation that elements tend to form characteristic numbers of bonds, as atoms would share electrons until achieving a stable electron configuration, typically an octet for second-row elements [6].
Resonance and Dynamic Bonding Character
Beyond simply identifying the electron pair as the fundamental unit of bonding, Lewis recognized the dynamic character of chemical bonds [2]. He described what he called "tautomerism between polar and non-polar" forms, suggesting that molecules could exist as resonance hybrids between purely covalent and purely ionic extremes [2]. This prescient concept directly anticipated the resonance theory that Pauling would later develop quantum mechanically [2]. Lewis further used this dynamic electron-pair mechanism to explain heterolytic bond cleavage in solution, where an electron pair moves completely to one atom during bond breaking [2]. This idea would later be formalized with the curved arrow notation invented by Robinson to describe reaction mechanisms [2].
Theoretical Evolution: From Lewis to Modern Valence Bond Theory
The Heitler-London Quantum Mechanical Foundation
The crucial transition from Lewis's qualitative but chemically intuitive model to a quantitative quantum theory began with the work of Walter Heitler and Fritz London in 1927 [2] [3]. They demonstrated that Lewis's electron-pair bond could be explained using Schrödinger's wave mechanics, specifically for the hydrogen molecule [7] [3]. The Heitler-London treatment described the electronic wavefunction as a covalent combination of localized basis functions on the bonding atoms [8]. For H₂, they expressed the wavefunction as:
ΦHL = |ab̅| - |a̅b|
where a and b are basis functions (1s atomic orbitals) localized on the two hydrogen atoms, and the bar indicates beta spin [8]. This wavefunction describes the covalent pairing of electron spins between the two atoms as envisioned by Lewis [8]. The resonance character of this wavefunction allowed electrons in fixed fragment orbitals to delocalize, lowering the kinetic energy in the case of H₂ [9]. This treatment provided the first quantum mechanical validation of Lewis's electron-pair bond, establishing that bond stability arose from the quantum mechanical phenomenon of resonance [3].
Pauling's Resonance Theory and Hybridization
Linus Pauling, who had been excited upon learning of the Heitler-London treatment, undertook an extensive program to generalize and popularize valence bond theory [2]. His work translated Lewis's chemical ideas into quantum mechanical language, making them accessible and useful to practicing chemists [2] [3]. Pauling introduced two critical extensions to the basic theory: resonance (1928) and orbital hybridization (1930) [7]. Resonance theory provided a quantum mechanical framework for Lewis's concept of dynamic bonding, allowing representation of molecules as quantum superpositions of multiple valence bond structures [7]. Hybridization (sp, sp², sp³) explained how atoms could achieve the appropriate geometry for bonding, such as the tetrahedral configuration of carbon in methane [7]. Pauling's 1939 textbook "On the Nature of the Chemical Bond" became the definitive work that dominated chemical thinking for decades [7].
Table 2: Evolution of Key Concepts from Lewis to Modern Valence Bond Theory
| Concept | Lewis's Pre-Quantum Formulation | Modern Quantum Refinement | Key Contributors |
|---|---|---|---|
| Chemical Bond | Electron pair shared between atoms | Quantum resonance between covalent/ionic structures | Lewis, Heitler, London, Pauling |
| Atomic Geometry | Cubical atom with electrons at vertices | Orbital hybridization (sp³, sp², sp) | Pauling, Slater |
| Resonance | Tautomerism between polar/non-polar forms | Quantum superposition of VB structures | Lewis, Pauling |
| Bond Types | Edge, corner, or face sharing between cubes | Sigma/pi bonds from orbital overlap | Lewis, Pauling |
Comparative Analysis: Cubical Model vs. Modern Computational Approaches
Conceptual Strengths and Limitations
When evaluated against modern computational approaches, Lewis's cubical model demonstrates both remarkable insight and significant limitations:
- Visualization Advantage: The cubical model provided an intuitive, tangible representation of valence that could be easily visualized and taught, unlike abstract mathematical formulations [5]
- Explanatory Power for Organic Molecules: The model successfully explained the tetrahedral geometry of carbon and the fundamental difference between single, double, and triple bonds [2]
- Quantitative Deficiencies: Unlike modern valence bond theory, the cubical model offered no mathematical framework for calculating bond energies or molecular properties [1]
- Limited Applicability: The model struggled with triple bonds and could not adequately explain transition metal bonding or spectroscopic properties [1] [4]
- Lack of Predictive Power: While excellent for rationalizing known compounds, the model had limited utility for predicting new types of chemical bonds [1]
Modern valence bond theory, implemented with computer programs competitive in accuracy with molecular orbital methods, has overcome these limitations while retaining the chemical intuition of Lewis's original approach [8].
Modern Computational Valence Bond Methods
Contemporary valence bond theory has seen a renaissance with the development of computational methods that preserve the locality of the electron-pair bond while achieving accuracy comparable to molecular orbital approaches [8]. Modern VB methods describe the electronic wavefunction as a linear combination of several valence bond structures, each representing a specific Lewis structure [8]. Unlike simple molecular orbital theory, which can overemphasize ionic character, VB theory correctly describes bond dissociation and provides a more intuitive picture of electron reorganization during chemical reactions [7] [8]. The relationship between MO and VB theories is formally established through a unitary transformation - at the same level of theory, they represent different but mathematically equivalent representations of the same wavefunction [8].
Table 3: Methodological Comparison of Bonding Theories
| Feature | Lewis Cubical Model | Modern Valence Bond Theory | Molecular Orbital Theory |
|---|---|---|---|
| Bond Representation | Electron pairs as shared cube elements | Linear combination of VB structures | Delocalized molecular orbitals |
| Mathematical Basis | Qualitative geometric model | Quantum wavefunction with electron correlation | Quantum wavefunction with orbital delocalization |
| Treatment of Aromaticity | Not addressed | Spin coupling of π orbitals | π-electron delocalization |
| Bond Dissociation | Not quantitatively addressed | Correctly dissociates to neutral atoms | Can incorrectly predict ionic dissociation |
| Computational Cost | Minimal | High (until recent developments) | Moderate |
Experimental and Computational Protocols in Modern Bonding Analysis
Energy Decomposition Analysis (EDA) Methodology
Modern analysis of chemical bonding origins utilizes sophisticated computational protocols such as the variational Energy Decomposition Analysis (EDA) based on Absolutely Localized Molecular Orbitals (ALMOs) [9]. This approach partitions the total interaction energy (ΔEINT) into physically meaningful components along the bond-forming pathway:
ΔEINT = ΔEPrep + ΔECov + ΔECon + ΔEPCT
where:
- ΔEPrep: Energy change for fragment geometric distortion and hybridization [9]
- ΔECov: Energy lowering from constructive quantum interference between fragment wavefunctions [9]
- ΔECon: Stabilization from orbital contraction [9]
- ΔEPCT: Energy lowering from polarization and charge transfer [9]
This methodology has revealed that unlike H₂ and H₂⁺, where kinetic energy lowering drives bond formation, bonds between heavier elements (e.g., H₃C-CH₃, F-F) often show kinetic energy increases during bond formation due to Pauli repulsion between bonding and core electrons [9].
Research Reagent Solutions: Computational Tools for Bonding Analysis
Table 4: Essential Computational Tools for Modern Valence Bond Research
| Research Tool | Function | Application in Bonding Analysis |
|---|---|---|
| VB-CI Methods | Configuration Interaction in Valence Bond basis | Accounts for electron correlation in bond breaking |
| ALMO-EDA | Energy Decomposition Analysis | Partitioning covalent, ionic, and steric contributions |
| CAS-VB | Complete Active Space Valence Bond | Multiconfigurational treatment of complex bonds |
| Fragment Orbital Methods | Use of molecular orbitals as basis functions | Analysis of bonding in large systems |
| Modern VB Codes | (e.g., XMVB, TURTLE) | Practical computation of VB wavefunctions |
Gilbert N. Lewis's pre-quantum models of the cubical atom and electron-pair bond represent far more than historical curiosities. They embody the essential chemical intuition about localized bonds between atoms that continues to influence how chemists design and understand molecules [5] [3]. While the specific geometric representation of the cubical atom was quickly superseded by quantum mechanical models, the core concept of the electron-pair bond has proven astonishingly durable [2] [3]. Modern valence bond theory, with its sophisticated computational implementations, remains fundamentally rooted in Lewis's original insight that chemical bonds form through electron pairs shared between atoms [8]. The trajectory from Lewis's cubes to modern computational VB methods demonstrates how chemical intuition can productively guide theoretical development, creating a synthesis of physical rigor and chemical insight that continues to advance our understanding of molecular structure [3].
The 1927 work by Walter Heitler and Fritz London on the hydrogen molecule (H₂) marks the foundational moment of quantum chemistry. For the first time, the "chemical bond," a concept long used by chemists without a fundamental physical explanation, was described in the rigorous language of quantum mechanics [10]. Prior to this breakthrough, the existence of stable molecules posed a significant challenge to classical physics, which could not explain why two neutral hydrogen atoms would form a bound state rather than repel each other. Heitler and London, working as postdocs with Erwin Schrödinger, discovered that the chemical bond originates in a quantum interference phenomenon between electron wave functions, a phenomenon they termed "Schwebungsphänomen" in their original German publication [10]. Their calculation demonstrated that the covalent bond arises from electron sharing between atoms, with the binding energy resulting from the cumulative effects of electron density buildup between nuclei and quantum mechanical exchange [11].
This pioneering work established the valence bond (VB) theory, which assumes strictly "correlated" electrons without ionic terms, in contrast to the molecular orbital approach that would emerge later [10]. The Heitler-London treatment represented a radical departure from classical thinking, providing the first quantum-mechanical account of why the H₂ molecule exists at all—a phenomenon that classical mechanics coupled with electrostatics had failed to explain [11]. Their work laid the groundwork for Linus Pauling's more extensive development of valence bond theory and his concepts of orbital hybridization and resonance, which would dominate chemical thinking for decades [10] [11].
Methodological Approaches: Valence Bond vs. Molecular Orbital Theory
The Heitler-London Valence Bond Method
The Heitler-London approach to the hydrogen molecule started with a simple but profound insight: when two hydrogen atoms are brought close together, their electron wave functions begin to overlap, leading to quantum mechanical interference that can either lower or raise the total energy of the system depending on the relative spin alignment of the electrons [11].
The mathematical foundation begins with the electronic Hamiltonian for H₂, which describes the kinetic and potential energies of all particles in the system [12]:
[ \hat{H} = -\frac{1}{2} \nabla^21 -\frac{1}{2} \nabla^22 -\frac{1}{r{1A}} -\frac{1}{r{2B}} -\frac{1}{r{2A}} -\frac{1}{r{1B}} +\frac{1}{r_{12}} +\frac{1}{R} ]
Heitler and London approximated the molecular wavefunction using atomic orbitals from the separated atoms:
[ \psi(r1,r2) = \psi{1s}(r{1A})\psi{1s}(r{2B}) ]
This wavefunction was then used in the variational integral to compute an approximate energy:
[ \tilde{E}(R) = \frac{\int{\psi \hat{H} \psi d\tau}}{\int{\psi^2 d\tau}} ]
Their key finding was that the singlet state (with opposed electron spins) produced a significant binding energy at a specific internuclear distance, while the triplet state (with parallel spins) led only to repulsion [11]. This spin-dependent interaction represented the first quantum mechanical explanation of why some atomic configurations form bonds while others do not.
The Molecular Orbital Alternative
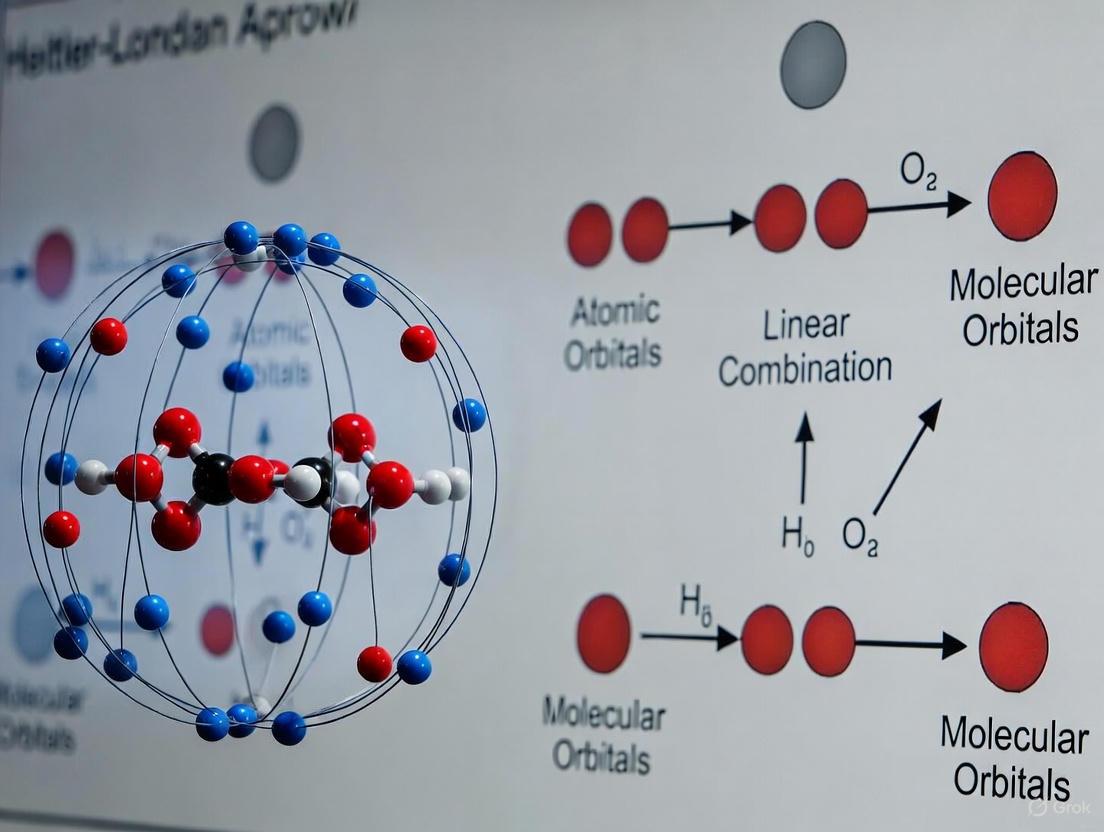
Shortly after Heitler and London's work, Friedrich Hund and Robert S. Mulliken developed an alternative approach: molecular orbital (MO) theory [10] [11]. Unlike the valence bond method, which emphasizes electron correlation and pairing, MO theory constructs orbitals that extend over the entire molecule by taking linear combinations of atomic orbitals (LCAO) [13].
For H₂⁺, the simplest molecular system, the molecular orbitals are formed as:
[ \psi+ = C+(1sA + 1sB) ] [ \psi{-} = C{-}(1sA - 1sB) ]
The bonding molecular orbital (ψ₊) has enhanced electron density between the nuclei, creating the attractive force that constitutes the chemical bond. In contrast, the antibonding orbital (ψ₋) has a nodal plane between the nuclei with diminished electron density in the bonding region, resulting in repulsion [13]. The MO method initially gave poorer results for H₂ but eventually dominated chemical thinking due to its systematic improvability and conceptual advantages for describing delocalized bonding [10] [11].
Figure 1: Quantum mechanical approaches to chemical bonding in H₂, showing how atomic orbitals combine to form molecular bonds through two different theoretical frameworks.
Quantitative Comparison of Methodological Performance
Computational Accuracy for H₂
The table below compares the performance of different quantum mechanical methods in describing the hydrogen molecule, based on historical development and theoretical refinements:
Table 1: Performance comparison of quantum chemical methods for H₂ description
| Method | Dissociation Energy (eV) | Equilibrium Bond Length (Å) | Key Features | Limitations |
|---|---|---|---|---|
| Heitler-London (1927) [11] | 3.14 | 0.87 | First quantum explanation of bonding; Introduced electron correlation | Underestimates binding energy; Oversimplified wavefunction |
| Simple MO Theory [11] | ~2.7 | ~0.85 | Conceptual simplicity; Systematic improvability | Poor initial results; Allows ionic terms with incorrect weights |
| James-Coolidge (1933) [11] | ~4.48 (matched experiment) | ~0.74 (matched experiment) | High accuracy with mechanical calculators; Included correlation effects | Computationally demanding; Complex mathematical form |
| Modern Calculations [14] | 4.746 (current accepted value) | 0.7406 (current accepted value) | Includes relativistic and QED effects; Accuracy to 9+ significant digits | Requires extensive computational resources |
The Heitler-London method, while groundbreaking, significantly underestimated the true binding energy of H₂ (3.14 eV calculated vs. 4.48 eV experimental) and overestimated the bond length (0.87 Å calculated vs. 0.74 Å experimental) [11]. However, it correctly predicted the existence of the chemical bond and provided a qualitative understanding of its origin. The systematic errors stemmed primarily from the limited flexibility of the wavefunction and the lack of ionic terms (H⁺H⁻ and H⁻H⁺) which become important at shorter distances [10].
Evolution of Theoretical Accuracy
The progression toward higher accuracy required increasingly sophisticated treatments:
- Configuration Interaction: Adding excited-state configurations to the wavefunction [15]
- Orbital Optimization: Self-consistent field methods to improve orbital shapes [15]
- Explicit Correlation: Direct inclusion of electron-electron distance in wavefunction [11]
- Relativistic Corrections: Accounting for effects described by quantum electrodynamics [14]
Modern quantum calculations of hydrogen molecule energy levels now achieve astonishing precision, with uncertainties at the level of 10⁻⁹ or better, strongly dependent on high-order relativistic and quantum electrodynamics (QED) corrections [14].
Experimental Protocols and Validation
Spectroscopic Validation Methods
The theoretical predictions of the Heitler-London model and its refinements required experimental validation through precision spectroscopy. Key methodological approaches include:
Cavity Ring-Down Spectroscopy: Ultra-sensitive technique used to achieve precision measurements of near-infrared electric quadrupole transitions in H₂ molecules with uncertainties at the state-of-the-art level [14]
Frequency Comb-Calibrated Saturation Absorption Spectroscopy: Combined with optical frequency combs to observe Doppler-free Lamb-dip spectra of overtone transitions in HD molecules, with frequency measurement accuracy reaching 10 significant digits [14]
Vacuum Ultraviolet Laser Systems: "Chirp"-free systems that improved the measurement accuracy of H₂ dissociation energy to the sub-MHz level, providing key experimental evidence for determining fundamental constants like the proton charge radius [14]
These experimental methods verify quantum theoretical calculations with up to 9-10 significant digits, providing stringent tests for quantum electrodynamics theory and enabling precise determination of fundamental constants such as the proton-to-electron mass ratio [14].
Potential Energy Curve Mapping
The potential energy curve of H₂ represents a fundamental experimental benchmark for theoretical methods [11]:
Figure 2: Methodology for mapping the H₂ potential energy curve, showing the relationship between atomic approach, energy minimization, and experimental validation.
The depth of the potential well at the equilibrium distance Rₑ represents the binding energy or dissociation energy Dₑ. For H₂, the accepted experimental values are Dₑ = 4.746 eV and Rₑ = 0.7406 Å [12] [14]. The potential energy curve follows the relationship E(R) = Eₑₗₑ꜀(R) + 1/R, where Eₑₗₑ꜀(R) is the electronic energy from solving the electronic Schrödinger equation, and 1/R represents the nuclear repulsion term [12].
Table 2: Key computational and experimental resources for hydrogen molecule research
| Tool/Resource | Function/Application | Relevance to H₂ Studies |
|---|---|---|
| LOBSTER Package [10] | Quantum-chemistry software for wave function-based bonding analysis | Calculates atomic charges, population analyses, and periodic bonding indicators for solid-state systems |
| Plane-Wave DFT Codes [10] | Electronic structure calculations for periodic systems | Superior efficiency for periodic electronic structures; requires unitary transformation to atomic orbitals for bonding analysis |
| Maximally Localized Wannier Functions (MLWF) [10] | Localized orbital construction in solids | Generates orbitals reflecting Lewis-type single, double, or triple bonds; extends localization ideas to solid state |
| Cavity Ring-Down Spectroscopy [14] | Ultra-sensitive absorption measurements | Precision measurements of H₂ transition frequencies with uncertainties at state-of-the-art levels |
| Frequency Comb Calibration [14] | Absolute frequency measurement | Enables Doppler-free spectroscopy with accuracy to 10 significant digits |
| Variational Method [12] | Quantum mechanical approximation technique | Foundation for improving wavefunction accuracy through systematic expansion |
Modern Applications and Research Frontiers
Fundamental Physical Tests
The hydrogen molecule continues to serve as a critical testing ground for fundamental physics:
Quantum Electrodynamics Tests: Precision spectroscopic measurements of H₂ provide stringent experimental tests for QED theory, with recent theoretical studies showing strong dependence on high-order relativistic and QED corrections [14]
Determination of Fundamental Constants: H₂ spectroscopy enables accurate determination of constants including the proton charge radius and proton-to-electron mass ratio, with potential implications for resolving the "proton radius puzzle" [14]
Beyond Standard Model Physics: Planned measurements aiming for accuracy beyond 10⁻¹⁰ may enable probing of "new physics" beyond the Standard Model of particle physics [14]
Bonding Analysis in Solids
The conceptual framework established by Heitler-London has extended far beyond the original H₂ molecule:
Solid-State Bonding Analysis: Orbital-based bonding analysis using tools like LOBSTER allows application of wavefunction-based bonding concepts to periodic solids, moving beyond the oversimplified ionic model that previously dominated solid-state chemistry [10]
Crystal Orbital Overlap Population (COOP): Generalized Mulliken overlap population for periodic wave functions, revolutionizing how solid-state chemists understand bonding in materials [10]
First-Principles Bond Orders: Quantitative bond indices derived from density matrix analysis, extending the qualitative concepts of valence bond theory to quantitative predictions in complex materials [10]
The 1927 Heitler-London treatment of H₂ represents far more than a historical milestone—it established the fundamental paradigm for understanding chemical bonding in quantum mechanical terms. While the simple valence bond approach has been superseded by more accurate methods for quantitative predictions, its conceptual framework remains vital for chemical intuition and reasoning.
The continuing evolution of H₂ research—from the qualitative breakthrough of Heitler-London to modern measurements with 10-digit accuracy—exemplifies the progressive refinement of scientific understanding. The hydrogen molecule continues to serve as a benchmark system for testing theoretical methods and experimental techniques, bridging the gap between fundamental quantum mechanics and practical chemical applications. As precision spectroscopy advances further, this simplest of neutral molecules may yet reveal new physics beyond our current understanding, continuing the legacy that began with Heitler and London's pioneering work nearly a century ago.
The Heitler-London approach, originating from the first quantum-mechanical treatment of the hydrogen molecule in 1927, provides the foundational framework for understanding covalent bonding [10]. This seminal work demonstrated that the chemical bond arises fundamentally from interfering wave functions, a phenomenon described as "Schwebungsphänomen" in the original German publication [10]. The approach established valence bond (VB) theory, which emphasizes electron correlation and pairing between specific atomic centers. Modern quantum chemistry has since developed this foundation into sophisticated computational methods that extend far beyond the original two-electron model. Within this evolutionary context, three interrelated concepts—covalent wavefunctions, exchange interaction, and resonance—emerge as pillars for describing molecular structure and stability from the perspective of electron delocalization. This guide objectively compares these conceptual frameworks, examining their theoretical underpinnings, quantitative expressions, and roles in contemporary valence bond research, particularly relevant for molecular design in pharmaceutical development.
Conceptual Frameworks & Comparative Analysis
The following table provides a systematic comparison of the three core concepts, highlighting their distinct roles and manifestations in quantum chemistry.
Table 1: Comparative Analysis of Key Quantum Chemical Concepts
| Feature | Covalent Wavefunctions | Exchange Interaction | Resonance | ||||
|---|---|---|---|---|---|---|---|
| Core Definition | Mathematical descriptions (ψ) of the quantum state of a system; antisymmetric for fermions [16] [17]. | A quantum constraint on indistinguishable particles, arising from symmetry and Coulomb interaction [18] [19]. | A description of bonding in molecules by combining several contributing structures into a hybrid [20] [21]. | ||||
| Primary Physical Origin | Solution to the Schrödinger equation; incorporates wave-particle duality [16]. | Combination of exchange symmetry (Pauli principle) and Coulomb repulsion [18]. | Electron delocalization that lowers the system's potential energy [21]. | ||||
| Key Mathematical Formulation | Normalized: ( \int | \Psi | ^2 d\tau = 1 ); Born rule: ( | \Psi | ^2 ) is probability density [16] [17]. | Exchange integral: ( J{\text{ex}} = \int \Phia^(\vec{r}_1)\Phi_b^(\vec{r}2) \left( \frac{1}{r{12}} \right) \Phib(\vec{r}1)\Phia(\vec{r}2) d^3r1 d^3r2 ) [18]. | Represented by a superposition of Lewis structures; the hybrid is the true structure [20] [21]. |
| Interpretation in Bonding | Describes electron sharing and interference; the foundation for molecular orbital construction [10]. | An effective "interaction" that aligns spins (ferromagnetism) and influences bond energetics [18] [19]. | Not a rapid isomerism; a single, averaged structure with intermediate bond orders/ lengths [21]. | ||||
| Role in Modern VB Theory | The fundamental object from which all properties are derived; transformed for analysis (e.g., localized orbitals) [10]. | Manifests as a stabilization energy in the antisymmetric spin-orbitals of the Heitler-London model [18] [19]. | Quantified via resonance energy; provides the conceptual link to charge-shift bonding [21] [22]. |
Quantitative Data & Experimental Protocols
Energetic and Geometric Manifestations
Quantitative data from both computation and experiment validate the predictions of these theories. The resonance hybrid, for instance, manifests in experimentally measurable bond lengths and orders, while exchange interactions produce definitive, measurable energy splittings.
Table 2: Experimental and Computational Signatures of Resonance and Exchange
| System/Measurement | Observable | Theoretical Prediction | Experimental/Computational Result |
|---|---|---|---|
| Nitrite Ion (NO₂⁻) | N–O Bond Length [20] [21] | Intermediate between single (∼145 pm) and double (∼115 pm) bond. | 125 pm (average of two equivalent bonds). |
| Benzene (C₆H₆) | C–C Bond Order [21] | Bond order of 1.5 from two equivalent Kekulé structures. | 1.5 (from measured bond length of ∼139 pm). |
| H₂ Molecule (Biradical) | Singlet-Triplet Energy Gap [23] | Exchange integral, ( J ), determines the gap: ( ES - ET \approx J ). | For ( J > 0 ), the singlet state (antiferromagnetic) is lower in energy. |
| Probability Density Analysis (PDA) | Charge-Shift Resonance Energy [22] | Correlation between PDA barrier ( \Delta \Phi ) and VB-based ( E_{CS} ). | PDA provides a real-space measure for charge-shift bonding independent of VB reference. |
Detailed Computational Methodologies
Protocol 1: Calculating Exchange Coupling in a Biradical
- System Preparation: Construct a molecular model with two unpaired electrons occupying spatially separated, nearly degenerate molecular orbitals (SOMOs).
- Wavefunction Calculation: Perform an unrestricted DFT or multi-configurational (e.g., CASSCF) calculation to obtain the electronic wavefunctions.
- Energy Evaluation: Calculate the total energies for the broken-symmetry singlet state and the triplet state.
- Exchange Integral Extraction: The exchange coupling constant ( J ) can be estimated using the Yamaguchi formula or similar approaches: ( J \approx (E{S} - E{T}) / (\langle S^2 \rangleT - \langle S^2 \rangleS) ), where ( E{S} ) and ( E{T} ) are the singlet and triplet state energies, and ( \langle S^2 \rangle ) are their respective spin expectation values [23].
Protocol 2: Quantifying Resonance with Valence Bond Theory
- Structure Definition: Identify all major and minor resonance contributors based on octet rule, formal charge minimization, and electronegativity rules [21].
- Wavefunction Calculation: Perform a Valence Bond Self-Consistent Field (VBSCF) calculation, using the contributing structures as a non-orthogonal basis.
- Energy Decomposition: Compute the energy for the full wavefunction and for the single most stable contributing structure.
- Resonance Energy Calculation: The resonance energy (or delocalization energy) is defined as the difference: ( E{\text{resonance}} = E{\text{single structure}} - E_{\text{full VB wavefunction}} ). A positive value indicates stabilization due to resonance [21] [22].
Visualization of Theoretical Relationships and Workflows
The following diagrams, generated with Graphviz DOT language, illustrate the logical relationships between these concepts and a generalized workflow for bonding analysis.
Conceptual Relationship Map
Diagram 1: A map showing how fundamental quantum principles give rise to the key concepts of exchange interaction and resonance.
Bonding Analysis Workflow
Diagram 2: A computational workflow for bonding analysis, showing parallel paths in Molecular Orbital (MO) and Valence Bond (VB) theory.
The Scientist's Toolkit: Essential Research Reagents & Materials
This section details key computational methods and theoretical tools used in modern valence bond research, which serve as the essential "reagents" for in-silico experimentation.
Table 3: Essential Computational Tools for Modern Valence Bond Research
| Tool/Resource | Type | Primary Function in Research | ||
|---|---|---|---|---|
| LOBSTER Package [10] | Software | Performs orbital-based bonding analysis for periodic solids, enabling COOP, bond orders, and crystal orbital Hamilton population from plane-wave DFT. | ||
| Maximally Localized Wannier Functions (MLWFs) [10] | Computational Method | Generates localized orbitals from delocalized Bloch states, bridging MO theory and chemical intuition for solids. | ||
| Probability Density Analysis (PDA) [22] | Analytical Framework | Identifies critical points in the many-electron probability density | Ψ | ² to extract VB-like resonance structures and charge-shift character from any wavefunction. |
| Valence Bond Self-Consistent Field (VBSCF) | Computational Method | Optimizes a wavefunction expressed as a linear combination of VB structures, providing accurate weights and resonance energies. | ||
| Charge-Shift Resonance Energy (E_CS) [22] | Quantitative Descriptor | A measure within VB theory to identify and characterize bonds where electron-pair fluctuation (resonance) is dominant over static covalent or ionic sharing. |
The quarter-century following the initial applications of quantum theory to chemical bonding witnessed the powerful influence of Linus Pauling's valence bond (VB) formulation, which provided an intuitive framework that would dominate chemical thinking for decades [24]. During the period known as "The Pauling Era," concepts such as hybridization of atomic orbitals and resonance theory emerged as transformative tools that allowed chemists to reconcile quantum mechanics with empirically observed molecular structures and properties [25]. Pauling's work, particularly his landmark 1931 paper "The Nature of the Chemical Bond" and his influential 1939 textbook, translated the complex mathematics of quantum mechanics into a chemical framework that researchers could readily apply to diverse problems—from molecular geometry to drug design [25] [7].
This guide examines Pauling's conceptual framework within the broader context of Heitler-London approach modern valence bond comparison research, objectively evaluating how hybridization and resonance theories performed against emerging alternatives and detailing the experimental protocols that validated these concepts.
Theoretical Foundations: Extending the Heitler-London Approach
The Heitler-London Foundation
The quantum mechanical description of chemical bonding began with Walter Heitler and Fritz London's 1927 paper on the hydrogen molecule, which Pauling himself called "the greatest single contribution to the clarification of the chemist's concept of valence" [25]. Their work provided the first quantum mechanical justification for Gilbert N. Lewis's electron-pair bond through mathematical treatment of the covalent bond in H₂ as a quantum phenomenon involving the resonance phenomenon—an interchange in position of the two electrons that reduces the system's energy and causes bond formation [25] [2].
The Heitler-London theory described the hydrogen molecule using a wavefunction that emphasized electron pairing and localization, establishing the foundation upon which Pauling would build his more comprehensive valence bond theory [8]. This approach stood in contrast to the emerging molecular orbital (MO) theory, which would later be developed by Hund, Mulliken, and others [2].
Pauling's Conceptual Advances
Pauling's revolutionary contribution was extending the Heitler-London approach beyond simple diatomic molecules to account for bonding in polyatomic molecules through two key concepts:
Orbital Hybridization: Pauling proposed that atoms could "hybridize" their valence orbitals—mixing directionless free-atom s, p, d orbitals to form equivalent directed orbitals—to explain the directional character of bonds in molecules like methane (CH₄) with its tetrahedral geometry [24] [25]. This concept grew from Pauling's chemical intuition that the energy separation between s and p orbitals was small compared to bond formation energy, making such mixing favorable despite physicists' initial reservations about combining different orbital types [25].
Resonance Theory: Building on Heisenberg's quantum mechanical concept of resonance, Pauling proposed that the electronic structure of many molecules could be described as a "resonance hybrid" intermediate between two or more theoretical Lewis structures [21]. This approach successfully explained the properties of molecules like benzene, whose characteristics appeared averaged between possible bonding patterns [24].
Table 1: Fundamental Concepts in Pauling's Valence Bond Framework
| Concept | Theoretical Basis | Key Innovation | Example Applications |
|---|---|---|---|
| Orbital Hybridization | Mixing of atomic orbitals from the same atom | Explained directional bonds and equivalent bonding in polyatomic molecules | Tetrahedral carbon (sp³), trigonal planar boron (sp²) |
| Resonance Theory | Quantum mechanical state superposition | Described molecular properties as intermediate between multiple Lewis structures | Benzene, carboxylate ions, amide groups |
| Electronegativity | Empirical scale of electron-attracting power | Predicted bond polarity and ionic character | Acid strength, dipole moments, bond energies |
Quantitative Comparison: Valence Bond Theory vs. Molecular Orbital Theory
The struggle for dominance between valence bond theory and molecular orbital theory represents one of the significant narratives in the development of modern chemical bonding theory [2]. Each approach offered distinct advantages and limitations for researchers studying molecular structure and properties.
Table 2: Quantitative Comparison of Valence Bond vs. Molecular Orbital Theories
| Parameter | Valence Bond Theory (Pauling) | Molecular Orbital Theory (Hund-Mulliken) |
|---|---|---|
| Theoretical Foundation | Localized electron-pair bonds, hybridization, resonance | Delocalized molecular orbitals covering entire molecules |
| Computational Efficiency (1930s-1950s) | More accessible for hand calculation and qualitative prediction | Initially less accessible; required more computational resources |
| Bond Description | Localized between atom pairs with specific directional character | Delocalized over entire molecule |
| Prediction of Molecular Geometry | Excellent through hybridization concept | Good through molecular symmetry considerations |
| Treatment of Aromaticity | Resonance between Kekulé and Dewar structures | π-electron delocalization in cyclic systems |
| Handling of Molecular Dissociation | Correctly predicts homonuclear dissociation to atoms | Crude MO approaches incorrectly predict dissociation to mixture of atoms and ions |
| Explanation of Spectroscopic Properties | Limited ability to explain electronic transitions | Superior framework for spectra and ionization properties |
| Treatment of Paramagnetism | Struggles with paramagnetic species | Naturally accounts for paramagnetism via unpaired electrons in MOs |
The decline of VB theory's popularity beginning in the 1960s can be largely attributed to the computational advantages of MO theory, which achieved more efficient numerical implementation on digital computers and became the dominant approach in quantum chemistry programs [24] [7]. However, modern valence bond theory has experienced a resurgence since the 1980s as computational challenges have been addressed [8] [7].
Experimental Protocols and Validation
Validating Hybridization Through Molecular Geometry
Pauling's concept of hybridization provided quantitative predictions about molecular geometry that could be tested experimentally:
Protocol: Determining Tetrahedral Hybridization in Methane
- Theoretical Prediction: Carbon (ground state: 2s²2p²) undergoes promotion to 2s¹2p³ configuration followed by sp³ hybridization, forming four equivalent orbitals directed at tetrahedral angles (109.5°) [26]
- Experimental Methods: X-ray diffraction and spectroscopy to measure bond angles and lengths
- Key Measurements: All C-H bonds in methane are equivalent length (110.2 pm) with H-C-H bond angles of 109.5°, consistent with sp³ hybridization
- Comparative Controls: Compare with BH₃ (trigonal planar, 120°) and BeH₂ (linear, 180°) showing different hybridization patterns
The robustness of hybridization concepts has been confirmed using modern computational methods, including natural bond orbital analysis, which demonstrates that Pauling's qualitative conceptions of directional hybridization are consistently manifested across diverse computational quantum chemistry methodologies [24].
Quantifying Resonance Stabilization
Resonance theory predicted stabilization energies that could be measured experimentally:
Protocol: Measuring Resonance Energy in Benzene
- Theoretical Framework: Represent benzene as resonance hybrid between two Kekulé structures with alternating single and double bonds [21]
- Experimental Approach: Compare hydrogenation energy of benzene to hypothetical cyclohexatriene without resonance stabilization
- Calculation Method: Measure enthalpy of hydrogenation for benzene and compare to cyclohexene as reference
- Results: Benzene shows 150 kJ/mol greater stability than predicted for non-resonating structure, confirming significant resonance stabilization
The development of Natural Resonance Theory (NRT) has provided quantitative tools to evaluate resonance weights in modern computational chemistry, validating Pauling's original intuitions about the significance of resonance in molecular stabilization [24].
Visualization of Theoretical Relationships
The following diagram illustrates the conceptual development from Heitler-London theory to Pauling's framework and its relationship to molecular orbital theory:
Theoretical Evolution of Bonding Concepts
Table 3: Key Research Reagent Solutions for Valence Bond Studies
| Research Tool | Function/Application | Significance in Bonding Research |
|---|---|---|
| Natural Bond Orbital (NBO) Analysis | Analyze wavefunctions for hybridization and bond characterization | Quantifies % s/p character in hybrids and resonance weights [24] |
| X-Ray Crystallography | Determine molecular geometry and bond parameters | Experimental validation of bond lengths and angles predicted by hybridization |
| Photoelectron Spectroscopy | Measure electron binding energies | Probes orbital energies and hybridization effects |
| Vibrational Spectroscopy | Characterize bond strengths and force constants | Provides experimental bond orders through frequency measurements |
| Magnetic Susceptibility Measurements | Detect unpaired electrons | Distinguishes between covalent and ionic bonding character [25] |
| Energy Decomposition Analysis (EDA) | Partition binding energy into components | Quantifies covalent, ionic, and steric contributions to bonds |
| Quantum Chemistry Software (Gaussian, Molpro) | Compute wavefunctions and electronic structure | Enables NBO/NRT analysis at various theory levels [24] |
Pauling's conceptual framework of hybridization and resonance, developed during what we now call The Pauling Era, provided an indispensable bridge between quantum mechanics and practical chemical research that continues to influence how researchers approach molecular design in fields including drug development [25]. While molecular orbital theory eventually gained dominance for computational quantum chemistry, Pauling's valence bond approach offered an intuitive and chemically meaningful framework that could be readily applied to predict molecular behavior and rationalize experimental observations [2] [7].
The enduring value of Pauling's contributions is confirmed by modern computational studies showing that his qualitative conceptions of hybridization and resonance are robustly manifested across diverse quantum chemistry methodologies [24]. For researchers today, understanding both the historical development and modern computational implementation of these concepts provides valuable perspective on the electronic structure of molecules relevant to pharmaceutical development and materials design.
Beyond the Diatomic: Modern Computational Methods and Expanding Applications of VB Theory
Valence Bond (VB) theory stands as one of the two fundamental quantum mechanical theories, alongside Molecular Orbital (MO) theory, developed to explain chemical bonding [27] [28]. Rooted in the pioneering work of Heitler and London in 1927, VB theory provides a conceptually accessible framework for understanding how covalent bonds form through the overlap of atomic orbitals [28] [29]. The theory's central premise states that "electrons in a molecule occupy atomic orbitals rather than molecular orbitals. The atomic orbitals overlap on the bond formation and the larger the overlap the stronger the bond" [27]. This foundational principle emphasizes the localized nature of chemical bonding in VB theory, where electrons remain associated with their original atomic orbitals rather than delocalizing over the entire molecule.
The Heitler-London approach to the hydrogen molecule provided the first quantum-mechanical treatment of the chemical bond, revealing that covalent bonding arises fundamentally from the interference of atomic wave functions—a phenomenon initially described as "Schwebungsphänomen" in the original German publication [10]. This early work demonstrated that the energy stabilization leading to bond formation emerges from the quantum mechanical interplay of electron pairs occupying overlapping atomic orbitals. Unlike the later developed Molecular Orbital theory, which treats electrons as delocalized throughout the entire molecule, VB theory maintains a more intuitive connection to Lewis structures, depicting bonds as shared electron pairs between specific atoms [28].
The conceptual framework of VB theory has evolved significantly beyond its initial formulation, with modern implementations incorporating more sophisticated mathematical treatments while retaining the core physical intuition of localized electron pairs. The "perfect" form of Heitler-London theory, as described by Epiotis, Larson, and Eaton, demonstrates how the apparent complexity of a complete valence bond basis set can be managed through the use of delocalized atomic orbitals, making the theory both conceptually accessible and computationally rigorous [30]. This evolution has maintained the theory's relevance in modern chemical research, particularly for understanding reaction mechanisms, electronic structures of complex molecules, and bonding in coordination compounds.
The Principle of Linear Combination in VB Structures
Fundamental Concept and Mathematical Basis
The linear combination of VB structures represents a cornerstone of modern valence bond theory, extending the simple Heitler-London model to account for the complex electronic structures of polyatomic molecules. In mathematical terms, a linear combination involves combining a set of mathematical entities by multiplying each by a scalar coefficient and summing the results [31]. Applied to valence bond theory, this principle allows chemists to represent the true wave function of a molecule as a weighted sum of multiple possible VB structures, each depicting a different electron distribution or bonding pattern.
This approach finds its theoretical justification in the quantum mechanical principle of superposition, which permits a system to exist in multiple states simultaneously. For a molecular system, the complete wave function Ψ can be expressed as:
Ψ = c₁Ψ₁ + c₂Ψ₂ + c₃Ψ₃ + ... + cₙΨₙ
where Ψ₁, Ψ₂, ..., Ψₙ represent the wave functions for different valence bond structures, and c₁, c₂, ..., cₙ are numerical coefficients indicating the relative contribution of each structure to the overall wave function [29]. The squares of these coefficients (|cᵢ|²) provide a measure of the relative weight or importance of each contributing structure in describing the molecule's true electronic distribution. This mathematical formalism enables valence bond theory to move beyond the limitations of single Lewis structures, providing a more nuanced and accurate description of molecular bonding.
Increased-Valence Structures and Resonance Theory
A particularly important application of the linear combination principle in VB theory emerges in the description of "increased-valence" structures for electron-rich molecules [29]. These representations provide a qualitative yet powerful method for depicting the electronic structures of molecules with four-electron three-center bonding units, such as FOOF, SF₄, ClF₃, and XeF₂. Increased-valence structures are formally equivalent to resonance between standard Lewis structures (Kekulé-type) and "long-bond" Lewis structures (Dewar-type), often incorporating Pauling "three-electron bond" structures as components [29].
The construction of increased-valence structures typically employs two primary techniques derived from the linear combination principle:
- Delocalization Approach: A non-bonding electron from a Kekulé-type Lewis structure is delocalized into a bonding localized molecular orbital (LMO), generating what is effectively a one-electron bond [29].
- Spin-Pairing Approach: The antibonding electron of a "three-electron bond" configuration spin-pairs with an electron from an adjacent atomic center [29].
These approaches demonstrate how the linear combination of simpler bonding representations generates more complex bonding descriptions that more accurately reflect molecular electronic structures. For example, in the case of FOOF (F₂O₂), increased-valence structures can be derived by considering the spin-pairing of antibonding electrons from the O₂ moiety with fluorine atoms, resulting in bonding descriptions that account for the molecule's unusual bond lengths and stability [29].
Table 1: Types of Valence Bond Structures and Their Characteristics
| Structure Type | Description | Example Molecules | Key Features |
|---|---|---|---|
| Standard Lewis | Obey the octet rule with maximum electron-pair bonds between adjacent atoms | CH₄, H₂O | Localized electron pairs, familiar dash symbols |
| Long-bond Lewis | Contain at least one electron-pair bond linking non-adjacent atoms | FOOF (certain representations) | May violate conventional bonding patterns |
| Increased-Valence | Resonance hybrid of standard and long-bond structures | FOOF, SF₄, XeF₂ | Describe hypervalent molecules without d-orbital participation |
The Maximum Overlap Condition in VB Theory
Fundamental Principle and Bond Strength Relationship
The condition of maximum overlap represents a fundamental determinant of bond strength and directionality in valence bond theory. This principle states that the strength of a covalent bond is directly proportional to the degree of overlap between the participating atomic orbitals [27] [28]. When two atoms approach each other during bond formation, their atomic orbitals interact through various overlapping patterns, with greater overlap leading to more effective electron pairing between nuclei and consequently stronger chemical bonds. The theoretical foundation for this relationship emerges from the quantum mechanical treatment of the hydrogen molecule by Heitler and London, which demonstrated that the energy stabilization associated with bond formation increases with the extent of orbital overlap [10].
The maximum overlap condition provides the theoretical rationale for the observed directional character of covalent bonds in molecules. Since atomic orbitals have specific spatial orientations (e.g., p orbitals are directional, while s orbitals are spherical), the extent of overlap varies significantly with the relative orientation of the approaching atoms. Bonds form along directions that maximize this overlap, naturally explaining the characteristic bond angles in molecules such as water (approximately 104.5°), ammonia (approximately 107°), and methane (109.5°). In the case of methane, VB theory explains the tetrahedral molecular geometry through sp³ hybridization, where carbon's 2s and 2p orbitals mix to form four equivalent hybrid orbitals oriented toward the corners of a tetrahedron, thereby maximizing overlap with hydrogen 1s orbitals [27] [28].
Sigma and Pi Bonds: Differential Overlap and Bonding Patterns
The maximum overlap condition further elucidates the distinction between sigma (σ) and pi (π) bonds in VB theory, which arise from different modes of orbital overlap. Sigma bonds form through head-to-head overlap of atomic orbitals along the internuclear axis, resulting in cylindrical symmetry and maximum orbital overlap [27] [28]. This direct overlap pattern produces the strongest type of covalent bond, forming the fundamental skeleton of molecular structures. In contrast, pi bonds result from lateral or side-by-side overlap of parallel p orbitals, with electron density concentrated above and below the internuclear axis [27]. This parallel overlap is less effective than head-on overlap, resulting in weaker bonds with different chemical properties.
The cumulative effect of these overlapping patterns explains the bonding patterns in molecules with multiple bonds. A single bond consists of one sigma bond, a double bond contains one sigma and one pi bond, while a triple bond comprises one sigma and two pi bonds [27]. This hierarchical bonding scheme directly correlates with bond lengths and strengths—as bond order increases, bond length decreases and bond strength increases—relationships that valence bond theory explains through the additive effects of multiple overlapping interactions. For example, the difference in bond length and strength between H₂ (single bond) and F₂ (weaker single bond) can be explained by differences in the overlapping orbitals involved—the covalent bond in HF forms through overlap of hydrogen's 1s orbital with fluorine's 2p orbital, with the degree of overlap determining bond strength [27].
Table 2: Bond Types and Overlap Characteristics in Valence Bond Theory
| Bond Type | Orbital Overlap | Electron Density | Bond Strength | Examples |
|---|---|---|---|---|
| Sigma (σ) | Head-on along internuclear axis | Concentrated along bond axis | Strongest | H₂, C-C in ethane |
| Pi (π) | Lateral/side-by-side | Above and below internuclear axis | Weaker than sigma | C=C in ethene |
| Single | One sigma overlap | Localized between two atoms | ~350-400 kJ/mol | H₂, F₂, HF |
| Double | One sigma + one pi | More diffuse | ~600-700 kJ/mol | O₂, C₂H₄ |
| Triple | One sigma + two pi | Even more diffuse | ~800-900 kJ/mol | N₂, C₂H₂ |
Comparative Analysis: VB Theory vs. Molecular Orbital Theory
Conceptual and Methodological Differences
Valence Bond theory and Molecular Orbital theory represent two complementary yet distinct approaches to understanding chemical bonding from a quantum mechanical perspective. While both theories aim to explain molecular structure and stability, they employ fundamentally different conceptual frameworks and mathematical formulations. VB theory emphasizes the localization of electrons in atomic orbitals, with chemical bonds forming through overlap between these orbitals on adjacent atoms [28]. This perspective maintains a direct connection to Lewis dot structures and provides an intuitive picture of bonding as shared electron pairs between specific atoms. In contrast, MO theory treats electrons as delocalized over the entire molecule, occupying molecular orbitals constructed from linear combinations of atomic orbitals (LCAO) from all constituent atoms [10] [28].
The divergent approaches of these theories lead to different descriptions of molecular electronic structure. VB theory describes bonding in terms of localized electron pairs formed between two atoms, often utilizing hybridization to explain molecular geometries that cannot be accounted for by pure atomic orbitals [28]. MO theory, developed by Hund and Mulliken in 1932, describes bonding through molecular orbitals that span the entire molecule, categorizing them as bonding, antibonding, or nonbonding based on their energy levels and phase relationships [10] [28]. The molecular orbital approach naturally accommodates delocalized bonding phenomena, such as those in aromatic compounds, without requiring additional concepts like resonance.
The mathematical foundations of these theories also differ significantly. Traditional VB calculations employ wavefunctions that maintain electron identity with specific atoms, leading to computational challenges for larger molecules. MO theory utilizes a delocalized approach from the outset, often resulting in more computationally tractable solutions, especially when combined with modern approximations like density functional theory [10]. However, advances in computational methods, including the "perfect" form of Heitler-London theory that uses delocalized atomic orbitals as a basis, have addressed some limitations of early VB implementations [30].
Relative Strengths and Limitations in Chemical Applications
Both VB and MO theories offer unique insights into chemical bonding, with their relative utility depending on the specific chemical phenomenon under investigation. VB theory excels in providing qualitative understanding of molecular geometry, reaction mechanisms, and bonding patterns in coordination compounds [28]. Its strong connection to traditional chemical intuition makes it particularly valuable for explaining stereochemistry, hypervalency in main-group compounds, and the concept of resonance in conjugated systems [29]. The theory's emphasis on localized bonds facilitates understanding of bond dissociation processes and radical reactions.
MO theory provides superior capabilities for interpreting molecular spectroscopy, predicting magnetic properties, and understanding delocalized bonding in extended systems [28]. Its description of bonding and antibonding orbitals offers straightforward explanations for phenomena such as paramagnetism in O₂, electronic spectra of complex molecules, and band structures in solids [10] [28]. MO theory's quantitative predictive power for properties like ionization potentials, electron affinities, and bond energies generally exceeds that of traditional VB approaches, particularly for systems with extensive electron delocalization.
Table 3: Comparative Analysis of Valence Bond and Molecular Orbital Theories
| Characteristic | Valence Bond Theory | Molecular Orbital Theory |
|---|---|---|
| Fundamental Focus | Overlap of atomic orbitals between adjacent atoms [28] | Molecular orbitals spanning entire molecule [28] |
| Electron Distribution | Localized in specific atomic orbitals [28] | Delocalized throughout molecular orbitals [28] |
| Bonding Picture | Localized electron pairs between two atoms [28] | Delocalized bonds involving multiple centers [28] |
| Hybridization | Utilized extensively to explain molecular shapes [28] | Not inherently required [28] |
| Resonance | Employs multiple VB structures [27] | Describes delocalization naturally with single determinant |
| Computational Tractability | Challenging for large/delocalized systems [28] | More amenable to computational treatment [28] |
| Explanatory Strengths | Molecular geometry, reaction mechanisms [28] | Spectroscopy, magnetic properties [28] |
Experimental Methodologies and Research Tools
Computational Approaches for VB Analysis
Modern computational chemistry provides powerful tools for investigating the principles of linear combination and maximum overlap in valence bond theory. While traditional VB computations faced challenges due to the non-orthogonality of atomic orbitals and the large number of configurations required for accurate descriptions, contemporary methods have overcome many of these limitations. Orbital-based bonding analysis in solids, as implemented in software packages like LOBSTER (Local Orbital Basis Suite Towards Electronic-Structure Reconstruction), enables researchers to extract chemical bonding information from plane-wave density functional theory calculations by transforming the results into a local orbital basis [10]. This approach facilitates the calculation of wave function-based atomic charges, various population analyses, periodic bonding indicators, and first-principles bond orders, bridging the gap between quantitative electronic structure calculations and qualitative chemical bonding models [10].
The LOBSTER package specifically addresses the challenge of analyzing periodic solid-state structures using valence bond concepts. Since electronic structures for periodic solids are most often calculated using plane waves (due to translational symmetry and Bloch's theorem), a unitary transformation to atomic or molecular orbitals is necessary for chemical bonding analysis [10]. This software implements major developments in wave function-based bonding analysis within a periodic solid-state context, allowing for two- and multi-centre bonding analysis, fragment-molecular analysis, and other techniques that align with traditional VB concepts [10]. These computational advances have been particularly valuable for understanding the bonding in solid-state systems deriving from carbonate chemistry and other extended materials where delocalized bonding plays a crucial role.
Spectroscopic and Structural Techniques
Experimental validation of valence bond concepts relies heavily on spectroscopic and structural techniques that probe molecular geometry, electron distribution, and bonding characteristics. X-ray crystallography provides direct evidence for bond lengths and angles, offering quantitative data that reflects the consequences of orbital overlap—shorter bonds typically indicate greater overlap and stronger bonding interactions. The unusual bond lengths in molecules like FOOF (O-F bond length of approximately 1.575 Å) provide critical testing grounds for increased-valence VB descriptions, which aim to explain these structural features without invoking valence shell expansion for second-row elements [29].
Spectroscopic methods including UV-visible, infrared, and nuclear magnetic resonance (NMR) spectroscopy offer complementary insights into electronic structure and bonding. UV-visible spectroscopy probes electronic transitions between molecular orbitals, providing indirect information about energy level separations that reflect bonding patterns. Infrared spectroscopy measures vibrational frequencies that correlate with bond strengths, offering experimental verification of the maximum overlap principle—stronger bonds resulting from greater orbital overlap typically exhibit higher vibrational frequencies. NMR spectroscopy, particularly chemical shift and coupling constant measurements, provides information about electron distribution around nuclei, helping to distinguish between bonding models. For fluorine-containing molecules, ¹⁹F NMR serves as a particularly sensitive probe of bonding environment, enabling researchers to test predictions from increased-valence VB structures [29].
Table 4: Essential Research Tools for Valence Bond Theory Investigations
| Research Tool | Primary Function | Application in VB Theory |
|---|---|---|
| LOBSTER Software | Orbital-based bonding analysis in periodic systems [10] | Extracts chemical bonding information from plane-wave DFT calculations |
| X-ray Crystallography | Determines molecular geometry with high precision | Provides bond length/angle data to test overlap predictions |
| NMR Spectroscopy | Probes local electronic environment around nuclei | Validates electron distribution in increased-valence structures |
| Vibrational Spectroscopy | Measures bond vibration frequencies | Correlates frequency with bond strength from orbital overlap |
| Photoelectron Spectroscopy | Measures ionization energies from specific orbitals | Probes energy levels related to bonding/antibonding character |
| Computational VB Packages | Performs modern valence bond calculations | Implements linear combination of VB structures quantitatively |
The core principles of linear combination of VB structures and the maximum overlap condition continue to provide fundamental insights into chemical bonding, maintaining the relevance of valence bond theory in modern chemical research. The linear combination principle extends the simple Heitler-London model to complex molecular systems through increased-valence representations and resonance concepts, offering a qualitatively intuitive yet quantitatively rigorous description of electronic structure [29]. Meanwhile, the maximum overlap condition establishes the direct relationship between orbital interaction and bond strength, explaining molecular geometries and reactivity patterns across diverse chemical systems [27] [28].
While molecular orbital theory has gained prominence for quantitative computational work and interpretation of spectroscopic data, valence bond theory remains indispensable for understanding chemical reactivity, molecular geometry, and bonding in electron-rich systems [28]. The conceptual accessibility of VB theory, particularly its connection to traditional Lewis structures and its ability to represent intermediate bonding situations through resonance, ensures its continued utility in chemical education and research. Modern computational implementations have addressed many historical limitations of the theory, enabling quantitative applications that retain the conceptual clarity of the valence bond approach [10] [30].
The ongoing development of both theoretical methodologies and experimental techniques promises to further refine our understanding of these fundamental principles. As computational power increases and experimental resolution improves, researchers can explore more complex bonding situations where the linear combination of VB structures and subtle variations in orbital overlap dictate molecular properties and reactivity. These advances ensure that the core principles of valence bond theory will continue to inform chemical research across disciplines including drug development, materials science, and catalysis, where understanding bonding at the molecular level enables rational design of new compounds with tailored properties and functions.
The Heitler-London (HL) model, foundational to modern valence bond (VB) theory, has long provided an intuitive, chemically accurate picture of covalent bonding. For decades, however, its computational implementation was hindered by programming complexities compared to the more straightforward algorithms of molecular orbital (MO) methods, limiting its use for high-accuracy calculations [8] [32]. Recent methodological breakthroughs are systematically overcoming these hurdles, enabling valence bond theory to achieve a new level of accuracy that is highly competitive with, and in some cases superior to, leading MO-based approaches, all while maintaining computational economy.
Historical Programming Challenges and Modern Solutions
The decline of VB theory's popularity in computational chemistry was not due to a lack of descriptive power but largely because MO methods were easier to program for digital computers [8]. MO theory's use of delocalized orbitals that span the entire molecule streamlined the computational process. In contrast, the core of traditional VB calculations involves a linear combination of several valence bond structures, each described using localized atomic orbitals, leading to a wavefunction that is inherently multi-configurational [8]. This formulation, while chemically intuitive, introduced significant programming complications, especially concerning the handling of non-orthogonal orbitals, which made VB computations more complex and computationally expensive to implement and run [32].
Modern advances have directly addressed these challenges. The development of sophisticated classical VB programs has closed the gap, making VB calculations competitive in accuracy and economy with post-Hartree-Fock methods [8]. Furthermore, the Density Functional Valence Bond (DFVB) approach represents a pivotal innovation. Methods like the Hamiltonian matrix correction-based DFVB (hc-DFVB) integrate the multi-configurational strength of VB wavefunctions with density functional theory (DFT) to efficiently account for dynamic electron correlation, which is crucial for quantitative accuracy [33]. This hybrid strategy delivers high-fidelity results without the prohibitive computational cost of traditional post-HCSCF methods like valence bond configuration interaction (VBCI) [33].
Quantitative Performance Comparison
The following tables summarize key experimental data and benchmarks comparing modern VB methods with other computational approaches.
Method Performance on Diatomic Molecules and Excited States
Table 1: Comparative performance of VB and other methods for molecular properties and excited states.
| Method | System(s) Studied | Key Performance Metric | Result / Accuracy | Comparative Note |
|---|---|---|---|---|
| Screening-Modified HL [34] | H₂ (ground state) | Bond Length, Binding Energy | "Substantially improved agreement with experimental bond length" | A simple analytical modification yielding high accuracy. |
| hc-DFVB [33] | C₂H, CN, BO, CO⁺ (excited states) | Excitation Energy | "Significantly better excitation energies compared to VBSCF"; correct state ordering. | Outperforms standard VBSCF; reliable for complex excited states. |
| VBSCF [33] | C₂H, CN, BO, CO⁺ (excited states) | Excitation Energy & State Ordering | Shows "some ordering inconsistencies" compared to hc-DFVB. | Provides a qualitative foundation but lacks quantitative dynamic correlation. |
| MO-based MRDFT | General strongly correlated systems | Treatment of Static & Dynamic Correlation | Faces "double-counting error" and "symmetry dilemma" [33]. | VB-based MRDFT (e.g., hc-DFVB) is designed to avoid these issues. |
Computational Economy and Application Scope
Table 2: Comparison of computational cost, scalability, and application scope.
| Feature / Method | Classical VBSCF / Post-VBSCF | Modern DFVB (e.g., hc-DFVB) | Traditional MO-CI/CASPT2 |
|---|---|---|---|
| Computational Cost | High (exponential scaling with active space) [33] | Moderate (DFT cost for correlation) [33] | High to Prohibitive (scaling with system size) [33] |
| Handling of Dynamic Correlation | Requires expensive post-VBSCF methods (VBCI, VBPT2) [33] | Integrated via DFT functionals [33] | Requires expensive post-MCSCF methods (MRCI, CASPT2) [33] |
| Ideal for System Size | Small to medium molecules [33] | Small to large systems (economical) [33] | Small to medium molecules (accurate but costly) [33] |
| Key Application Strength | Qualitative bonding insight, static correlation [8] | Strongly correlated excited states, avoided crossings [33] | Spectroscopy, multi-reference character [33] |
Experimental Protocols and Workflows
Protocol: Screening-Modified Heitler-London Calculation
This protocol details the procedure for improving the standard HL model with an effective screening parameter, as demonstrated for the H₂ molecule [34].
- Wavefunction Initialization: Begin with the standard HL wavefunction, (\psi{\pm}(\vec{r}1, \vec{r}2) = N{\pm} [\phi(r{1A})\phi(r{2B}) \pm \phi(r{1B})\phi(r{2A})]), where (\phi(r_{ij})) is the 1s atomic orbital [34].
- Parameter Introduction: Modify the atomic orbitals to include a single variational parameter, (\alpha), acting as an effective nuclear charge: (\phi(r{ij}) = \sqrt{\frac{1}{\pi}} e^{-\alpha r{ij}}) [34] [35].
- Variational Optimization: For a fixed inter-proton distance (R), optimize the parameter (\alpha) to minimize the total energy. This can be achieved using a variational quantum Monte Carlo (VQMC) approach, which samples the electronic coordinates to compute the energy expectation value [34] [35].
- Function Construction: Repeat the optimization across a range of (R) values to construct a function (\alpha(R)) that describes how screening changes with bond length [34].
- Property Calculation: Use the optimized (\alpha(R))-dependent wavefunction to compute molecular properties like the potential energy curve, from which the bond length, dissociation energy, and vibrational frequency are derived [34] [35].
Protocol: Hamiltonian-Corrected DFVB (hc-DFVB) Calculation
This protocol outlines the hc-DFVB method for studying systems with strong electron correlation and excited states [33].
- VB Structure Generation: For the target molecule, generate a complete set of VB structures that span the defined active space. These structures can be built from localized atomic orbitals or fragment orbitals [33].
- VBSCF Calculation: Perform a Valence Bond Self-Consistent Field (VBSCF) calculation. This optimizes the orbitals and coefficients for the VB structures, providing a wavefunction that accounts for static electron correlation [33].
- Hamiltonian Matrix Correction: Construct an effective Hamiltonian matrix where the diagonal elements are replaced with energies calculated using a DFT functional for each VB determinant. This step incorporates dynamic correlation energy in a size-consistent manner [33].
- Diagonalization and State Interaction: Diagonalize the corrected Hamiltonian matrix. This final step produces the final adiabatic states (energies and wavefunctions) and inherently includes multi-state interactions, which is crucial for describing avoided crossings and conical intersections [33].
Diagram illustrating the step-by-step workflow for the hc-DFVB computational protocol, highlighting key stages of structure generation, correlation integration, and state resolution.
The Scientist's Toolkit: Essential Research Reagents and Materials
Table 3: Key computational tools and concepts in modern valence bond theory.
| Tool / Concept | Function / Role in Calculation |
|---|---|
| VB Structures | The fundamental "building blocks" of the VB wavefunction. Each structure represents a specific, classical Lewis-type electron pairing scheme [8]. |
| Effective Nuclear Charge (α) | A variational parameter in modified HL models that accounts for the screening of the nucleus by other electrons, allowing atomic orbitals to contract or expand for lower energy [34] [35]. |
| Variational Quantum Monte Carlo (VQMC) | A computational method used to optimize wavefunction parameters (like α). It uses stochastic (random) sampling to compute quantum mechanical expectation values efficiently [34]. |
| Hamiltonian Matrix Correction (hc) | The core of the hc-DFVB method. It replaces diagonal elements of the VB Hamiltonian with energies from DFT, seamlessly integrating dynamic correlation [33]. |
| Fragment Orbitals | Pre-defined molecular orbitals from parts of a larger system, used as a basis set for constructing VB wavefunctions for complex molecules, enhancing computational efficiency [8]. |
The computational landscape for valence bond theory has been fundamentally transformed. The programming hurdles that once relegated VB theory to a conceptual role have been surmounted by modern implementations that are both robust and economical. The advent of methods like hc-DFVB, which synergistically combines the intuitive, multi-reference picture of VB with the computational efficiency of DFT, demonstrates that VB theory is not only back but is a powerful tool for tackling complex quantum chemical problems—from ground-state bonding to challenging excited-state interactions—with precision that rivals the best contemporary MO-based approaches.
Valence Bond (VB) theory, with its roots in the seminal work of Heitler and London on the hydrogen molecule, has evolved into a sophisticated computational framework capable of providing deep insights into chemical bonding and reactivity [2] [14] [36]. While the classical VB approach, as championed by Pauling, utilized localized, non-orthogonal atomic orbitals, modern developments have introduced powerful alternatives that balance chemical intuition with computational tractability [37]. Two such advanced frameworks are Spin-Coupled Valence Bond and methods employing Delocalized Fragment Orbitals. These approaches represent a significant evolution from the original Heitler-London model, extending its core principles to complex molecular systems while addressing historical limitations related to computational cost and the description of electron correlation [38] [37]. This guide objectively compares these methodologies, detailing their theoretical foundations, performance characteristics, and practical applications in chemical research, particularly in areas relevant to drug development.
Theoretical Framework and Comparative Methodology
Spin-Coupled Valence Bond Theory
Spin-Coupled Valence Bond represents a modern VB method that captures essential electron correlation through the description of non-orthogonal, atom-centered orbitals coupled with a sophisticated treatment of spin interactions [38] [37]. Unlike early VB methods that fix orbital forms, SCVB optimizes both the orbitals and the spin coupling between them, providing a more flexible and accurate wavefunction. This approach maintains the localized bonding picture central to classical VB theory while offering a improved description of bond breaking and multi-reference character [37]. The mathematical formulation typically involves a multi-configurational wavefunction where electrons in active orbitals are coupled in different spin patterns, with all orbitals and coefficients variationally optimized.
Fragment Orbital Methods in Valence Bond
Fragment Orbital methods represent a different evolution within modern VB theory, utilizing delocalized orbitals to reduce computational complexity [38]. These approaches, including the Fragment Molecular Orbital method developed by Hoffmann and coworkers, partition molecular systems into logical subunits or fragments [10]. The total wavefunction is then constructed from orbitals that can be delocalized over these fragments, rather than strictly localized on individual atoms. This method is particularly valuable for analyzing inter-fragment interactions in large systems, such as enzyme-substrate complexes or supramolecular assemblies [10]. By moving to a fragment basis rather than an atomic one, these methods maintain computational feasibility while preserving much of the chemical interpretability of VB theory.
Table: Fundamental Characteristics of Advanced VB Methods
| Feature | Spin-Coupled VB | Fragment Orbital VB |
|---|---|---|
| Orbital Type | Non-orthogonal, atom-centered | Delocalized over molecular fragments |
| Electron Correlation | Extensive through orbital and spin optimization | Moderate, depends on fragment size and treatment |
| Computational Scaling | Higher due to non-orthogonality | Improved through fragment approximation |
| Chemical Interpretability | High - maintains localized bond picture | Moderate - focuses on fragment interactions |
| Primary Application Domain | Bond breaking, diradicals, excited states | Large systems, enzyme catalysis, supramolecular chemistry |
Comparative Performance Analysis
Computational Efficiency and Accuracy
The computational performance of these methods varies significantly due to their different theoretical constructs. Spin-Coupled VB methods face challenges with computational complexity arising from the non-orthogonality of the atomic orbitals [37]. This non-orthogonality necessitates expensive integral transformations and more complex Hamiltonian matrix elements. However, this computational cost brings substantial benefits in accuracy, particularly for electron correlation effects [38].
Fragment Orbital methods offer substantially improved computational efficiency by reducing the number of interacting units through fragment delocalization [10] [38]. By treating groups of atoms as single fragments, these methods decrease the configuration space and simplify the wavefunction construction. The trade-off involves potential loss of detail in bonding descriptions between atoms within the same fragment. For very large systems such as enzyme-active sites or supramolecular complexes, Fragment Orbital methods often represent the only practical VB-based approach [10] [39].
Table: Computational Performance Comparison
| Performance Metric | Spin-Coupled VB | Fragment Orbital VB |
|---|---|---|
| System Size Limit | ~12-14 electrons (full configuration expansion) | Much larger systems feasible |
| Bond Dissociation | Excellent - describes entire potential curve | Good for equilibrium, limited for full dissociation |
| Static Correlation | Excellent through multi-configurational treatment | Moderate, depends on fragment definitions |
| Dynamic Correlation | Requires additional corrections (VBCI, VBPT2) | Similar need for dynamic correlation recovery |
| Software Availability | Specialized codes (CASVB, BOVB) | Implemented in major packages (LOBSTER) |
Chemical Interpretation and Bonding Analysis
The interpretative power of VB methods represents one of their primary advantages over Molecular Orbital-based approaches. Spin-Coupled VB provides exceptional insight into bond formation processes and electronic structures that challenge single-reference methods [37]. For example, SCVB naturally describes the intricate bonding situation in molecules like O₂, where it can represent the bonding as two 3-electron-2-center bonds, resolving the historical limitations of early VB theory in explaining oxygen's paramagnetism [37].
Fragment Orbital methods excel in analyzing intermolecular interactions and chemical reactivity in complex systems [10] [39]. By focusing on fragment-based interactions, these methods bridge the gap between quantum mechanical descriptions and chemical intuition about functional group interactions. This makes them particularly valuable for studying enzyme catalysis, where the interaction between specific molecular fragments (substrate, cofactors, key amino acids) drives reactivity [39].
Evolution of Modern Valence Bond Methods from Heitler-London Foundations
Experimental Protocols and Applications
Protocol: Spin-Coupled VB for Bond Breaking Analysis
The application of Spin-Coupled VB to study bond dissociation follows a well-defined computational protocol:
Active Space Selection: Identify the electrons and orbitals involved in the bond cleavage process. For a single bond, this typically includes the two electrons and two orbitals directly participating in the bond.
Orbital Optimization: Variationally optimize the forms of the active orbitals without constraints of orthogonality, allowing them to adapt to the changing bonding environment along the dissociation coordinate.
Spin Function Optimization: Optimize the spin coupling patterns between the unpaired electrons, allowing for all possible spin alignments consistent with the total spin symmetry.
Configuration Interaction: Include additional configurations beyond the fundamental spin-coupled state to capture dynamic correlation effects, typically through Valence Bond Configuration Interaction or Valence Bond Perturbation Theory [37].
Property Calculation: Compute relevant properties (energies, densities, spin distributions) along the reaction coordinate to analyze the bonding evolution.
This protocol has been successfully applied to challenging systems such as the C₂ molecule, where SCVB provides evidence supporting a bond order greater than two, resolving longstanding controversies about its electronic structure [36].
Protocol: Fragment Orbital VB for Enzyme Catalysis
For studying enzymatic reactions using Fragment Orbital VB methods, the following protocol is recommended:
System Fragmentation: Divide the enzyme-substrate complex into logical fragments based on chemical functionality - typically separating substrate, cofactor, and key amino acid side chains [39].
Fragment Orbital Calculation: Compute the orbitals for each fragment in isolation or in a simplified embedding environment to capture their essential electronic character.
Interaction Analysis: Calculate the interaction matrix elements between fragment orbitals to identify dominant bonding and antibonding interactions driving the catalytic process.
VB Wavefunction Construction: Build the total VB wavefunction from the fragment orbitals, typically focusing on key resonance structures that represent different bonding patterns along the reaction pathway.
Energy Decomposition: Analyze the contribution of different resonance structures to the transition state stabilization, identifying electronic factors responsible for catalytic efficiency [39].
This approach has proven particularly powerful in studying cytochrome P450 enzymes, where Fragment Orbital VB methods have elucidated the electronic origins of activation barriers in hydrogen abstraction reactions [39].
Table: Application Performance in Key Chemical Systems
| Chemical System/Property | Spin-Coupled VB Performance | Fragment Orbital VB Performance |
|---|---|---|
| H₂ Bond Dissociation | Excellent - recovers full correlation energy | Limited - fragment approximation less suitable |
| O₂ Electronic Structure | Excellent - correct diradical description | Good - correct spin state with appropriate fragments |
| Enzyme Catalysis Mechanisms | Limited by system size | Excellent - captures key interactions efficiently |
| Aromaticity | Good - but computationally demanding | Very good - efficient for large conjugated systems |
| Transition Metal Complexes | Good for small active sites | Better for complete ligand field analysis |
Research Reagent Solutions
Table: Essential Computational Tools for Advanced VB Research
| Tool/Software | Function | Method Compatibility |
|---|---|---|
| LOBSTER Package | Periodic bonding analysis, orbital transformation | Primarily Fragment Orbital Methods |
| CASVB Module | Expresses MO-based wavefunctions in VB terms | Both SCVB and Fragment Methods |
| BOVB Methods | Breathing orbital treatment for dynamic correlation | Spin-Coupled VB |
| VBSCF Methods | Self-consistent field optimization for VB wavefunctions | Both SCVB and Fragment Methods |
| XMVB Program | Ab initio VB calculations with various correlation treatments | Both SCVB and Fragment Methods |
Method Selection Guide Based on Research Objectives
Spin-Coupled Valence Bond and Fragment Orbital methods represent complementary evolutions of the foundational Heitler-London approach, each extending its capabilities to address different challenges in computational chemistry. Spin-Coupled VB maintains the strongest connection to the original localized bonding picture, providing unparalleled insights into electron correlation and bond formation at the cost of higher computational demands. Fragment Orbital VB sacrifices some granularity in bonding description to achieve practical applicability to large systems, particularly biological assemblies and materials.
The continuing development of both methodologies suggests a future where VB theory reclaims its position as an equal partner to Molecular Orbital theory, with each providing unique insights into different aspects of molecular structure and reactivity. For drug development professionals, Fragment Orbital methods offer immediate practical value for studying enzyme-substrate interactions and catalytic mechanisms. For fundamental studies of bonding and reactivity, Spin-Coupled VB remains the gold standard for accuracy and interpretability within the valence bond framework.
The Heitler-London theory, originating from the first quantum-mechanical treatment of the hydrogen molecule in 1927, represents the foundational framework for understanding the chemical bond [10] [2]. This pioneering work demonstrated that bonding occurs primarily through the pairing of electrons between atoms, with the highest electron density located between the bonded nuclei [40]. The core insight of Heitler and London was recognizing that covalent bonding stems from the interference of atomic wave functions—a phenomenon they termed "Schwebungsphänomen" in the original German publication [10]. This valence bond (VB) approach, emphasizing strictly correlated electrons occupying atom-centered orbitals, explained chemical bonding in terms highly compatible with Lewis's electron-pair bond model [41] [2].
Despite its groundbreaking nature, the original Heitler-London approach possessed significant limitations. It could not adequately explain the formation of odd-electron molecules, coordinate bonds, pi-bonding systems, or molecular stereochemistry [40]. These shortcomings prompted successive theoretical developments, including Pauling and Slater's improvements incorporating directional bonding concepts through orbital hybridization [40], and the parallel development of molecular orbital (MO) theory by Hund, Mulliken, and others [10] [2]. Modern valence bond theory has evolved into a computationally sophisticated framework that competes in accuracy with post-Hartree-Fock methods while retaining the chemically intuitive appeal of localized bonds [8]. This article examines how this evolved Heitler-London paradigm, through its modern VB successors, provides unique insights into aromatic systems, reaction mechanisms, and biomolecular interactions compared to alternative computational approaches.
Theoretical Frameworks: Comparative Principles and Methodologies
Valence Bond Theory Evolution from Heitler-London
The modern valence bond theory represents a significant evolution from the original Heitler-London model. While the initial approach described H₂ using a purely covalent wavefunction (Φₕₗ) combining two VB determinants [8], contemporary implementations address its limitations through several key advancements:
Multi-structure approach: Modern VB overcomes the original method's inability to describe diverse bonding situations by representing the total wavefunction as a linear combination of several valence bond structures, incorporating both covalent and ionic contributions [8]. For H₂, this appears as Φᵥᵦₜ = λΦₕₗ + μΦᵢ, where λ and μ are coefficients determining covalent versus ionic character [8].
Delocalized basis functions: Contemporary implementations employ more flexible basis sets, including delocalized atomic orbitals (Coulson-Fischer theory) or molecular orbital fragments, overcoming the simple atomic orbital limitations of the original approach [8].
Computational efficiency: New algorithms and programming approaches have made VB calculations competitive with MO-based methods in accuracy and economy, reversing the early computational disadvantages that led to MO theory's dominance [8].
Competing Theoretical Frameworks
Table: Comparison of Quantum Chemical Methods for Molecular Systems
| Method | Theoretical Basis | Bond Description | Strengths | Limitations |
|---|---|---|---|---|
| Modern VB Theory | Linear combination of VB structures [8] | Localized electron pairs, resonance hybrids [8] | Chemical intuitiveness, accurate bond dissociation [8] | Computational complexity, non-orthogonality issues [36] |
| Molecular Orbital Theory | Linear combination of atomic orbitals [10] [8] | Delocalized molecular orbitals [10] | Computational efficiency, straightforward interpretation of spectroscopy [10] [2] | Incorrect dissociation limits (single-determinant) [8] |
| Density Functional Theory | Electron density functional [10] | Electron density distribution [10] | Favourable cost-accuracy ratio [36] | Difficult to interpret bonding, functional dependence [10] |
Quantum Information Theory Framework
A recently developed approach applies quantum information theory (QIT) to chemical bonding, using orbital entanglement patterns to recover both Lewis and beyond-Lewis bonding structures [41]. This framework introduces maximally entangled atomic orbitals (MEAOs), with multipartite entanglement serving as a comprehensive index of bond strength [41]. The method effectively characterizes bonding not only at equilibrium geometries but also during chemical reactions and in complex phenomena like aromaticity, providing a unified perspective that bridges conceptual gaps between traditional theories [41].
Experimental and Computational Methodologies
Precision Spectroscopy of Fundamental Systems
Precision spectroscopy of simple molecular systems like hydrogen provides critical experimental benchmarks for theoretical methods. Advanced techniques include:
Cavity ring-down spectroscopy: Achieves precision measurements of near-infrared electric quadrupole transitions in H₂ molecules with uncertainties at state-of-the-art levels, verifying quantum theoretical calculations with up to 9 significant digits [14].
Frequency comb-calibrated saturation spectroscopy: Enables Doppler-free Lamb-dip spectrum observations of overtone transitions in HD molecules with frequency measurement accuracy reaching 10 significant digits [14].
Vacuum ultraviolet laser systems: "Chirp"-free systems improve measurement accuracy of H₂ dissociation energy to sub-MHz levels, providing key experimental evidence for fundamental constants [14].
These spectroscopic methods validate theoretical predictions by comparing experimental results with first-principles calculations incorporating relativistic and quantum electrodynamics (QED) corrections [14].
Computational Bonding Analysis Protocols
Table: Computational Methodologies for Bonding Analysis
| Method | Key Features | Electron Correlation Treatment | Size Consistency | Typical Applications |
|---|---|---|---|---|
| RHF/UHF SCF | Single determinant, restricted or unrestricted orbitals [36] | Limited (none for RHF) [36] | Yes [36] | Initial geometry optimization, molecular properties [36] |
| CASSCF | Multi-configurational, active space selection [36] | Static correlation within active space [36] | Yes [36] | Bond breaking, diradicals, excited states [36] |
| MRCI | Multi-reference with configuration interaction [36] | Static and dynamic correlation [36] | No (corrected) [36] | Accurate potential energy surfaces [36] |
| CCSD(T) | Single-reference coupled cluster [36] | Dynamic correlation (gold standard) [36] | Yes [36] | Equilibrium properties, weak interactions [36] |
| DFT | Electron density functional [36] | Approximate via functional [36] | Yes [36] | Large systems, transition metals [36] |
| Modern VB | Valence bond structures, non-orthogonal orbitals [8] | Configurations in VB space [8] | Yes [8] | Bonding analysis, reaction mechanisms [8] |
For solid-state systems, the LOBSTER package performs orbital-based bonding analysis by transforming plane-wave DFT results into local atomic orbitals, enabling population analyses, bond orders, and crystal orbital overlap population (COOP) calculations [10].
Research Reagent Solutions for Computational Chemistry
Table: Essential Computational Tools for Bonding Analysis
| Tool/Code | Function | Application Context |
|---|---|---|
| LOBSTER | Orbital-based bonding analysis for periodic solids [10] | Solid-state bonding analysis [10] |
| MOLPRO | High-accuracy ab initio calculations [36] | Molecular electronic structure [36] |
| CASVB | Valence bond analysis of MO-based wavefunctions [36] | VB interpretation of multi-configurational calculations [36] |
| Aug-cc-pwCVTZ basis sets | Polarized triple-zeta with core-valence correlation [36] | Accurate correlation-consistent calculations [36] |
Comparative Performance in Key Chemical Systems
Aromatic Systems and Delocalization
Aromaticity represents a critical test case for theoretical methods, with modern VB theory providing particularly insightful perspectives:
Resonance theory foundation: VB theory naturally incorporates the resonance concept that Pauling described as essential for understanding aromatic stabilization, providing a theoretical foundation for this "chemical feeling" developed through practice [42].
Kekulé structures in benzene: Modern VB treatments of benzene explicitly include both Kekulé and Dewar structures, quantitatively describing the resonance energy that stabilizes the aromatic system [8]. This approach accurately predicts benzene's symmetric structure and bond length equalization.
Beyond-Lewis bonding: The quantum information theory framework reveals multicenter entanglement patterns in aromatic systems, recovering both traditional Lewis and more complex bonding descriptions without a priori chemical assumptions [41].
Reaction Mechanisms and Bond Formation
The description of chemical reaction pathways highlights fundamental differences between theoretical approaches:
Size consistency and dissociation: Modern VB theory correctly describes bond dissociation, unlike single-determinant MO methods [8]. For H₂, MO theory with configuration interaction becomes equivalent to VB description, allowing proper dissociation [8].
Transition state characterization: VB methods provide detailed insights into transition state electronic structure through resonance between reactant, product, and ionic structures, offering intuitive understanding of reaction barriers [8].
Diradical and open-shell systems: For challenging cases like singlet oxygen, modern VB correctly predicts the triplet ground state with two three-electron π-bonds, overcoming limitations of simple Lewis structure depictions [8].
Complex Bonding Situations
Specific molecular systems reveal distinctive capabilities of different theoretical approaches:
Dicarbon (C₂) controversy: The bonding in C₂ presents a challenge, with qualitative MO theory suggesting a double bond while some modern VB calculations indicate quadruple bond character [36]. Generalized valence bond (GVB) studies propose an alternative interpretation of a traditional σ-bond with antiferromagnetically coupled electrons in remaining orbitals [36].
Solid-state materials: For extended systems, VB-inspired approaches like COOP (Crystal Orbital Overlap Population) transformed solid-state chemistry by moving beyond oversimplified ionic models to describe covalent interactions in materials like GaP [10].
Emerging Frontiers and Future Directions
Quantum Information Theory in Bonding Analysis
The emerging quantum information theory approach represents a paradigm shift in bonding analysis:
Orbital entanglement: QIT characterizes bonding through orbital entanglement patterns, with maximally entangled atomic orbitals (MEAOs) recovering both traditional and beyond-Lewis bonding concepts [41].
Multipartite entanglement: This serves as a comprehensive index of bond strength, effective for equilibrium geometries, transition states, and complex phenomena like aromaticity [41].
Unified framework: The QIT approach has potential to elevate Hilbert space atomic partitioning to match the prevalence of real-space partitioning in atoms-in-molecules theory, offering rigorous, quantitative descriptors for fuzzy chemical concepts [41].
Advanced Spectroscopic Validation
Future developments will leverage increasingly precise experimental data:
Fundamental constant determination: Precision spectroscopy of hydrogen molecules enables accurate determination of fundamental constants like the proton-to-electron mass ratio, providing stringent tests for theoretical predictions [14].
Beyond Standard Model physics: Advances in measurement accuracy beyond 10⁻¹⁰ may reveal "new physics" beyond the Standard Model, testing the limits of current quantum chemical methods [14].
Methodological Integration
The convergence of theoretical approaches promises enhanced capabilities:
Unitary transformation bridge: The formal equivalence between MO and VB theories through unitary transformations ensures they describe the same wavefunction differently, enabling cross-fertilization of concepts and methodologies [8].
Multi-method workflows: Integrated computational approaches combining VB interpretability with MO/DFT efficiency provide comprehensive bonding analyses across molecular and solid-state systems [10] [36].
The Heitler-London approach, through its evolution into modern valence bond theory, remains a vital framework for understanding chemical bonding across diverse molecular systems. While molecular orbital theory and density functional methods dominate computational practice for practical reasons, modern VB provides unique chemical insights particularly valuable for understanding aromaticity, reaction mechanisms, and complex bonding situations. The recent integration of quantum information concepts with chemical bonding analysis promises to bridge remaining conceptual gaps, offering rigorous quantification of traditionally qualitative chemical concepts. For researchers in drug development and molecular design, this multi-perspective approach enables more nuanced understanding of biomolecular interactions, where the complementary strengths of different theoretical frameworks can be leveraged for specific challenges. As precision spectroscopic validation continues to advance, further refinement of these computational tools will enhance their predictive power for both fundamental studies and applied molecular design.
Addressing Perceived Failures: How Modern VB Theory Solves Historical Challenges
Molecular oxygen (O₂) stands as a remarkable exception in the realm of diatomic molecules due to its triplet ground state, a property first explained through molecular orbital theory by Mulliken in the 1920s [43]. Unlike most molecules encountered in daily life that exist in a singlet state, ground-state oxygen contains two unpaired electrons, making it a stable diradical [44]. This triplet configuration, denoted as³Σ⁻g, fundamentally influences oxygen's chemical behavior, including its paramagnetism and relatively slow reaction kinetics with singlet-state molecules [44] [45]. The Heitler-London approach to valence bond (VB) theory, while successfully explaining the electron-pair bond in hydrogen, requires significant modification to account for this unusual electronic structure, primarily through the concept of three-electron bonding [44] [2].
This article examines the triplet ground state of oxygen through the lens of modern valence bond theory, comparing its explanatory power with molecular orbital theory and presenting key experimental data that illuminate the electronic structure and reactivity of this biologically and industrially crucial molecule.
Theoretical Frameworks: Explaining Oxygen's Electronic Structure
Molecular Orbital Theory Perspective
Molecular orbital (MO) theory provides the most direct explanation for oxygen's paramagnetic triplet ground state. According to MO theory, the combination of two oxygen atoms results in molecular orbitals where the two highest-energy electrons occupy two degenerate π* antibonding orbitals separately [44]. In accordance with Hund's rules, these electrons remain unpaired and spin-parallel, giving rise to a total electron spin of S=1 [44]. This configuration corresponds to three degenerate spin states (triplet state), which is energetically more favorable than the singlet configuration where the electrons would be paired [44].
The MO description reveals a bond order of 2 for O₂, resulting from one full σ bond and two π half-bonds, each accounted for by two-center three-electron (2c-3e) bonding [44]. This successfully explains both the paramagnetism observed experimentally and the relatively short bond length (121 pm) compared to what would be expected for a single bond (147.5 pm in hydrogen peroxide) [44].
Valence Bond Theory and the Three-Electron Bond
Valence bond theory, with its roots in the Heitler-London description of H₂, traditionally describes bonding through electron pairs localized between atoms [46] [2]. For oxygen, the standard Lewis structure O=O fails to accurately represent the molecule because it shows all electrons paired, contradicting experimental evidence of paramagnetism [44]. Similarly, the alternative structure •O–O• suggests single-bond character inconsistent with the short bond length [44].
Modern VB theory accommodates oxygen's electronic structure through the concept of three-electron bonds, represented by Pauling using three closely spaced collinear dots [44]. In this description, each 2c-3e bond consists of two electrons in a π bonding orbital and one electron in a π* antibonding orbital, giving a net bond order contribution of 1/2 [44]. The combination of one full σ bond and two of these half-bonds yields the correct overall bond order of 2.
Table 1: Comparison of Theoretical Descriptions of Dioxygen Bonding
| Theoretical Approach | Description of Bonding | Predicted Bond Order | Accounts for Paramagnetism |
|---|---|---|---|
| Simple Lewis Theory | O=O with all electrons paired | 2 | No |
| Molecular Orbital Theory | (σ)²(σ)²(σ)²(π)⁴(π)² with unpaired electrons | 2 | Yes |
| Valence Bond (3-e⁻ bond) | One σ bond + two 2c-3e π bonds | 2 | Yes |
Historical Context: VB-MO Theory Debates
The development of quantum mechanical theories for chemical bonding involved significant competition between VB theory, championed by Linus Pauling, and MO theory, developed by Robert Mulliken and others [2]. Until the 1950s, VB theory dominated chemical thinking because it used language familiar to chemists and effectively explained molecular geometries [2]. However, MO theory gradually gained prominence due to its more straightforward explanation of paramagnetic molecules like oxygen and its implementation in computational methods [2] [43]. Modern computational chemistry has since witnessed a renaissance of VB theory, with both approaches now recognized as valuable tools for understanding molecular structure [2].
Experimental Evidence and Methodologies
Magnetic Susceptibility Measurements
A straightforward experimental demonstration of oxygen's triplet ground state involves observing its behavior in liquid form. When liquid oxygen is poured between the poles of strong magnets, it can be suspended or its stream deflected due to the net magnetic moment arising from the total electron spin S=1 [44]. This paramagnetic behavior directly reflects the presence of unpaired electrons and provides simple yet compelling evidence for the triplet state.
Spectroscopic Characterization
Spectroscopic techniques provide more quantitative evidence for oxygen's electronic structure. The energy difference between the triplet ground state (³Σ⁻g) and the first excited singlet state (¹Δg) is approximately 0.97 eV [47]. This significant energy gap explains the kinetic stability of triplet oxygen at ambient temperatures despite its thermodynamic tendency to form more stable oxidation products.
Spectroscopic observations also identify a second, higher-energy singlet state (¹Σ⁺g), though this is less relevant to most chemical and biological processes [45]. The electronic transitions between these states provide the theoretical foundation for various oxygen-sensing technologies.
Kinetic Studies of Oxygen Evolution
Recent research published in Nature (2025) has revealed how Marcus kinetics control whether singlet or triplet oxygen evolves from superoxide disproportionation [47]. This work demonstrates that the release of triplet versus singlet oxygen follows individual Marcus normal and inverted region behavior, with the crossover from predominantly triplet to predominantly singlet oxygen evolution occurring at higher driving forces [47].
The kinetic constant for superoxide oxidation can be described by: [ k{1+3} = k3 + k1 = Z{\text{el},3} \cdot e^{-\frac{(\Delta G^\circ + \lambda3)^2}{4RT\lambda3}} + Z{\text{el},1} \cdot e^{-\frac{(\Delta G^\circ + \Delta G{1\leftarrow 3}^\circ + \lambda1)^2}{4RT\lambda1}} ] where the subscripts 3 and 1 denote values for triplet and singlet oxygen, respectively [47].
Table 2: Experimental Evidence for Oxygen's Triplet State
| Experimental Method | Observation | Interpretation |
|---|---|---|
| Magnetic Susceptibility | Liquid oxygen attracted to magnetic fields | Presence of unpaired electrons (paramagnetism) |
| Spectroscopy | Energy gap of 0.97 eV between ³Σ⁻g and ¹Δg states | Triplet state is ground state; singlet is excited |
| Kinetic Studies | Marcus parabolic behavior in superoxide oxidation | Separate pathways for triplet vs. singlet O₂ evolution |
Chemical Reactivity and Biological Implications
Spin-Forbidden Reactions
The unusual electron configuration of triplet oxygen prevents it from reacting directly with many singlet-state molecules because such reactions would violate the conservation of spin angular momentum [44]. A direct reaction between triplet oxygen and a singlet molecule would require a triplet transition state, creating a significant energy barrier [44]. This explains why molecular oxygen can coexist with organic materials at ambient temperatures despite the thermodynamic favorability of oxidation reactions.
At higher temperatures or in the presence of catalysts that facilitate spin inversion, these reactions proceed more readily, as evidenced by the characteristic autoignition temperatures of flammable materials [44]. This kinetic stability, derived from the triplet ground state, is essential for aerobic life, as it prevents uncontrolled oxidation of biological molecules.
Reactive Oxygen Species in Biological Systems
The reduction of oxygen in biological systems proceeds through several reactive oxygen species (ROS), including superoxide (O₂⁻), peroxide (O₂²⁻), and singlet oxygen (¹O₂) [47]. These species play dual roles in cellular processes, acting as both damaging oxidants and important signaling molecules [47].
The spin conservation rules during superoxide disproportionation determine whether triplet or singlet oxygen forms [47]. Recent research has shown that the relative yields of these species are governed by Marcus kinetics, with implications for understanding and controlling oxidative damage in cellular systems [47].
Applications in Photodynamic Therapy
The photophysics of oxygen is exploited in photodynamic therapy (PDT) for cancer treatment [48]. In PDT, photosensitizers absorb light and transfer energy to triplet oxygen, generating singlet oxygen that destroys cancer cells [48]. Fullerenes and their derivatives, particularly open-cage fullerenes, have shown enhanced efficiency in generating reactive oxygen species via both type I (electron transfer) and type II (energy transfer) mechanisms [48].
Open-cage C₆₀ fullerenes demonstrate greater efficiency for photoinduced ROS generation than pristine C₆₀ due to increased rates of intersystem crossing and lower reduction potentials, facilitating more efficient generation of radical anions [48]. This application directly leverages the spin-state properties of molecular oxygen for therapeutic purposes.
The Scientist's Toolkit: Key Research Reagents and Methods
Table 3: Essential Reagents and Methods for Oxygen State Research
| Reagent/Method | Function/Application | Key Features |
|---|---|---|
| Tetramethylrhodamine | Fluorophore for triplet lifetime measurements | Oxygen-dependent triplet-state quenching [49] |
| Open-cage Fullerenes | Photosensitizers for ROS generation studies | Enhanced intersystem crossing; defined structures [48] |
| ESR Spin Trapping | Detection of reactive oxygen species | Identifies and quantifies specific ROS [48] |
| Marcus Kinetics Analysis | Modeling electron transfer reactions | Parabolic relationship between driving force and rate [47] |
| Triplet Imaging | Measuring oxygen consumption in cells | Based on oxygen-induced triplet lifetime changes [49] |
Comparative Theoretical Performance
Computational Approaches
Modern computational chemistry employs both valence bond and molecular orbital methods to study oxygen redox chemistry. While MO-based methods (including DFT) are more commonly used for practical computations, modern VB theory provides valuable conceptual insights, particularly for understanding diradical character and excited states [2].
The Heitler-London model, though historically significant for introducing the concept of the electron-pair bond, fails to correctly predict the relative energies of singlet and triplet states in the hydrogen molecule at large internuclear distances [50]. This limitation arises from inadequate treatment of electron correlation effects in the original model [50].
Predictive Power for Reactivity
Valence bond theory with three-electron bonding concepts successfully rationalizes the kinetic persistence of triplet oxygen despite its thermodynamic reactivity [44]. The spin-forbidden nature of reactions between triplet oxygen and singlet molecules creates a kinetic barrier that explains the stability of organic materials in air at room temperature [44].
Molecular orbital theory, particularly frontier orbital analysis, provides additional insights into reaction pathways and the role of catalysts in facilitating spin inversion during oxygen reactions [43]. Both theoretical frameworks contribute complementary understanding to oxygen redox chemistry.
The triplet ground state of oxygen, explained through the framework of three-electron π-bonding in modern valence bond theory, represents a fascinating example of how electronic structure dictates chemical behavior. While molecular orbital theory provides a more straightforward explanation for oxygen's paramagnetism, valence bond theory offers valuable conceptual insights into the nature of the oxygen-oxygen bond and the kinetic stability of molecular oxygen.
Recent advances in understanding Marcus kinetics control of singlet versus triplet oxygen evolution from superoxide have opened new avenues for controlling oxygen redox chemistry in biological systems, energy storage technologies, and therapeutic applications. The continued development of both theoretical and experimental approaches ensures that oxygen, despite being one of the most extensively studied molecules in chemistry, continues to reveal new surprises and applications.
The photoelectron (PE) spectrum of methane has long served as a critical experimental benchmark challenging the simplistic sp³ hybridization model of chemical bonding. This guide objectively compares the explanatory power of simple hybridization, molecular orbital (MO) theory, and modern valence bond (VB) theory in interpreting this spectrum. The data reveal that while the hybrid orbital model predicts four equivalent bonds, the experimentally observed two-peak structure with 3:1 intensity ratio finds a more natural explanation in delocalized orbital descriptions. Modern VB theory, employing resonance among multiple bond-pair structures, successfully reconciles the experimental data with a localized bonding picture, demonstrating its continued relevance in quantum chemistry.
The photoelectron spectrum of methane provides a pivotal test case for evaluating theoretical bonding models. When methane molecules are irradiated with ultraviolet light, the one-photon process ejects electrons from molecular orbitals, creating molecular cations [51]. The kinetic energies of these ejected electrons are measured to produce the photoelectron spectrum, which directly probes the electronic energy levels of the molecule.
For decades, the sp³ hybridization model has dominated pedagogical descriptions of methane's bonding, proposing four equivalent carbon-hydrogen bonds arranged tetrahedrally. This model successfully predicts molecular geometry but faces a significant challenge: it implies that all bonding electrons occupy identical energy environments, which should yield a single ionization peak in the photoelectron spectrum (possibly with vibrational fine structure) [52]. Experimental evidence, however, contradicts this prediction.
Experimental Data: The Photoelectron Spectrum of Methane
Key Spectral Features
The low-energy portion of methane's photoelectron spectrum, corresponding to ionization from valence bonding orbitals, displays two distinct peaks with the following characteristics:
Table 1: Experimental Photoelectron Spectral Data for Methane
| Spectral Feature | Ionization Energy | Relative Intensity | Proposed Origin |
|---|---|---|---|
| Peak 1 | Lower binding energy | 3 | Triply degenerate T₂ orbital |
| Peak 2 | Higher binding energy | 1 | Singly degenerate A₁ orbital |
The observed 3:1 intensity ratio provides crucial quantitative evidence that the bonding electrons in methane occupy two distinct energy environments rather than four equivalent orbitals [51] [52]. This intensity ratio reflects the degeneracy of the originating molecular orbitals—three electrons from one set and one from another.
Experimental Methodology
Photoelectron Spectroscopy Protocol:
- Photon Source: Ultraviolet radiation with sufficient energy to eject valence electrons (typically He(I) radiation at 21.2 eV)
- Target Species: Gaseous methane molecules at low pressure
- Detection Method: Measurement of kinetic energies of photoejected electrons
- Energy Calibration: Reference to known ionization standards
- Data Processing: Conversion of electron kinetic energies to ionization potentials via the relationship: IE = hν - KE, where IE is ionization energy, hν is photon energy, and KE is electron kinetic energy
The experiment measures the one-photon ionization process: CH₄ + hν → [CH₄]⁺ + e⁻, with the spectrum reflecting the electronic structure of neutral methane ground state through the "frozen orbital" approximation, rather than the geometry-relaxed cation [51].
Theoretical Models: Comparative Analysis
Simple Hybridization Model
The sp³ hybridization model proposes that carbon mixes its one 2s and three 2p orbitals to form four equivalent orbitals with 25% s-character and 75% p-character, directed toward the corners of a regular tetrahedron.
Prediction: Four equivalent bonding environments → Single ionization peak in PE spectrum.
Contradiction with Experiment: The observed two-peak structure with 3:1 intensity ratio directly contradicts this prediction, revealing a fundamental limitation of the simple hybridization model [52].
Molecular Orbital Theory Approach
Molecular orbital theory, combined with group theory, provides an alternative description that successfully predicts the observed spectrum:
Table 2: Molecular Orbital Description of Methane Bonding
| Orbital Symmetry | Degeneracy | Energy Order | Spectral Contribution |
|---|---|---|---|
| T₂ | Triply degenerate | Higher energy (less stable) | Peak with relative intensity 3 |
| A₁ | Singly degenerate | Lower energy (more stable) | Peak with relative intensity 1 |
In the Td point group of methane, the maximum degeneracy is three (a T representation). The bonding molecular orbitals formed from the carbon 2s and 2p orbitals and hydrogen 1s orbitals transform as the A₁ and T₂ irreducible representations, naturally explaining the observed spectral features [52]. The four equivalent C-H bonds emerge from the combination of these symmetry-adapted molecular orbitals, though the individual orbitals differ in energy and symmetry.
Modern Valence Bond Theory Resolution
Modern valence bond theory reconciles the photoelectron spectrum with a localized bonding picture through resonance theory. Rather than invoking delocalized molecular orbitals, VB theory describes the ionized states ([CH₄]⁺) as resonance hybrids among four structures, each attributing the ejected electron to a different sp³ hybrid orbital [51] [8].
Key VB Insight: A linear combination of these four resonance structures, conserving the number of structures, leads to a triply degenerate T₂ state and an A₁ state [51]. The energy difference between each ionized state and the ground state corresponds to the ionization energies observed in the photoelectron spectrum.
Mathematical Representation: The VB wavefunction for methane cation can be represented as: ΨVB = ΣcᵢΦᵢ where Φᵢ represents different resonance structures with the hole (missing electron) localized on different C-H bonds, and cᵢ are coefficients determined by variational calculation.
Comparative Theoretical Framework
Relationship Between VB and MO Theories
Modern computational chemistry reveals that MO and VB theories are related by a unitary transformation and, at equivalent levels of theory, describe the same wavefunction in different forms [8]. For the hydrogen molecule, the relationship can be expressed as:
MO Wavefunction: ΦMO = (|ab̄| - |āb|) + (|aā| + |bb̄|)
VB Wavefunction: ΦVBT = λ(|ab̄| - |āb|) + μ(|aā| + |bb̄|)
where the MO description represents a special case of VB with equal covalent and ionic contributions (λ = μ) [8]. This mathematical relationship extends to more complex molecules like methane, explaining why both approaches ultimately converge when properly implemented.
Diagram: Theoretical Relationship and Experimental Workflow
Theoretical Approaches to Methane PE Spectrum
Research Reagents and Instrumentation
Table 3: Essential Research Reagents and Materials for Photoelectron Spectroscopy
| Item | Function/Specification | Application Context |
|---|---|---|
| Ultra-pure Methane Gas | ≥99.995% purity, minimal impurities | Sample material for photoelectron spectroscopy |
| Helium I Radiation Source | 21.22 eV photon energy | UV photon source for ejecting valence electrons |
| Electron Energy Analyzer | High resolution (≤10 meV) | Measurement of photoelectron kinetic energies |
| High Vacuum System | Pressure ≤10⁻⁸ torr | Mean free path preservation for ejected electrons |
| Calibration Reference | Inert gas with known ionization energies | Energy scale calibration (e.g., Ar, Xe) |
| Computational Software | Ab initio quantum chemistry packages | Theoretical calculation of ionization energies |
The photoelectron spectrum of methane serves as a critical benchmark demonstrating that simple hybridization models, while pedagogically useful for predicting molecular geometry, provide an incomplete description of electronic structure. The experimentally observed 3:1 peak intensity ratio finds natural explanation in both molecular orbital theory (through symmetry-adapted delocalized orbitals) and modern valence bond theory (through resonance among localized bond structures). Contemporary quantum chemistry reveals that these apparently competing theories represent complementary mathematical descriptions of the same physical reality, related by unitary transformations. For researchers investigating molecular electronic structure, this case study emphasizes the importance of employing multiple theoretical frameworks and validating models against spectroscopic evidence, particularly when designing catalysts for methane conversion or developing novel bonding descriptions for complex molecular systems.
This guide provides a comparative analysis of Valence Bond (VB) theory and Molecular Orbital (MO) theory, focusing on their performance in describing homolytic bond dissociation and calculating bond dissociation energies (BDEs). For researchers and drug development professionals, accurately predicting bond strength and radical formation pathways is crucial for designing stable molecular architectures and understanding degradation mechanisms. The data and methodologies presented herein demonstrate that modern VB methods offer a more physically intuitive and accurate description of bond cleavage processes, particularly for homolytic reactions, compared to standard MO approaches.
The accurate description of chemical bond formation and dissociation is a cornerstone of molecular design. Two primary theoretical frameworks have emerged: Valence Bond (VB) Theory and Molecular Orbital (MO) Theory. While both apply quantum mechanics to explain chemical bonding, their fundamental approaches and strengths differ significantly.
Valence Bond Theory has its roots in the work of Heitler and London, who in 1927 provided the first quantum mechanical description of the covalent bond in the hydrogen molecule. [7] Linus Pauling later developed these ideas, incorporating the concepts of resonance and orbital hybridization. [53] [7] VB theory describes a covalent bond as the overlap of half-filled valence atomic orbitals from adjacent atoms, resulting in an electron pair localized between them. [7] An essential aspect of VB theory is the condition of maximum overlap, which leads to the formation of the strongest possible bonds. [53] [7]
Molecular Orbital Theory, introduced by Mulliken and Hund, offers an alternative approach where electrons are placed in delocalized orbitals that can extend over the entire molecule. [7] [54]
A key historical critique is that Pauling's powerful advocacy for VB theory "set it back fifteen years" by temporarily hindering the adoption of MO theory. [54] However, modern developments have led to a resurgence of VB methods, which now compete effectively with MO-based calculations, especially for modeling chemical reactions. [7] [55]
Comparative Analysis: VB vs. MO in Bond Dissociation
The core difference between the two theories becomes most apparent when modeling the breaking of a chemical bond.
Describing Homolytic Cleavage
Homolytic cleavage is a bond-breaking process where the electron pair is split evenly, with each fragment retaining one electron. This process generates highly reactive radical intermediates. [56]
- VB Description: VB theory correctly predicts the homolytic dissociation of homonuclear diatomic molecules into separate atoms. Its wavefunction is built from a combination of covalent and ionic structures, providing a natural and intuitive picture of the bond separation into neutral atoms. [7]
- MO Description: Simple MO theory provides a less accurate description of this process. For example, the MO wavefunction for dihydrogen is an equal mixture of covalent and ionic structures, leading to the incorrect prediction that the molecule would dissociate into a mixture of hydrogen atoms and hydrogen ions (H⁺ and H⁻). [7]
The Bond Dissociation Energy (BDE) Metric
The bond dissociation energy (BDE) is a critical quantitative measure for this discussion. It is defined as the standard enthalpy change when a bond A−B is cleaved by homolysis to give two radical fragments, A• and B•. [57] BDEs are a key indicator of bond strength and are vital for predicting the stability of molecules and the feasibility of radical reactions in both synthesis and degradation pathways. [58]
Table 1: Representative Bond-Dissociation Enthalpies (DH°₂₉₈)
| Bond | Molecule | Bond-Dissociation Enthalpy (kcal/mol) | Bond-Dissociation Enthalpy (kJ/mol) |
|---|---|---|---|
| H-H | Hydrogen | 104.2 | 435.8 |
| C-C | Typical Alkane | 83-90 | 347-377 |
| C-F | CH₃F | 115 | 481 |
| O-H | Water | 119 | 497 |
| F-F | Fluorine | 37 | 155 |
Experimental and Computational Methodologies
Computational Protocols for BDE Determination
Computing BDEs is a relatively straightforward process that involves calculating the energy difference between a parent molecule and its radical fragments. [58] The following workflow, as implemented by Rowan Scientific, outlines established methods for computing BDEs.
Diagram Title: Computational Workflow for BDE Calculation
The accuracy of this process depends on the computational methods used. The table below summarizes different workflow modes, balancing speed and precision. [58]
Table 2: Computational Workflow Modes for BDE Calculation
| Mode | Optimization Method | Single-Point Energy Method | Optimize Fragments? |
|---|---|---|---|
| Reckless | GFN-FF | GFN2-xTB | No |
| Rapid | GFN2-xTB | r²SCAN-3c | Yes |
| Careful | r²SCAN-3c | ωB97X-3c | Yes |
| Meticulous | ωB97X-3c | ωB97M-D3(BJ)/def2-TZVPPD | Yes |
Modern Valence Bond Methods
Modern VB theory has overcome early limitations by incorporating electron correlation more effectively. The Breathing-Orbital Valence Bond (BOVB) method is a key advancement. [55]
- Principle: BOVB allows each Lewis structure (covalent, ionic) in the VB wavefunction to have its own specific set of orbitals. These orbitals can "breathe"—change in size and shape—to instantaneously adapt to the electron fluctuations inherent in bond formation and breaking. [55]
- Advantage: This incorporates dynamic correlation energy, leading to accurate bond dissociation energies while retaining the intuitive, compact wavefunction of classical VB theory. The BOVB method has been successfully applied to two-electron bonds, odd-electron bonds, and resonance energies, yielding results that compare well with high-level experimental and computational data. [55]
The Scientist's Toolkit: Essential Research Reagents and Materials
The following table details key reagents and computational tools referenced in the studies cited, which are essential for experimental and theoretical investigations into homolytic bond cleavage.
Table 3: Key Research Reagent Solutions
| Reagent / Tool | Function / Application | Example / Note |
|---|---|---|
| Diboron(4) Compounds | Source of boryl radicals via homolytic B-B bond cleavage. [59] | B₂pin₂ (bis(pinacolato)diboron) used in metal-free borylation of aryl halides. [59] |
| N-Heterocyclic Carbenes (NHCs) | Facilitate homolytic cleavage of bonds and stabilize radical intermediates. [59] | Me₂ImMe used to induce radical formation from B₂pin₂. [59] |
| Computational Workflows | Calculate BDEs and model reaction pathways for molecular design. [58] | Rowan Scientific's workflows (Rapid, Careful, Meticulous modes) for predicting bond stability. [58] |
| Radical Traps | Experimental detection and verification of radical species. | TEMPO used to trap aryl radicals, providing evidence for a radical mechanism. [59] |
For researchers and drug development professionals requiring a precise understanding of bond strength and radical-mediated processes, the choice of theoretical model is critical. The evidence from homolytic bond dissociation reveals the clear superiority of the Valence Bond approach. While MO theory is a powerful and widely used framework, VB theory, particularly in its modern implementations like BOVB, provides a more physically intuitive and chemically accurate description of bond cleavage into neutral radicals. This makes VB theory an indispensable tool for the rational design of robust molecules, from pharmaceuticals to materials, where predicting and preventing radical-based degradation is paramount.
Modern Valence Bond (VB) theory represents a sophisticated computational approach that describes molecular electronic structures through linear combinations of specific bonding patterns, most notably covalent and ionic structures [8]. This theoretical framework has its direct origins in the pioneering 1927 work of Walter Heitler and Fritz London, who provided the first quantum mechanical treatment of the chemical bond in the hydrogen molecule [3]. The Heitler-London approach fundamentally demonstrated that the stability of the chemical bond arises from a quantum resonance phenomenon—a discovery that forged an inseparable link between classical chemistry and quantum physics [3].
The core innovation of the Heitler-London method was its conceptualization of the covalent bond as an electron-pair bond formed through the overlap of individual atomic orbitals, allowing two electrons to be shared between atoms [2] [3]. This perspective contrasted sharply with the emerging molecular orbital theory, preserving a more direct connection to the chemical intuition of bonds between specific atom pairs. Linus Pauling later dramatically expanded this foundation through his resonance theory, which systematically incorporated ionic structures to accurately model bond polarity and molecular stability [2]. Within this framework, the optimization challenge becomes determining the optimal weighting coefficients for covalent versus ionic contributions to achieve the most accurate representation of the true molecular wavefunction.
Theoretical Framework: Covalent and Ionic Wavefunction Structures
Fundamental Wavefunction Components
In Valence Bond Theory, the complete wavefunction is constructed from distinct structural components that represent different electron distribution patterns. For a fundamental system like the hydrogen molecule, three primary wavefunction structures form the building blocks of the complete description.
Covalent Structure (Heitler-London): This component, denoted as ΦHL, represents the pure covalent bonding situation where electrons are perfectly shared between two atoms. Mathematically, it is expressed as ΦHL = |ab̄| - |āb|, where a and b are atomic orbitals on the two hydrogen atoms, and the overbar indicates beta spin [8]. This wavefunction describes two electrons with paired spins shared between the atoms without any charge separation.
Ionic Structures: These components represent situations where both electrons reside on a single atom, creating charge-separated species. The ionic wavefunction is expressed as ΦI = |aā| + |b̄b| [8]. For H₂, this corresponds to the limiting forms H⁺H⁻ and H⁻H⁺, representing the two possible charge distributions where one atom bears both electrons.
Triplet Structure: A third, repulsive component ΦT = |ab̄| + |āb| describes a triplet state where electron spins are parallel [8]. This configuration is highly energetically unfavorable for bond formation and typically makes negligible contribution to the ground state wavefunction.
The Complete Valence Bond Wavefunction
The complete VB wavefunction synthesizes these components through a linear combination: ΦVBT = λΦHL + μΦI, where λ and μ are variational coefficients that determine the relative weighting of covalent versus ionic contributions [8]. The optimization process involves varying these coefficients to minimize the total energy of the system. In the specific case of H₂, the optimal coefficients are approximately λ ≈ 0.75 and μ ≈ 0.25, indicating a predominantly covalent bond with significant ionic character [8]. This quantitative balance demonstrates that even in seemingly purely covalent bonds like H₂, ionic contributions play an essential role in accurate wavefunction description.
Table: Wavefunction Components in Valence Bond Theory
| Component Type | Mathematical Representation | Physical Significance | Relative Weight in H₂ |
|---|---|---|---|
| Covalent (Heitler-London) | ΦHL = |ab̄| - |āb| | Perfect electron sharing | ~75% |
| Ionic | ΦI = |aā| + |b̄b| | Charge separation | ~25% |
| Triplet | ΦT = |ab̄| + |āb| | Repulsive parallel spins | Negligible |
Optimization Strategies for Wavefunction Balance
Computational Optimization Approaches
Modern computational methods employ sophisticated algorithms to determine the optimal balance between covalent and ionic structures in valence bond wavefunctions. These optimization strategies have evolved significantly from early manual calculations to contemporary automated approaches.
Variational Energy Minimization: The fundamental approach involves treating the coefficients λ and μ as variational parameters that are systematically adjusted to minimize the total energy of the system [8]. The energy is calculated as the expectation value of the Hamiltonian operator, E = ⟨ΦVBT|Ĥ|ΦVBT⟩/⟨ΦVBT|ΦVBT⟩, and minimum energy corresponds to the most accurate wavefunction.
Configuration Interaction Expansion: For complex molecules, multiple covalent and ionic structures contribute to the complete wavefunction, requiring a generalized form ΦVBT = ΣciΦi, where ci are the determined coefficients [8]. Modern VB implementations use efficient algorithms to handle these multi-structure optimizations.
Coulson-Fischer Delocalization: Advanced VB methods incorporate partially delocalized orbitals rather than strictly atomic orbitals, allowing for more efficient representation of electron distribution and reducing the number of ionic structures required for accurate description [8].
Basis Set Selection and Optimization
The choice of basis functions significantly impacts the efficiency and accuracy of VB calculations involving covalent-ionic balance.
Atomic Orbital Basis: Traditional VB uses atomic orbitals (e.g., 1s, 2p, 3d) centered on constituent atoms as basis functions [8]. While chemically intuitive, this approach often requires more ionic structures to achieve accuracy.
Delocalized Atomic Orbitals: Coulson-Fischer theory employs optimized orbitals that are slightly delocalized across multiple atoms, providing more compact wavefunction representations with fewer terms [8].
Fragment Molecular Orbitals: For large systems, using pre-computed molecular orbitals of molecular fragments as basis functions can improve computational efficiency while maintaining accuracy [8].
Table: Optimization Methods for Covalent-Ionic Balance
| Method | Key Features | Computational Cost | Accuracy |
|---|---|---|---|
| Pure Heitler-London | Only covalent terms; no optimization | Low | Poor for polar bonds |
| Single-Structure VB | λ and μ optimized for minimal set | Medium | Good for simple diatomics |
| Multi-Structure VB | Multiple covalent/ionic structures | High | Excellent for complex systems |
| VB with Delocalized Orbitals | Fewer structures needed | Medium-High | Excellent with proper optimization |
Comparative Analysis: Valence Bond vs. Molecular Orbital Approaches
Fundamental Differences in Wavefunction Construction
The valence bond and molecular orbital approaches represent two distinct yet mathematically related strategies for constructing molecular wavefunctions, with significant implications for how covalent and ionic character is represented.
Conceptual Foundations: Valence Bond Theory builds the molecular wavefunction from localized bonds between specific atom pairs, directly extending the chemist's intuition of electron-pair bonds [8]. In contrast, Molecular Orbital Theory constructs the wavefunction from delocalized orbitals that extend over the entire molecule, emphasizing the molecular entity as a whole rather than individual bonds [2].
Mathematical Relationship: Despite their different conceptual frameworks, VB and MO theories are related by a unitary transformation at equivalent levels of theory [8]. This mathematical relationship ensures they can describe the same physical reality through different representations.
Covalent-Ionic Representation: In VB theory, the balance between covalent and ionic character is explicitly represented through different resonance structures with optimized weights [8]. MO theory represents this balance implicitly through orbital occupation patterns and configuration interaction.
Performance in Specific Chemical Systems
Both theoretical approaches have distinct strengths and limitations when applied to different chemical systems, particularly regarding their handling of covalent-ionic balance.
Diatomic Molecules: For H₂, simple MO theory (without configuration interaction) predicts equal covalent and ionic contributions (λ = μ = 1), which remains fixed regardless of bond distance [8]. VB theory correctly describes the dissociation limit and allows for distance-dependent optimization of the covalent-ionic balance.
Organic π-Systems: MO theory traditionally excelled at describing delocalized systems like benzene, where early VB implementations required multiple resonance structures [8]. Modern VB methods with optimized structures now achieve comparable accuracy with compact wavefunctions.
Open-Shell Systems: VB theory provides more intuitive description of diradicals and open-shell systems, where the balance between covalent and ionic structures directly correlates with spin coupling patterns [8].
Table: Comparative Analysis of VB vs. MO Theory
| Feature | Valence Bond Theory | Molecular Orbital Theory |
|---|---|---|
| Bond Description | Localized electron pairs between atoms | Delocalized orbitals over entire molecule |
| Covalent-Ionic Balance | Explicit through resonance structures | Implicit through orbital coefficients |
| Wavefunction Form | Linear combination of VB structures | Single Slater determinant (basic) or CI expansion |
| H₂ Wavefunction | ΦVBT = λ(\|ab̄\|-\|āb\|) + μ(\|aā\|+\|b̄b\|) | ΦMOT = \|σσ̄\| = (\|ab̄\|-\|āb\|) + (\|aā\|+\|b̄b\|) |
| Dissociation Behavior | Correctly separates to neutral atoms | Incorrectly yields 50% ionic character at dissociation (basic MO) |
| Chemical Intuitiveness | High - aligns with traditional bond concepts | Lower - requires reinterpretation for chemical concepts |
Experimental Protocols for Valence Bond Analysis
Computational Implementation Methodology
Modern valence bond calculations follow specific methodological protocols to optimize the balance between covalent and ionic structures in molecular wavefunctions.
Diagram: Valence Bond Calculation Workflow
The computational protocol begins with molecular geometry specification and basis set selection, typically using atomic orbitals or delocalized basis functions [8]. Researchers then enumerate all significant resonance structures, including both covalent and ionic forms, with initial weighting coefficients. The algorithm constructs the Hamiltonian matrix and computes the total energy expectation value, then iteratively adjusts the coefficients to minimize energy until convergence criteria are satisfied.
Benchmarking and Validation Procedures
Validation of optimized valence bond wavefunctions requires comparison against both experimental data and high-level theoretical calculations.
Energy Comparison: The optimized VB energy is compared against experimental formation energies and high-level coupled-cluster calculations to validate accuracy [8].
Property Prediction: The wavefunction is used to compute molecular properties including dipole moments, bond lengths, and vibrational frequencies for comparison with experimental measurements.
Wavefunction Analysis: Advanced analysis techniques quantify the bond orders, electron densities, and covalent-ionic character for chemical interpretation.
Research Reagent Solutions for Valence Bond Studies
Table: Essential Computational Tools for Valence Bond Research
| Tool Category | Specific Examples | Function in VB Research |
|---|---|---|
| VB Software Packages | XMVB, TURTLE, CRUNCH | Specialized VB computation with structure optimization |
| Electronic Structure Suites | GAMESS, Gaussian, Q-Chem | Implement VB methods alongside other quantum approaches |
| Basis Set Libraries | cc-pVDZ, cc-pVTZ, aug-cc-pVXZ | Provide atomic orbital basis for VB expansions |
| Wavefunction Analysis | AIM, NBO, MULTIWFN | Analyze optimized VB wavefunctions and bonding character |
| Molecular Visualization | VMD, GaussView, ChemCraft | Visualize VB structures and electron distributions |
The optimization of covalent and ionic structures in valence bond wavefunctions remains an active research area with continuing developments. Modern valence bond methods have overcome historical limitations through improved computational algorithms that efficiently handle the multi-structure optimization problem [8]. The unique strength of VB theory lies in its direct chemical interpretability—the optimized coefficients λ and μ provide quantitative measures of bond character that align with chemical intuition.
Future directions include the development of more efficient optimization algorithms for large systems, improved basis sets specifically optimized for VB calculations, and more sophisticated approaches for automating resonance structure selection. The integration of machine learning techniques for preliminary structure screening shows particular promise for accelerating VB computations while maintaining the rigorous theoretical foundation established by Heitler and London nearly a century ago.
As computational power continues to grow and algorithms become more refined, valence bond theory with optimized covalent-ionic balance is poised to provide increasingly accurate descriptions of molecular structure and reactivity while maintaining its fundamental connection to the chemical concept of the electron-pair bond.
VB vs. MO: A Rigorous Comparative Analysis of Two Quantum Chemical Frameworks
The valence bond (VB) and molecular orbital (MO) theories emerged as the two foundational quantum mechanical frameworks for describing the chemical bond, originating from the pioneering work of Heitler-London and Hund-Mulliken, respectively, in the late 1920s [2]. For decades, these theories were perceived as competing descriptions of chemical bonding, with VB theory dominating the chemical literature until the 1950s before being largely eclipsed by the more computationally tractable MO theory [2]. This historical competition created the persistent misconception that VB and MO theories represent fundamentally different physical descriptions of molecular systems. However, modern quantum chemistry has revealed that at equivalent levels of theory, these approaches are mathematically equivalent—connected by a unitary transformation—and differ primarily in their conceptual representation of the electronic wavefunction [8].
The core distinction lies in their initial construction of the wavefunction. VB theory builds the wavefunction from valence bond structures that maintain atomic identity, describing the electronic wavefunction as a linear combination of several valence bond structures, each representing chemically intuitive bonding patterns [8]. In contrast, MO theory begins with delocalized molecular orbitals that extend over the entire molecule, constructing the wavefunction from a linear combination of basis functions centered on the various atoms in a species [8]. Despite these different starting points, both theories ultimately converge toward the same exact wavefunction when systematically improved, revealing their fundamental mathematical connection.
Theoretical Foundations: VB and MO Wavefunctions
Valence Bond Theory Framework
The modern valence bond theory approach has its roots in the seminal 1927 paper by Heitler and London, which provided the first quantum mechanical treatment of the hydrogen molecule [60]. In this framework, the electronic wavefunction is expressed as a linear combination of VB structures, each describable using linear combinations of atomic orbitals, delocalized atomic orbitals, or even molecular orbital fragments [8].
For the hydrogen molecule, the simplest VB wavefunction combines covalent and ionic structures:
[ \Phi_{VBT} = \lambda(\left\vert a\overline{b}\right\vert - \left\vert \overline{a}b\right\vert) + \mu(\left\vert a\overline{a}\right\vert + \left\vert b\overline{b}\right\vert) ]
where λ and μ are coefficients determining the relative contributions of covalent (first term) and ionic (second term) structures [8]. For H₂, λ ≈ 0.75 and μ ≈ 0.25, indicating the predominantly covalent character of the bond [8].
Molecular Orbital Theory Framework
The molecular orbital approach, developed by Hund and Mulliken, constructs molecular orbitals as linear combinations of atomic orbitals (LCAO) [8]. For H₂, the bonding (σ) and antibonding (σ*) molecular orbitals are formed:
[ \sigma = a + b ] [ \sigma^* = a - b ]
The ground state wavefunction in MO theory is represented as a Slater determinant with the σ orbital doubly occupied:
[ \Phi_{MOT} = \left\vert \sigma\overline{\sigma}\right\vert ]
This molecular orbital wavefunction can be expanded to reveal its relationship to the VB description [8].
The Unitary Transformation: Connecting VB and MO Theories
Mathematical Equivalence
The fundamental connection between VB and MO theories lies in the unitary transformation that relates their wavefunction representations. As explicitly demonstrated by expanding the MO wavefunction for H₂:
[ \Phi_{MOT} = (\left\vert a\overline{b}\right\vert - \left\vert \overline{a}b\right\vert) + (\left\vert a\overline{a}\right\vert + \left\vert b\overline{b}\right\vert) ]
This expansion reveals that the simple MO wavefunction implicitly contains both covalent and ionic terms in equal proportions [8]. Comparing this with the VB wavefunction:
[ \Phi{VBT} = \lambda(\left\vert a\overline{b}\right\vert - \left\vert \overline{a}b\right\vert) + \mu(\left\vert a\overline{a}\right\vert + \left\vert b\overline{b}\right\vert) ] [ \Phi{MOT} = (\left\vert a\overline{b}\right\vert - \left\vert \overline{a}b\right\vert) + (\left\vert a\overline{a}\right\vert + \left\vert b\overline{b}\right\vert) ]
Clearly demonstrates that the simplest MO description is equivalent to a VB wavefunction with fixed coefficients (λ = μ = 1), rather than variationally optimized values [8]. This mathematical relationship extends to more complex molecules through unitary transformations that localize the delocalized molecular orbitals into bond-oriented functions [61].
Theoretical Implications and Practical Consequences
The unitary transformation connecting VB and MO theories ensures that at the same level of theory, they describe the same wavefunction, albeit in different mathematical forms [8]. This mathematical equivalence, however, manifests differently in practical applications:
- Computational Tractability: MO theory with orthogonal orbitals proved more amenable to computational implementation, contributing significantly to its dominance in quantum chemistry [8] [61]
- Chemical Intuition: VB theory more directly connects to classical chemical concepts like hybridization and Lewis electron-pair theory [61]
- Interpretive Frameworks: Both theories can be extended to higher accuracy—VB through multi-structure approaches, MO through configuration interaction—with both converging to the exact solution [8]
Comparative Analysis: Key Differentiators and Convergence
Table 1: Fundamental Characteristics of VB and MO Theories
| Feature | Valence Bond Theory | Molecular Orbital Theory |
|---|---|---|
| Fundamental Approach | Localized bonds from atomic orbitals | Delocalized molecular orbitals from LCAO |
| Wavefunction Form | Linear combination of VB structures [8] | Slater determinant of MOs [8] |
| Initial Description of H₂ | Covalent-ionic superposition [8] | Doubly-occupied bonding orbital [8] |
| Bond Dissociation | Correctly describes separation to atoms | Simple description fails without CI [8] |
| Computational Implementation | Historically challenging due to non-orthogonal basis [61] | More computationally tractable [8] |
| Connection to Chemical Concepts | Direct linkage to Lewis structures and hybridization [61] | More abstract connection to chemical intuition |
Table 2: Wavefunction Composition for H₂ at Different Theory Levels
| Theory Level | Covalent Contribution | Ionic Contribution | Description |
|---|---|---|---|
| Simple VBT | λ ≈ 0.75 | μ ≈ 0.25 | Variationally optimized [8] |
| Simple MOT | 1.0 | 1.0 | Equal contributions [8] |
| MO-CI | Variable | Variable | Matches VBT at completion [8] |
| Exact | λ (exact) | μ (exact) | Identical regardless of path |
The perceived "failures" of either theory often stem from incomplete application rather than fundamental limitations. For instance, the inability of simple MO theory to correctly describe bond dissociation arises from the fixed covalent-ionic ratio in the wavefunction, which is remedied by including configuration interaction (MO-CI) [8]. Similarly, early criticisms of VB theory for failing to explain the triplet ground state of O₂ or the photoelectron spectrum of methane resulted from oversimplified applications rather than inherent theoretical deficiencies [8].
Computational Methodologies and Experimental Protocols
Modern Computational Approaches
Contemporary quantum chemistry has developed sophisticated methods for implementing both VB and MO theories at high levels of accuracy:
- Modern VB Theory: Utilizes non-orthogonal orbital bases and efficient algorithms to handle the computational complexity [8]. The generalized valence bond (GVB) wavefunction represents a special form of the multi-configurational self-consistent field (MCSCF) wavefunction [61]
- Periodic Systems: For solid-state calculations, the LOBSTER package performs bonding analysis by transforming plane-wave DFT results into an local orbital basis, enabling COOP (Crystal Orbital Overlap Population) and other bonding indicators [10]
- Wavefunction Analysis: Tools like localized molecular orbitals and Wannier functions create connections between the delocalized MO perspective and localized bonding descriptions [10]
Research Reagent Solutions: Essential Computational Tools
Table 3: Key Computational Tools for Bonding Analysis
| Tool/Method | Function | Application Context |
|---|---|---|
| LOBSTER Package | Transforms plane-wave results to local orbital basis [10] | Solid-state bonding analysis |
| Maximally Localized Wannier Functions | Generates localized orbitals from periodic calculations [10] | Solid-state systems |
| Mulliken Population Analysis | Calculates atomic charges and overlap populations [10] | Molecular systems |
| COOP Analysis | Crystal Orbital Overlap Population for solids [10] | Periodic systems |
| GVB Methods | Generalized Valence Bond calculations [61] | Molecular systems |
| Fragment Molecular Orbital Method | Analyses interactions between molecular fragments [10] | Large molecular systems |
The unitary transformation connecting valence bond and molecular orbital theories reveals their fundamental mathematical equivalence, demonstrating that they represent complementary perspectives on the same underlying quantum reality. While their historical development emphasized perceived differences and competition, modern quantum chemistry recognizes both as valuable frameworks within a unified theoretical understanding of chemical bonding.
The choice between VB and MO approaches in contemporary research depends primarily on the specific chemical question being addressed and computational considerations rather than fundamental theoretical superiority. For problems where chemical intuition and bond-localized perspectives are valuable, modern VB theory offers powerful insights, while for spectroscopic properties and delocalized systems, MO theory often provides more natural descriptions. Both theories, when properly applied at equivalent levels of sophistication, converge to the same description of molecular electronic structure, connected through the fundamental unitary transformation that reveals their underlying mathematical equivalence.
This unified perspective enriches our understanding of chemical bonding, allowing researchers to select the most appropriate conceptual and computational framework for their specific needs while recognizing the fundamental coherence of quantum chemical theory.
The hydrogen molecule (H₂) represents the simplest and most fundamental case of a covalent chemical bond, serving as the critical test system for the development of quantum chemical theories. The accurate description of its bonding mechanism posed a significant challenge in the early 20th century, ultimately leading to two complementary theoretical frameworks: the Heitler-London valence bond (VB) approach and molecular orbital (MO) theory [60] [11]. Both methodologies originate from quantum mechanics but offer distinctly different conceptual pathways to explain how two hydrogen atoms form a stable molecule. This comparison guide objectively analyzes the performance of these two dominant bonding descriptions using H₂ as the benchmark system, providing researchers with a clear understanding of their respective strengths, limitations, and computational implications within modern chemical research.
Theoretical Frameworks and Historical Development
Valence Bond Theory: The Heitler-London Approach
The valence bond approach emerged from the pioneering 1927 work of Walter Heitler and Fritz London, which provided the first successful quantum mechanical treatment of the hydrogen molecule [60] [11]. Their methodology centered on the concept of electron pairing between atomic orbitals of separate atoms. In the Heitler-London model, the wavefunction is constructed from a covalent combination of the two hydrogen 1s orbitals, effectively describing the bond as arising from the overlap and electron pair sharing between these localized orbitals [8].
The revolutionary aspect of this work was its demonstration that the covalent bond stability originates from quantum mechanical exchange effects - an energy lowering phenomenon that occurs when electron spins pair and their wavefunctions overlap. This approach naturally aligned with Gilbert N. Lewis's earlier concept of electron pair bonds and formed the foundation for Linus Pauling's extensive development of valence bond theory, including the concepts of hybridization and resonance [62]. For H₂, the Heitler-London calculation yielded a dissociation energy of 3.14 eV and a bond length of 0.87 Å, reasonably approximating the experimental values of 4.48 eV and 0.74 Å, respectively [11].
Molecular Orbital Theory: The Hund-Mulliken Approach
Molecular orbital theory, developed primarily by Friedrich Hund and Robert S. Mulliken, adopts a fundamentally different perspective by considering electrons as being delocalized throughout the entire molecule [63] [11]. Rather than focusing on electron pairs between specific atoms, MO theory constructs molecular orbitals as linear combinations of atomic orbitals (LCAO), resulting in orbitals that extend over multiple atomic centers.
For H₂, the two 1s atomic orbitals combine to form two molecular orbitals: a bonding σ orbital (with no nodal plane between nuclei) and an antibonding σ* orbital (with a nodal plane between nuclei) [63]. In the ground state, both electrons occupy the bonding orbital, resulting in a net stabilization. The initial MO treatment of H₂ was less accurate than the Heitler-London approach, but with systematic improvements including configuration interaction, it achieved comparable accuracy [11]. MO theory's strength lies in its ability to naturally describe delocalized bonding in more complex molecules and its more straightforward computational implementation.
Quantitative Comparison of Theoretical Predictions
Computational Performance for H₂
Table 1: Quantitative Comparison of VB and MO Predictions for H₂
| Parameter | Experimental Value | Heitler-London VB | Simple MO | Modern Calculations |
|---|---|---|---|---|
| Dissociation Energy (eV) | 4.48 [11] | 3.14 eV [11] | ~2.7 eV [63] | 4.48 eV (both methods) [11] |
| Bond Length (Å) | 0.74 [11] | 0.87 Å [11] | ~0.85 Å [63] | 0.74 Å (both methods) [11] |
| Bond Order | 1 | 1 [64] | 1 [64] | 1 (both methods) |
| Wavefunction Description | - | Covalent + ionic terms [8] | Doubly occupied σ orbital [63] | Equivalent at high theory [8] |
| Electron Distribution | Localized between nuclei | Localized between nuclei [65] | Delocalized molecular orbital [63] | Equivalent descriptions |
Table 2: Methodological Comparison of VB and MO Approaches
| Characteristic | Valence Bond Theory | Molecular Orbital Theory | ||
|---|---|---|---|---|
| Fundamental Unit | Electron pairs between atoms [65] | Delocalized molecular orbitals [63] | ||
| Bond Description | Localized bonds, resonance structures [8] | Orbital energy diagram, bonding/antibonding [63] | ||
| H₂ Wavefunction | ΦVBT = λΦcovalent + μΦ_ionic [8] | Φ_MOT = | σŌÆŹ | [8] |
| Computational Scaling | Historically more complex [8] | More straightforward [8] | ||
| Chemical Intuitiveness | High for localized bonds [65] | High for delocalized systems [63] | ||
| Treatment of Dissociation | Correctly separates to atoms [8] | Incorrectly gives ionic terms [8] |
Diagram: Theoretical Approaches to H₂ Bonding
Experimental Protocols and Methodologies
Key Experimental Determinations for H₂
The quantitative parameters for the H₂ molecule have been established through various experimental techniques, providing the benchmark data against which theoretical methods are evaluated. The dissociation energy of 4.48 eV (104 kcal/mol) represents the energy required to separate the molecule into two hydrogen atoms and was determined through spectroscopic measurements and thermodynamic studies [11]. The bond length of 0.74 Å was established using rotational spectroscopy and diffraction techniques, which precisely measure the average distance between the two hydrogen nuclei in the ground vibrational state [11].
Spectroscopic methods, particularly vibrational and rotational spectroscopy, have provided crucial information about the potential energy surface of H₂, including the anharmonicity of the bond vibration and the exact energy differences between quantum states. These experimental data were essential for validating the early quantum mechanical calculations of Heitler-London and James-Coolidge, with the latter achieving remarkable agreement with experimental values through extensive computation using hand-cranked mechanical calculators [11].
Computational Methodologies
Valence Bond Computational Approach
The modern valence bond approach implements the Heitler-London methodology with computational improvements that maintain the localized character of the bonding description. The wavefunction is expressed as a linear combination of valence bond structures, with the relative weights of covalent and ionic contributions determined variationally [8]. For H₂, this takes the form:
Φ_VBT = λ(|abŌÆŹ| - |āb|) + μ(|aaŌÆŹ| + |bbŌÆŹ|)
where λ and μ are coefficients optimized for energy minimization, a and b represent the 1s orbitals on the two hydrogen atoms, and the bar indicates electron spin [8]. In the case of H₂, the optimized coefficients are approximately λ ≈ 0.75 and μ ≈ 0.25, indicating predominantly covalent character with minor ionic contribution [8].
Molecular Orbital Computational Approach
The molecular orbital method employs a different strategy, constructing delocalized orbitals first and then populating them with electrons. For H₂, the σ bonding and σ* antibonding orbitals are formed as:
σ = a + b σ* = a - b
The ground state wavefunction is represented as a Slater determinant with the σ orbital doubly occupied: Φ_MOT = |σŌÆŹ| [8]. When analyzed in terms of atomic orbitals, this simple MO wavefunction contains equal covalent and ionic contributions, which represents a limitation at the simple theory level but can be corrected through configuration interaction methods that mix in excited state configurations [8].
Table 3: Essential Computational and Experimental Resources for H₂ Bonding Studies
| Resource Category | Specific Tools/Methods | Application in H₂ Research | Key Function |
|---|---|---|---|
| Computational Chemistry Software | Modern VB programs (GAMESS, VB2000) [8] | Accurate VB calculations with delocalized orbitals | Implement improved VB algorithms competitive with MO methods |
| MO-CI packages (Gaussian, GAMESS) [8] | Molecular orbital calculations with configuration interaction | Account for electron correlation in MO framework | |
| Extended Hückel methods [11] | Semi-empirical MO calculations for larger systems | Rapid geometry and energy predictions | |
| Theoretical Analysis Methods | Bond Order Analysis [64] | Quantifying bond strength: (bonding e⁻ - antibonding e⁻)/2 | Provide quantitative measure of bond multiplicity |
| Potential Energy Curve Mapping [11] | Modeling energy as function of internuclear distance | Visualize bond formation, dissociation energy, vibrational states | |
| Electron Density Analysis [41] | Topological analysis of electron distribution | Characterize bond critical points and electron localization | |
| Experimental Characterization | Rotational-Vibrational Spectroscopy [11] | Precise bond length and dissociation energy measurement | Validate theoretical predictions against experimental data |
| Photoelectron Spectroscopy [8] | Measuring molecular orbital energies | Experimental probe of electronic structure | |
| Quantum Information Approaches | Orbital Entanglement Measures [41] | Analyzing electron correlation patterns | Provide novel perspective on bond nature using QIT tools |
Modern Developments and Research Applications
Contemporary Theoretical Frameworks
Modern theoretical developments have largely reconciled the historical competition between VB and MO theories, demonstrating their fundamental equivalence at high levels of theory [8]. Through unitary transformations, a molecular orbital wavefunction can be converted into a valence bond representation, and vice versa, confirming that both are equally valid descriptions of the same physical reality [8]. This theoretical unification has been facilitated by computational advances that have made valence bond calculations more competitive with the dominant molecular orbital methods [8].
Recent approaches from quantum information theory have provided novel insights into chemical bonding, analyzing bonds through the lens of orbital entanglement patterns [41]. These methods quantify the nonlocal correlations between atomic orbitals in molecules, offering a fresh perspective on both traditional two-center bonds and multicenter bonding phenomena. For H₂, this approach recovers the classic electron-pair bond but within a more general framework that can be systematically applied to more complex bonding situations [41].
Diagram: Modern Unified View of Chemical Bonding
The side-by-side comparison of valence bond and molecular orbital theories using the H₂ molecule as a benchmark system reveals that both approaches, when implemented at appropriate levels of theory, converge to the same quantitative description of chemical bonding [8]. The historical perception of VB theory's limitations primarily resulted from computational implementation challenges rather than fundamental theoretical deficiencies [8]. For researchers in chemical and pharmaceutical development, this understanding is crucial for selecting appropriate computational methods for specific applications.
Valence bond theory offers more chemically intuitive descriptions for localized bonds and reaction mechanisms, aligning closely with traditional structural representations [65] [8]. Molecular orbital theory provides a more straightforward pathway for computational implementation and naturally describes delocalized bonding in conjugated systems and extended solids [63] [8]. The modern unification of these perspectives, complemented by emerging approaches from quantum information theory [41], provides researchers with a comprehensive toolkit for tackling complex bonding situations in drug design, materials science, and catalytic development. The H₂ case study continues to serve as the fundamental reference point for evaluating new theoretical methodologies and computational approaches in chemical bonding research.
The Heitler-London (HL) approach, introduced in 1927, and Molecular Orbital (MO) theory represent two foundational, yet philosophically distinct, quantum mechanical descriptions of the chemical bond [2] [10]. For decades, these theories have been perceived as competitors, with their struggle for dominance shaped by their respective proponents, Linus Pauling for Valence Bond (VB) theory and Robert Mulliken for MO theory [2]. This guide provides an objective comparison of modern Valence Bond theory, which builds upon the HL approach, and MO theory. Framed within contemporary research, it evaluates their performance based on conceptual intuitiveness, predictive power for molecular properties, and applicability in modern scientific fields like drug development. The resurgence of VB theory, powered by improved computational methods, allows for a fresh and balanced assessment of its strengths and weaknesses against the currently more prevalent MO theory [8] [2].
Theoretical Foundations and Historical Context
The Valence Bond theory originated directly from the pioneering work of Heitler and London on the hydrogen molecule [2] [12]. Their 1927 paper provided the first quantum-mechanical treatment of the covalent bond, describing it as an electron pair formed by the overlap of atomic orbitals from two hydrogen atoms, with the electrons correlating their spins [34] [10]. This "electron-pair" model was deeply aligned with the chemical intuition of Lewis and was later generalized by Pauling into a comprehensive theory incorporating concepts like hybridization and resonance [2] [66].
Concurrently, Mulliken and Hund developed MO theory, which took a fundamentally different approach by first constructing orbitals that are delocalized over the entire molecule [10]. In this framework, electrons are placed in these molecular orbitals and are treated as independent of each other, naturally allowing for ionic terms that the original HL model omitted [8] [10].
A key mathematical insight is that at high levels of theory, VB and MO are formally equivalent and related by a unitary transformation, meaning they can describe the same wavefunction but in different forms [8]. For instance, the simple MO description of H₂, when transformed, is equivalent to a VB wavefunction with fixed, equal covalent and ionic contributions [8]. The historical dominance of MO theory from the 1950s onward was not due to inherent superiority but largely because of its computational advantages—MO methods were easier to program for digital computers [8] [2].
Comparative Performance Analysis
Conceptual Strengths and Chemical Intuitiveness
Valence Bond theory excels in providing a conceptually intuitive picture that aligns closely with a chemist's empirical understanding.
- Localized Bonds and Resonance: VB theory describes molecules using localized two-center bonds, making it exceptionally powerful for visualizing and rationalizing molecular geometry, reaction mechanisms, and the concept of resonance between different Lewis structures [66]. This provides an "unrivaled explanatory power" for ground-state properties and organic reaction pathways [66].
- Direct Bond Description: The theory retains the classic Lewis pair bond as its central unit, offering a direct and visual representation of bonding that is easily communicated and taught [2] [66].
In contrast, Molecular Orbital theory's primary conceptual strength lies in its elegant interpretation of molecular spectra and delocalized systems.
- Spectroscopic Prediction: MO theory provides a natural framework for interpreting electronic spectra and photoelectron spectroscopy, as molecular orbitals can be directly linked to ionization energies and excited states [8] [66].
- Delocalized Systems: It seamlessly describes delocalized π-systems in aromatic molecules and conjugated polymers, as well as the bonding in extended solids, where the concept of localized bonds becomes cumbersome [8] [10].
Quantitative Accuracy and Predictive Power
The perception that VB theory is quantitatively inferior is largely a historical artifact. Modern VB implementations are competitive in accuracy with their MO counterparts.
Table 1: Quantitative Performance for the H₂ Molecule
| Theory/Method | Bond Length (Å) | Dissociation Energy (eV) | Key Feature |
|---|---|---|---|
| Heitler-London (Original) | ~0.90 [12] | ~0.25 [12] | Purely covalent, underestimates binding |
| Simple MO (Hartree-Fock) | - | - | Fixes covalent-ionic ratio at 1:1 [8] |
| MO with CI | - | - | Matches high-level VB accuracy [8] |
| Screening-Modified HL [35] | ~0.74 [35] | - | Incorporates dynamic effective nuclear charge |
Modern VB methods like the Xiamen Valence Bond (XMVB) package achieve high accuracy by moving beyond the original HL model. These methods natively include static electron correlation—a key advantage over basic MO methods—because even a single VB structure is inherently a multi-determinant wavefunction [66]. For challenging cases like the triplet ground state of O₂, a proper VB calculation correctly predicts the ground state using three-electron π-bonds, contrary to the common misconception that VB theory fails for oxygen [8].
Computational Efficiency and Application Scope
A key practical differentiator is computational cost, which has heavily influenced the adoption of these methods.
- MO Theory Efficiency: The standard Hartree-Fock method in MO theory uses a single Slater determinant with orthogonal orbitals, leading to relatively low computational scaling and making it the default in most quantum chemistry software [66].
- VB Theory Overhead: Ab initio VB methods are inherently multi-determinant, even for a single VB structure. This results in a higher computational cost, which has limited the development and availability of VB software [66].
Table 2: Computational and Applicability Comparison
| Aspect | Valence Bond (VB) Theory | Molecular Orbital (MO) Theory |
|---|---|---|
| Primary Strength | Explanatory power; intuitive for ground states & reactions [66] | Predictive power for spectra & delocalized systems [8] [66] |
| Typical Wavefunction | Multi-determinant (native static correlation) [66] | Single determinant (requires add-ons for correlation) |
| Computational Scaling | Higher (non-orthogonal orbitals) [66] | Lower (orthogonal orbitals) [8] [66] |
| O₂ Ground State | Correctly predicts triplet with 3-electron bonds [8] | Correctly predicts triplet |
| Handling of Aromatics | Requires resonance of Kekulé/Dewar structures | Natural description via delocalized π-MOs |
| Software Availability | Limited (e.g., XMVB) [66] | Widespread (e.g., Gaussian, GAMESS) |
Experimental Protocols and Methodologies
Protocol: Valence Bond Computation with XMVB
The Xiamen Valence Bond (XMVB) package is a leading tool for modern ab initio VB calculations [66]. A typical computational workflow is as follows.
Diagram 1: VB Computation Workflow
- Input Preparation: Define the molecular structure and select an atomic orbital basis set. The choice of basis functions (localized atomic orbitals, delocalized orbitals, or fragment orbitals) is flexible [8] [66].
- Structure Generation: Specify the set of valence bond structures to be included in the calculation. For a molecule like H₂, this typically includes the covalent (Heitler-London) structure and the ionic structures [8].
- VBSCF Calculation: Execute a Valence Bond Self-Consistent Field (VBSCF) calculation. This method optimizes both the coefficients of the VB structures and the orbitals within those structures simultaneously. It is physically analogous to the Multi-Configuration SCF (MCSCF) method in MO theory [66].
- Post-SCF Analysis: Analyze the wavefunction to determine the relative weights of the different VB structures. For example, in H₂, the coefficient λ for the covalent structure is larger than the coefficient μ for the ionic structures, quantifying the bond's covalency [8].
- Advanced Corrections (Optional): For higher accuracy, especially in describing bond dissociation, methods like Breathing Orbital VB (BOVB) or Valence Bond Configuration Interaction (VBCI) can be used to incorporate dynamic electron correlation [66].
Protocol: Bond Energy Calculation via Variational Quantum Monte Carlo
Recent research revisiting the HL model employs sophisticated computational methods to improve upon its original accuracy [67] [34] [35]. The following protocol outlines such an approach.
- Wavefunction Ansatz: A trial wavefunction is constructed based on the HL form, ψ±(r→₁,r→₂) = N± [ϕ(r₁ₐ)ϕ(r₂ᵦ) ± ϕ(r₁ᵦ)ϕ(r₂ₐ)], but with a critical modification: the atomic orbital ϕ(rᵢⱼ) = (1/√π) e^(-α rᵢⱼ) includes a variational parameter α that acts as an effective nuclear charge to account for electronic screening effects [34] [35].
- Variational Optimization: The Variational Quantum Monte Carlo (VQMC) method is used to compute the expectation value of the energy for a given α and internuclear distance R. The parameter α is optimized at each R to minimize the total energy [35].
- Property Calculation: The optimized wavefunction is used to calculate the potential energy curve, from which key molecular properties—bond length (Rₑ), binding energy (Dₑ), and vibrational frequency—are determined [35].
- Result: This screening-modified HL model yields results, such as a bond length of approximately 0.74 Å, that show substantially improved agreement with experimental data compared to the original HL model [35].
The Scientist's Toolkit: Essential Research Reagents
Table 3: Key Computational Tools for Bonding Analysis
| Tool/Solution | Function | Primary Theory |
|---|---|---|
| XMVB Package [66] | Specialized software for ab initio VB calculations (VBSCF, BOVB, VBCI). | Valence Bond |
| LOBSTER [10] | Performs wavefunction-based bonding analysis (population analysis, bond orders) on periodic solids. | Molecular Orbital |
| Gaussian/GAMESS | General-purpose quantum chemistry packages, predominantly featuring MO-based methods. | Molecular Orbital |
| Variational Quantum Monte Carlo (VQMC) [35] | Stochastic method to optimize and evaluate complex trial wavefunctions (e.g., modified HL). | Both |
Applications in Drug Development and Materials Science
The principles of quantum mechanics, including bonding theories, have become indispensable in pharmaceutical science, enabling researchers to understand and predict drug-target interactions at an atomic level [68].
- Enzyme Catalysis: Quantum tunneling, a phenomenon that can be probed with these theories, explains reaction rates in enzymes like soybean lipoxygenase, which exhibits a large kinetic isotope effect. Inhibitors can be designed to disrupt the optimal tunneling geometry, increasing their potency [68].
- Drug-Target Binding: Key interactions, such as hydrogen bonding (e.g., in the antibiotic vancomycin) and π-stacking, depend on quantum mechanical electron distributions. Accurate prediction of these interactions often requires hybrid QM/MM (Quantum Mechanics/Molecular Mechanics) methods, where the active site is treated quantum-mechanically [68].
- Materials Design: In solid-state chemistry, tools like LOBSTER enable bonding analysis in periodic materials, moving beyond simplistic ionic models to a nuanced understanding of covalent and ionic contributions in solids, which is crucial for designing new functional materials [10].
Valence Bond theory and Molecular Orbital theory are not mutually exclusive but are complementary frameworks for describing molecular electronic structure [8] [2]. The choice between them is a trade-off between chemical intuitiveness and predictive scope for spectroscopy and delocalization.
Modern Valence Bond theory, empowered by advanced computational methods, has overcome many of its historical quantitative limitations and remains unparalleled for providing intuitive insights into chemical bonding and reactivity [8] [66]. Its language of localized bonds and resonance is deeply embedded in chemical reasoning. Conversely, Molecular Orbital theory offers a more direct path to calculating spectroscopic properties and understanding delocalized electronic structures, maintaining its status as the more widely implemented computational workhorse [8] [66]. For the practicing scientist, the optimal approach is often a pragmatic one, leveraging the strengths of each theory to illuminate different aspects of a complex chemical system.
The field of computational chemistry is built upon three foundational theoretical frameworks: Valence Bond (VB) theory, Molecular Orbital (MO) theory, and Density Functional Theory (DFT). While MO theory and DFT currently dominate computational practices, modern valence bond theory has experienced a significant resurgence, challenging the perception of these methods as competing rather than complementary approaches [8] [69]. This guide provides an objective comparison of these methodologies within the context of modern chemical research, examining their theoretical foundations, practical performance, and synergistic applications.
The intellectual lineage of these theories traces back to the pioneering work of Heitler and London, who in 1927 published the first quantum mechanical treatment of the hydrogen molecule, establishing the fundamental principles of covalent bonding [10] [69]. This "Heitler-London" approach formed the cornerstone of what would become modern valence bond theory, introducing the concept of electron pairing as the basis for chemical bond formation [69] [70].
Theoretical Foundations and Historical Development
Valence Bond Theory: The Chemical Intuition Preserver
Valence Bond theory emerged directly from the Heitler-London description of H₂, where the wavefunction is constructed as a linear combination of covalent and ionic structures [8] [69]. The theory describes the electronic wavefunction as a linear combination of several valence bond structures, each representing an alternative pairing scheme for the electrons [8] [70]. This approach maintains a direct connection to classical chemical concepts, representing bonds as electron pairs localized between atoms.
The VB framework introduced pivotal chemical concepts including hybridization, resonance, and electronegativity through the work of Pauling, who recognized the connection between the Heitler-London wavefunction and Lewis's shared electron pairs [69]. A fundamental strength of VB theory lies in its adherence to the permutation symmetry inherent in the many-electron Hamiltonian, a feature not always preserved in MO wavefunctions [69].
Molecular Orbital Theory: The Delocalized Framework
Molecular Orbital theory, developed by Mulliken, Hund, and others, adopts a fundamentally different perspective, viewing molecules as collections of nuclei surrounded by electrons in delocalized orbitals [10] [36]. Instead of localizing electron pairs between specific atoms, MOT constructs molecular orbitals as linear combinations of atomic orbitals (LCAO) that extend over the entire molecule [8] [36].
This delocalized approach proved particularly successful in explaining spectroscopic phenomena, aromatic systems, and pericyclic reactions, contributing to its rapid adoption as the dominant theoretical framework [10]. The computational simplicity of working with orthogonal orbitals in MO theory further accelerated its popularity as digital computers became available for quantum chemical calculations [8].
Density Functional Theory: The Electron Density Alternative
Density Functional Theory represents a third paradigm, shifting focus from the complex N-electron wavefunction to the simpler electron density as the fundamental variable [10]. Originating from metal physics, DFT gained widespread adoption following the formulation of the Hohenberg-Kohn theorems and Kohn-Sham equations, which established that all ground-state properties are uniquely determined by the electron density [10] [36].
Unlike wavefunction-based methods, DFT inherently includes electron correlation through the exchange-correlation functional, offering a compelling balance between computational cost and accuracy [36]. This advantage has made DFT the predominant method for calculating electronic structures in periodic solids and large molecular systems [10].
Mathematical Relationship Between VB and MO Theories
Despite their different conceptual frameworks, VB and MO theories are fundamentally related through a unitary transformation [8]. At equivalent levels of theory, the two approaches describe the same wavefunction, merely representing it in different forms [8]. This relationship becomes evident when examining the hydrogen molecule:
- The MO wavefunction for H₂ (doubly occupied σ orbital) can be mathematically transformed into a VB-style wavefunction containing both covalent and ionic terms [8].
- Simple MO theory often overemphasizes ionic character in bonds, a deficiency corrected through configuration interaction (MO-CI), which brings the description in line with VB theory [8].
Table: Fundamental Characteristics of Computational Theories
| Theory | Fundamental Quantity | Bond Description | Key Strength |
|---|---|---|---|
| Valence Bond (VB) | Many-electron wavefunction | Localized electron pairs | Chemical intuition, permutation symmetry |
| Molecular Orbital (MO) | Delocalized orbitals | Electrons in molecular orbitals | Spectral prediction, computational simplicity |
| Density Functional (DFT) | Electron density | Implicit through density | Electron correlation, solid-state applications |
Methodological Comparison: Performance Metrics and Limitations
Computational Efficiency and Application Scope
The practical implementation of these theories reveals significant differences in computational efficiency and suitability for various chemical systems. MO-based methods, particularly Hartree-Fock with post-hoc correlation corrections, dominate molecular quantum chemistry due to their computational tractability and well-defined systematic improvement pathways [36]. Modern VB theory, however, has overcome earlier computational limitations through improved programming approaches, becoming competitive with MO methods in accuracy and economy [8].
DFT occupies a unique position, offering favorable scaling while incorporating electron correlation, making it particularly suitable for extended systems and solids [10]. This advantage explains DFT's dominance in materials science and solid-state chemistry, where plane-wave basis sets naturally accommodate periodic boundary conditions [10].
Bonding Analysis and Chemical Interpretation
For chemical bonding analysis, each method offers distinct advantages. VB theory provides a more intuitive description of bond formation and dissociation, particularly for systems where electron pairing plays a crucial role [8] [69]. MO theory excels in describing delocalized bonding, aromatic systems, and molecular spectroscopy [10] [36].
DFT's foundation on electron density presents interpretative challenges for bonding analysis, as the density lacks phase information essential for distinguishing bonding from antibonding interactions [10]. To address this limitation, computational tools like the LOBSTER package perform unitary transformations from plane-wave DFT results to local orbitals, enabling MO-style bonding analysis in solid-state systems [10].
Known Limitations and Systematic Deficiencies
Each methodology exhibits characteristic limitations that researchers must consider when selecting computational approaches:
- VB Limitations: Traditional VB implementations struggled with computational efficiency and the representation of delocalized systems, though modern approaches have largely addressed these issues [8] [69].
- MO Limitations: Standard MO methods can poorly describe bond dissociation and systems with significant static correlation, such as the C₂ molecule, which requires multi-configurational treatment [36].
- DFT Limitations: The accuracy of DFT depends critically on the exchange-correlation functional, with common functionals failing for dispersion interactions, strongly correlated systems, and transition states [36].
Table: Performance Comparison for Representative Chemical Systems
| System/Property | VB Performance | MO Performance | DFT Performance |
|---|---|---|---|
| H₂ Bond Dissociation | Excellent description | Requires CI for accuracy | Good with appropriate functionals |
| O₂ Triplet Ground State | Correctly predicts [8] | Correctly predicts [36] | Varies with functional |
| C₂ Bond Description | Multiple interpretations [36] | Requires multi-reference [36] | Challenging for standard functionals |
| Benzene Aromaticity | Resonance description | Delocalized π-system | Good structural prediction |
| Solid-State Band Structure | Limited application | Limited without periodicity | Excellent, method of choice |
Case Studies: Experimental Validation and Protocols
Dopant Engineering in Metal Oxide Photocatalysts
Recent research on Mo-doped NiFe₂O₄ photocatalysts demonstrates the complementary application of DFT and time-dependent DFT (TD-DFT) for predicting material properties before experimental synthesis [71]. The computational protocol involved:
- Model Construction: Building crystal structures of pristine and Mo-doped NiFe₂O₄ with dopant atoms substituting at Fe³⁺ octahedral sites [71].
- Electronic Structure Calculation: Using DFT to compute band structures, density of states, and charge redistribution following doping [71].
- Excited-State Modeling: Applying TD-DFT to simulate electronic excitations and predict interactions with H₂O₂ for hydroxyl radical generation [71].
- Property Prediction: Calculating flat band potentials, semiconductor type, and charge transfer resistance to forecast photocatalytic efficiency [71].
Experimental validation confirmed that Mo-doped NiFe₂O₄ nanoparticles exhibited enhanced photo-Fenton degradation of tetracycline, with performance metrics aligning with computational predictions [71]. This integrated approach demonstrates DFT's predictive capability for guiding materials design, particularly in optimizing dopant selection for specific applications.
Dual-Doping Strategy in BiVO₄ Photoanodes
A sophisticated dual-doping study on BiVO₄/FeCoNiOx photoanodes for water splitting employed DFT calculations to decipher the synergistic effects of Mo doping in both the semiconductor bulk and cocatalyst surface [72]. The methodology encompassed:
- Bulk Doping Analysis: Modeling Mo substitution at V sites in BiVO₄ to confirm n-type doping behavior and enhanced charge transport [72].
- Surface Doping Investigation: Simulating Mo incorporation in FeCoNiOx to examine electron redistribution and creation of localized electron reservoirs [72].
- Interface Modeling: Computing charge transfer barriers at the Mo:FeCoNiOx/Mo:BiVO₄ interface to understand enhanced hole extraction [72].
- Reaction Mechanism Elucidation: Calculating reaction energy profiles for oxygen evolution, revealing a reduced ΔG*OH barrier of 1.26 eV on reconfigured Fe sites [72].
The computationally guided design yielded a photoanode achieving 7.15 mA cm⁻² at 1.23 V versus RHE, approaching the theoretical maximum for BiVO₄, with experimental measurements confirming enhanced carrier separation efficiency (84.9% to 96.5%) and 2.7-fold faster hole migration [72].
Strain Engineering in Perovskite Materials
DFT's strength in property prediction for materials under external perturbations is exemplified by a study on strained APbBr₃ (A = K, Rb, Cs) perovskites [73]. The research protocol included:
- Structure Optimization: Determining optimum lattice constants using the GGA-PBE exchange-correlation functional with ultrasoft pseudopotentials [73].
- Controlled Strain Application: Systematically varying lattice constants along one axis (c) and three axes (a, b, c) from -45% to +45% of optimized values [73].
- Electronic Structure Monitoring: Computing band structures, density of states, and charge density redistribution under applied strain [73].
- Phase Transition Identification: Detecting critical points where materials transitioned between semiconducting, metallic, and topological insulator states [73].
Results revealed that band gap energies for KPbBr₃, RbPbBr₃, and CsPbBr₃ could be systematically tuned through strain engineering, with specific lattice deformations inducing conductor and n-type semiconductor behavior [73]. This demonstrates DFT's unique capability for predicting property modifications under non-equilibrium conditions.
Integrated Workflows: Complementary Methodologies in Practice
Modern computational research increasingly employs hybrid strategies that leverage the complementary strengths of multiple theoretical approaches. The following workflow illustrates a typical integrated computational methodology for materials design:
Integrated Computational Workflow Diagram Title: Multi-Method Research Strategy
This integrated approach is exemplified by orbital-based bonding analysis in solids, where plane-wave DFT calculations are transformed via the LOBSTER package into local orbital representations compatible with MO-style analysis [10]. This workflow enables:
- Initial Conceptualization: Using VB principles to formulate bonding hypotheses based on chemical intuition [69].
- Structure Optimization: Employing DFT for efficient geometry optimization and electron density calculation [10].
- Bonding Analysis: Transforming DFT results into localized orbitals for MO-style bonding indicators like COOP (Crystal Orbital Overlap Population) and bond orders [10].
- Property Prediction: Combining insights from all methods to predict spectroscopic behavior, reactivity, and materials properties [10] [36].
Essential Computational Research Reagents
Table: Key Computational Tools and Their Functions
| Research Tool | Theory Basis | Primary Function | Application Examples |
|---|---|---|---|
| LOBSTER Package | DFT/MO | Orbital-based bonding analysis in solids | COOP, bond orders, population analysis [10] |
| Gaussian | MO/DFT | Molecular quantum chemistry | SCF, CI, CC, DFT calculations [36] |
| MOLPRO | MO/VB | Advanced correlation methods | CASSCF, MRCI, coupled cluster [36] |
| VASP | DFT | Solid-state electronic structure | Plane-wave calculations for periodic systems [73] |
| CASVB | VB/MO | Valence bond analysis | Transforming MO wavefunctions to VB structures [36] |
| Quantum ESPRESSO | DFT | First-principles materials modeling | Nanoscale electronic structure [73] |
The modern computational chemistry landscape reflects a sophisticated division of labor among VB, MO, and DFT methodologies, each offering distinct advantages for specific research challenges. Valence Bond theory maintains its relevance through strong connections to chemical intuition and conceptual clarity in bonding descriptions [8] [69]. Molecular Orbital theory provides the foundational framework for spectroscopic interpretation and systematic quantum chemical calculations [10] [36]. Density Functional Theory delivers practical efficiency for complex systems and solid-state materials [10] [73].
Rather than representing competing methodologies, these theoretical frameworks form a complementary toolkit, with integrated approaches increasingly driving computational innovation. The strategic researcher selects and combines these methods based on the specific research question: VB for bonding insight, MO for spectral prediction and systematic improvement, and DFT for materials design and complex systems. This synergistic integration, guided by the fundamental theoretical relationships between the approaches, continues to expand the frontiers of computational chemistry across molecular science, materials research, and drug development.
Conclusion
The journey from the Heitler-London approach to modern valence bond theory marks a significant renaissance, transforming it from a historically overshadowed model into a sophisticated and computationally powerful framework. The key takeaway is that modern VB theory, when applied at a high level of theory, is mathematically equivalent to MO theory but offers a more intuitive, chemically grounded picture of electron-pair bonds, resonance, and bond formation/breaking. This clarity is particularly valuable for modeling chemical reactions and understanding electronic reorganization. For biomedical and clinical research, the future implications are substantial. Modern VB's ability to accurately describe charge transfer, diradical states, and intricate bonding patterns in transition states can provide deeper insights into enzyme mechanisms, drug-receptor interactions, and the reactivity of novel therapeutic compounds. Its continued development promises to offer unique tools for the rational design of molecules with tailored properties in drug development.
References
- 1. Cubical atom [https://en.wikipedia.org/wiki/Cubical_atom]
- 2. Valence Bond Theory—Its Birth, Struggles with Molecular ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC8001733/]
- 3. Chemical reductionism revisited: Lewis, Pauling and the ... [https://www.sciencedirect.com/science/article/abs/pii/S003936810700088X]
- 4. Lewis Atomic Model (1902 AD) [https://rinconeducativo.org/en/recursos-educativos/lewis-atomic-model-1902-ad/]
- 5. The Cubical Atom [https://www.dailykos.com/stories/2018/3/5/1404649/-The-Cubical-Atom]
- 6. 1.2: Lewis Theory of Bonding [https://chem.libretexts.org/Courses/Douglas_College/DC%3A_Chem_2330_(O'Connor)/1%3A_Introduction_and_Review/1.2%3A_Lewis_Theory_of_Bonding]
- 7. Valence bond theory [https://en.wikipedia.org/wiki/Valence_bond_theory]
- 8. Modern valence bond theory [https://en.wikipedia.org/wiki/Modern_valence_bond_theory]
- 9. Clarifying the quantum mechanical origin of the covalent ... [https://www.nature.com/articles/s41467-020-18670-8]
- 10. Orbital-based bonding analysis in solids - RSC Publishing [https://pubs.rsc.org/en/content/articlehtml/2025/sc/d5sc02936h]
- 11. Bonding to Hydrogen [https://www.americanscientist.org/article/bonding-to-hydrogen]
- 12. 1.10: The Chemical Bond [https://chem.libretexts.org/Bookshelves/Physical_and_Theoretical_Chemistry_Textbook_Maps/Quantum_Chemistry_(Blinder)/01%3A_Chapters/1.10%3A_The_Chemical_Bond]
- 13. 10.4: The Case of H₂ [https://chem.libretexts.org/Bookshelves/Physical_and_Theoretical_Chemistry_Textbook_Maps/Book%3A_Quantum_States_of_Atoms_and_Molecules_(Zielinksi_et_al)/10%3A_Theories_of_Electronic_Molecular_Structure/10.04%3A_The_Case_of_H]
- 14. Precision Spectroscopy of Few-Electron Systems and ... [https://atta.ustc.edu.cn/en-us/research/H2spec.html]
- 15. H 2 system in two different MO basis sets [https://www.sciencedirect.com/science/article/abs/pii/0009261471870495]
- 16. Wave function [https://en.wikipedia.org/wiki/Wave_function]
- 17. 7.2: Wave functions [https://phys.libretexts.org/Bookshelves/University_Physics/University_Physics_(OpenStax)/University_Physics_III_-_Optics_and_Modern_Physics_(OpenStax)/07%3A_Quantum_Mechanics/7.02%3A_Wavefunctions]
- 18. Exchange interaction [https://en.wikipedia.org/wiki/Exchange_interaction]
- 19. quantum chemistry - What is the exchange ? - Chemistry... interaction [https://chemistry.stackexchange.com/questions/61176/what-is-the-exchange-interaction]
- 20. 8.6: Resonance Structures [https://chem.libretexts.org/Bookshelves/General_Chemistry/Map%3A_Chemistry_-_The_Central_Science_(Brown_et_al.)/08%3A_Basic_Concepts_of_Chemical_Bonding/8.06%3A_Resonance_Structures]
- 21. Resonance (chemistry) [https://en.wikipedia.org/wiki/Resonance_(chemistry)]
- 22. Identifying a real space measure of charge-shift bonding with... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11168100/]
- 23. 5.1: Exchange interaction [https://chem.libretexts.org/Bookshelves/Physical_and_Theoretical_Chemistry_Textbook_Maps/Electron_Paramagnetic_Resonance_(Jenschke)/05%3A_Electron-Electron_Interactions/5.01%3A_Exchange_interaction]
- 24. Pauling's Conceptions of Hybridization and Resonance in ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC8303469/]
- 25. Pauling Develops His Theory of the Chemical Bond [https://www.ebsco.com/research-starters/history/pauling-develops-his-theory-chemical-bond]
- 26. 10.7: Valence Bond Theory- Hybridization of Atomic Orbitals [https://chem.libretexts.org/Bookshelves/General_Chemistry/Map%3A_A_Molecular_Approach_(Tro)/10%3A_Chemical_Bonding_II-_Valance_Bond_Theory_and_Molecular_Orbital_Theory/10.07%3A_Valence_Bond_Theory-_Hybridization_of_Atomic_Orbitals]
- 27. Valence Bond ( VB ) Theory - Assignment Point [https://assignmentpoint.com/valence-bond-vb-theory/]
- 28. Valence Bond vs Molecular Orbital Theory: 10... - VIVA DIFFERENCES [https://vivadifferences.com/valence-bond-theory-vs-molecular-orbital-theory-with-examples/]
- 29. Increased valence (qualitative valence bond) descriptions ... [https://www.sciencedirect.com/science/article/abs/pii/S0022113903000125]
- 30. The “Perfect” Form of Heitler-London Theory [https://link.springer.com/chapter/10.1007/978-3-642-93213-7_10]
- 31. : Definition & Equation - GeeksforGeeks Linear Combinations [https://www.geeksforgeeks.org/engineering-mathematics/linear-combinations/]
- 32. Valence Bond Theory—Its Birth, Struggles with Molecular ... [https://www.mdpi.com/1420-3049/26/6/1624?type=check_update&version=2]
- 33. A Density Functional Valence Bond Study on the Excited ... [https://www.mdpi.com/1420-3049/30/3/489]
- 34. Screening in the Heitler-London Model: Revisiting ... [https://arxiv.org/html/2511.01132v1]
- 35. Heitler-london Model Revisiting: Screening Effects In ... [https://quantumzeitgeist.com/variational-quantum-heitler-london-model-revisiting-screening-effects-hydrogen-molecules/]
- 36. The Electronic Structure and Bonding in Some Small Molecules [https://pmc.ncbi.nlm.nih.gov/articles/PMC11902227/]
- 37. 1. What is Valence Bond (VB) Theory? [http://xmvb.xmu.edu.cn/xmvb-course-en/chapt1.html]
- 38. Valence Bond Methods - an overview [https://www.sciencedirect.com/topics/chemistry/valence-bond-methods]
- 39. Valence Bond Insights into the H-Abstraction Barrier in ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12114163/]
- 40. The Important Features of The Heitler | PDF [https://www.scribd.com/document/462153637/The-important-features-of-the-Heitler]
- 41. Chemical Bonding Concepts from Quantum Information ... [https://arxiv.org/html/2501.15699v1]
- 42. Aromaticity and its chemical manifestations [https://www.sciencedirect.com/science/article/pii/S1380732399800211]
- 43. 4.1: Introduction - Chemistry LibreTexts [https://chem.libretexts.org/Bookshelves/General_Chemistry/Book%3A_Structure_and_Reactivity_in_Organic_Biological_and_Inorganic_Chemistry_(Schaller)/V%3A__Reactivity_in_Organic_Biological_and_Inorganic_Chemistry_3/04%3A_Oxygen_Binding_and_Reduction/4.01%3A_Introduction]
- 44. Triplet oxygen [https://en.wikipedia.org/wiki/Triplet_oxygen]
- 45. Why is oxygen in a triplet state and what are the ... [https://physics.stackexchange.com/questions/21577/why-is-oxygen-in-a-triplet-state-and-what-are-the-consequences]
- 46. Valence Bond Theory [http://lampz.tugraz.at/~hadley/ss1/molecules/valencebond.php]
- 47. Marcus kinetics control singlet and triplet oxygen evolving ... [https://www.nature.com/articles/s41586-025-09587-7]
- 48. Enhancement of photoinduced reactive oxygen species ... [https://pubs.rsc.org/en/content/articlehtml/2025/sc/d4sc05428h]
- 49. Triplet Imaging of Oxygen Consumption during the ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC2808489/]
- 50. Direct Exchange Interaction in Heitler-London Model [https://kk-sinha.blogspot.com/2019/01/direct-exchange-interaction-in-heitler.html]
- 51. Are the bonding orbitals in methane equivalent [https://chemistry.stackexchange.com/questions/32038/are-the-bonding-orbitals-in-methane-equivalent-photoelectron-spectrum]
- 52. Rethinking Hybridization [https://chem.libretexts.org/Courses/BethuneCookman_University/BCU%3A_CH_332_Physical_Chemistry_II/AGENDA/Rethinking_Hybridization]
- 53. 11.2: Introduction to the Valence-Bond Method [https://chem.libretexts.org/Bookshelves/General_Chemistry/Map%3A_General_Chemistry_(Petrucci_et_al.)/11%3A_Chemical_Bonding_II%3A_Additional_Aspects/11.2%3A_Introduction_to_the_Valence-Bond_Method]
- 54. A Critical Look at Linus Pauling's Influence on the ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC8348226/]
- 55. Breathing-orbital valence bond method – a modern ... [https://link.springer.com/article/10.1007/s00214-002-0364-8]
- 56. 9.1: Homolytic and Heterolytic Cleavage [https://chem.libretexts.org/Bookshelves/Organic_Chemistry/Organic_Chemistry_I_(Liu)/09%3A_Free_Radical_Substitution_Reaction_of_Alkanes/9.01%3A_Homolytic_and_Heterolytic_Cleavage]
- 57. Bond-dissociation energy [https://en.wikipedia.org/wiki/Bond-dissociation_energy]
- 58. Bond-Dissociation Energies (BDEs) - Rowan Scientific [https://www.rowansci.com/tools/bond-dissociation-energies]
- 59. NHC induced radical formation via homolytic cleavage of B–B ... [https://pubs.rsc.org/en/content/articlelanding/2022/sc/d2sc02096c]
- 60. The Origin, Development and Significance of the Heitler- ... [https://link.springer.com/chapter/10.1007/978-94-009-0949-6_8]
- 61. What is actually the difference between valence bond ... [https://chemistry.stackexchange.com/questions/33879/what-is-actually-the-difference-between-valence-bond-theory-and-molecular-orbita]
- 62. The Valence - Bond to Approach in Complexes Bonding [https://chemed.chem.purdue.edu/genchem/topicreview/bp/ch12/valence.php]
- 63. Chemical bonding - Molecular Orbitals, H2, He2 [https://www.britannica.com/science/chemical-bonding/Molecular-orbitals-of-H2-and-He2]
- 64. Section 4.2.2: Homonuclear Diatomic Molecules [https://chem.libretexts.org/Courses/Centre_College/CHE_332%3A_Inorganic_Chemistry/04%3A_Molecular_Orbitals/4.02%3A_Molecular_Orbital_Theory_of_Diatomic_Molecules/4.2.02%3A_Homonuclear_Diatomic_Molecules]
- 65. 3.3: Valence Bond Theory [https://chem.libretexts.org/Courses/Smith_College/CHM_118%3A_Advanced_General_Chemistry_(Strom)/03%3A_Advanced_Theories_of_Covalent_Bonding/3.03%3A_Valence_Bond_Theory]
- 66. Valence Bond Calculation - an overview [https://www.sciencedirect.com/topics/chemistry/valence-bond-calculation]
- 67. [2511.01132] Screening in the Heitler-London Model [https://arxiv.org/abs/2511.01132]
- 68. The Quantum Paradox in Pharmaceutical Science [https://pmc.ncbi.nlm.nih.gov/articles/PMC12111262/]
- 69. The Valence-Bond (VB) Model and Its Intimate ... [https://www.mdpi.com/1420-3049/26/15/4524]
- 70. Valence bond theory a re-examination of concepts and ... [https://www.sciencedirect.com/science/chapter/bookseries/abs/pii/S1380732399800156]
- 71. Photo-Fenton degradation on Mo-doped NiFe 2 O 4 ... [https://pubs.rsc.org/en/content/articlehtml/2025/dt/d4dt03359k]
- 72. Dual Mo‐Doping in BiVO4/FeCoNiOx Photoanode Enables ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12533305/]
- 73. A DFT study of the effect of strain on the structural and ... [https://www.nature.com/articles/s41598-025-11320-3]